| Table of Contents |  |
|
Case Report
| ||||||
| Hysteroscopically-guided subchorionic methotrexate administration: A new technique for termination of cesarean scar pregnancy | ||||||
| Giancarlo Garuti1, Stefania Calabrese1, Lorenzo Quirino1, Marco Di Mario1 | ||||||
|
1MD, Obstetrics and Gynecology Department, Public Hospital of Lodi, Via Savoia 1, 26900-Lodi, Italy.
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article: |
| Garuti G, Calabrese S, Quirino L, Mario MD. Hysteroscopically-guided subchorionic methotrexate administration: A new technique for termination of cesarean scar pregnancy. J Case Rep Images Gynecol Obstet 2016;2:1–6. |
|
Abstract
|
|
Introduction:
Cesarean scar pregnancy (CSP) is a rare but life-threatening condition, due to the risk of uterine rupture or for the development of placenta previa/accreta. The mainstay of management to spare fertility is early diagnosis and pregnancy termination. Although several conservative treatments have been experienced, no consensus on the best therapeutic approach is currently shared. The medical therapy for CSP termination is based on systemic or local Methotrexate (MTX) administration. Hysteroscopy allows the precise visualization of placental implantation and it can be used to drive MTX injection selectively within the intervillous placental spaces. This procedure increases drug concentration delivered to the target tissue, potentially improving its therapeutic index.
Case Report: A 36-year-old female with a history of cesarean section was admitted to our department with a diagnosis of viable 7th weeks CSP. The patient gave her consent to pregnancy termination by a hysteroscopically-guided MTX administration within the cervico-isthmic placental implantation site, followed by its resectoscopic removal. In an office setting, 80 mg of MTX were injected through a needle adaptable to the 5-Fr operative channel of hysteroscope, under the chorionic membrane of placental implantation site. The duration of the procedure took three minutes and it resulted easy, painless and uneventful. An early embryo demise was obtained; the human chorionic gonadotropin-beta subunit (beta-hCG) serum trend showed a rapid pregnancy termination and no patient complaints were recorded. After 28 days, a resectoscopic removal of the gestational sac and placenta was safely accomplished. Conclusion: Selective hysteroscopic administration of MTX within intervillous spaces of the ectopic placental implantation is effective and it can be considered for CSP termination. | |
|
Keywords:
Cesarean scar pregnancy, Ectopic pregnancy, Hysteroscopy, Methotrexate, Office hysteroscopy, Resectoscopy
| |
|
Introduction
| ||||||
|
Cesarean scar pregnancy (CSP) is a rare but potentially life-threatening condition, caused by a placental implantation within a retracted scar of cervico-isthmic uterine junction, deriving from a previous Cesarean Delivery (CD) [1]. Firstly described in 1978, CSP was more frequently reported in the last decades, due to the increasing rates of CD. In patients with history of CD, the CSP incidence varies from 1:500 to 1:1500 [1] [2][3]. If untreated, CSP exposes the patient to the risk of uterine rupture and hemorrhage during the first pregnancy months or to the development of a placenta previa/accreta near pregnancy termination [4] [5]. The mainstay management aimed to spare fertility and to reduce maternal morbidity, is an early diagnosis and pregnancy termination [6]. The current treatment of CSP is based on case reports and small case series, with more than 30 therapeutic options described [1] [3]. Even if recent studies compared different managements of CSP, no guidelines have been given until now [7] [8]. The dihydrofolate reductase (DHFR) enzyme inhibitor, Methotrexate (MTX), administered by systemic, loco-regional or intragestational-sac routes, is the cornerstone of medical therapy, followed or not by surgical removal [1] [3][6][9]. Due to the reversible and competitive nature of MTX binding to DHFR, drug concentration within target-cell is a major determinant of cytotoxicity [10]. Among the surgical techniques suggested for CSP removal (uterine dilatation and curettage, laparotomy, laparoscopy, vaginal excision and hysteroscopy), hysteroscopy resulted to be effective and showed a low morbidity rate [3] [11][12]. Based on the knowledge of MTX pharmacokinetics [10] and assuming that its selective administration within the placental intervillous spaces can enhance its cytocidal effects, we report the case of a patient with a viable CSP managed by hysteroscopically-guided sub-chorionic MTX administration, delivered at the placental implantation. The procedure was completed by the resectoscopic removal of the terminated pregnancy. | ||||||
|
Case Report
| ||||||
|
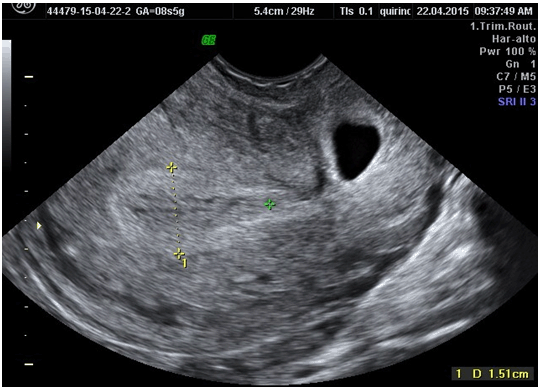
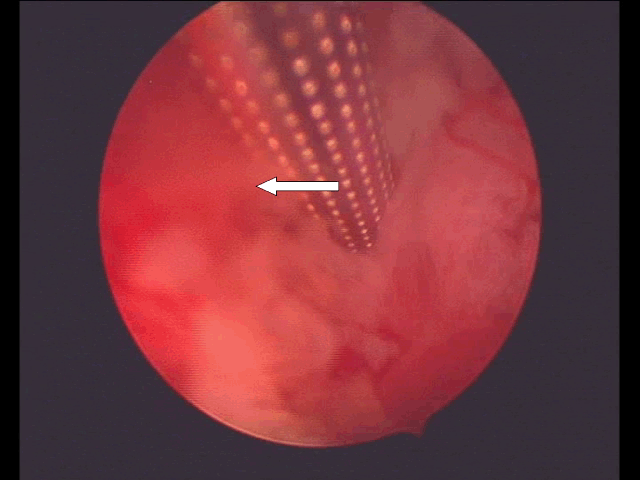
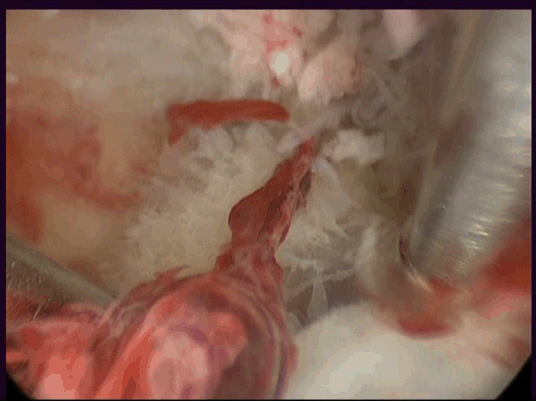
In April, 2015, a 36-year-old female with a positive urine pregnancy test and 8 weeks of amenorrhea, was admitted to our Department due to pelvic cramping pain and ultrasound evidences of a viable CSP. No gynecological complaints were recorded; her past obstetric history revealed a CD due to breech presentation and a vaginal delivery 11 and 3 years before, respectively. Pelvic examination detected a movable uterus with a smooth, painless and soft mass measuring about 2.5 cm, arising from the cervico-isthmic area and expanding towards the bladder base. Serum beta-subunit of human Chorionic gonadotropin (beta-hCG) concentration was of 18000 mUI/ml. Transvaginal ultrasonography revealed empty endometrial cavity and empty cervical canal, the gestational sac measuring 17x14 mm was embedded deep within the uterine wall at the level of the cervico-isthmic junction, bulging ventrally towards the bladder (Figure 1). It contained an embryonic pole showing heart activity and measuring 4.6 mm, corresponding to 7th pregnancy weeks (Figure 2). The measurement of myometrial thickness between gestational sac and bladder base was 1.5 mm. Color-Doppler and spectral analysis showed a high blood-flow with low resistances around the cervico-isthmic area. Based on these findings and according to published data, a diagnosis of viable CSP was made [6]. With the assumption that pregnancy termination was indicated and no guidelines were provided to treat CSP, we proposed to the patient local MTX administration through hysteroscopy guidance, followed by the resectoscopic removal of pregnancy. The patient signed an informed consent and an informative chart about the risk of hemorrhage. The first procedure was carried out in an office setting. By the vaginoscopic technique without analgesia or anesthesia, a double-flow 5-mm hysteroscope (Karl Storz, Tuttlingen, Germany) was used to administer 50 mg/m2 of MTX in 2 cc of saline, through a 17-gauge needle adaptable to the 5Fr operative channel. Continuous saline flow was delivered at working pressure set at 60 mm/Hg by an electronic irrigation-suction device. After the visual confirmation of a ventral uterine pregnancy implantation within the cesarean scar, we entered the chorionic sac by using hysteroscopy scissors, opening sequentially capsular decidua, chorion leave and chorionic membrane. After needle insertion within the operative channel, MTX has been injected 2–3 mm deep the chorionic membrane at the level of anterior implantation of the placenta, i.e. directly within the placental intervillous spaces (Figure 3). The procedure lasted three minutes, it was painless, and the patient was discharged after two hours of uneventful observation. Clinical, sonographic and beta-hCG monitoring were recommended after 4, 7, 14 and 21 days from MTX administration. During the follow-up period a light vaginal bleeding was the only complaint recorded. No embryonic heart activity was found after four days while the persistence of gestational sac within the cesarean scar was found until the 21st day. Beta-hCG increased to 29000 mUI/ml after four days, decreasing to 18000 mUI/ml, 5000 mUI/ml and 3000 mUI/ml after 7, 14 and 21 days, respectively. After 28 days, observing a further drop of beta-hCG to 500 mUI/ml, the hysteroscopy removal of CSP was planned. The intervention was carried-out under conscious sedation, using a 27Fr resectoscope armed with a 4 mm bipolar loop (Versapoint Bipolar System, Gynecare, Ethicon Inc., Menlo Park, CA, USA) set at 100 W power. Due to the soft consistency of the cervix, its dilatation was avoided. Saline solution was delivered as uterine distension medium at 60 mm/Hg working pressure. After CSP identification (Figure 4) and (Figure 5) the intervention was almost entirely managed by using the cold loop to separate villous trophoblast from the cervico-isthmic niche, following a loose cleavage plane maintained between tissues (Figure 6). The application of coagulating current for bleeding control was not necessary. The intervention lasted for eight minutes, an uneventful recovery followed and the patient was discharged on the first postoperative day. Villous trophoblast and embryonic tissues were found at pathology assessment. After intervention, beta-hCG decreased to a non-pregnancy range after 15 days. The first menstrual period was recorded after 32 days and no abnormalities were found both at physical and ultrasound examinations after 40 days from intervention. | ||||||
| ||||||
| ||||||
|
| ||||||
|
| ||||||
| ||||||
| ||||||
|
Discussion
| ||||||
|
The main metabolic pathway leading to MTX cytotoxicity is represented by a tight but reversible inhibition of DHFR. The DHFR enzyme plays a key role in maintaining intracellular folate homeostasis and it is responsible for the conversion of dihydrofolate substrates to tetrahydrofolates. Tetrahydrofolates play an essential role in synthesis of DNA and RNA precursors such as purine and pyrimidine rings. With respect to natural dihydrofolate substrates, the competitive reversible nature of the MTX binding to DHFR, leads to two critical determinants of cytotoxicity: drug cell concentration and duration of drug cell exposure [10]. These pharmacokinetic principles, combined with the short drug half-life and the poor blood supply to scar tissue surrounding a CSP, are the main causes of the low therapeutic index associated with systemic MTX administration [1] [3][10]. Driving MTX directly to the target tissue may lead to an excess of free drug cell concentration required to fully inhibit the enzyme, potentially enhancing its clinical effectiveness. Accordingly, ultrasound-guided MTX administration within the gestational sac showed an improvement of clinical results with respect to systemic MTX and it is now considered the first choice for CSP conservative treatment [3][4] [13][14]. However, as reported in 26–39% of cases [13][14] pregnancy absorption takes long time and possible failures require additional treatments. The target tissue of MTX is the villous trophoblast of the blastocyst implantation site, leading to the placental differentiation within the cesarean scar niche. The placental growth inhibition leads to pregnancy termination and it represents the key measure to reduce the risks of uterine wall rupture and hemorrhage. Previous reports related to hystero-embryoscopy settings showed that miniaturized hysteroscopes enable an easy access to the gestational sac [15]. Based on these assumptions, we believed that MTX administration in subchorionic space at placental implantation site (i.e., directly within the intervillous spaces), may optimize the cytotoxicity, enhancing the drug concentrations delivered to the target tissues. In the case here presented, hysteroscopy easily assessed the CSP anatomy and it allowed a selective MTX administration to the underlying villous trophoblast by identifying the placental implantation site. With regard to hysteroscopy guidance, ultrasound techniques of intra-gestational and/or peri-gestational sac MTX administration may be less specific in driving selectively the drug. This is mainly due to the possible difficulty in positioning precisely the needle tip within the placental implantation site [3] [13]. Subchorionic hysteroscopic MTX administration resulted technically easy, quick, painless, safe and effective; moreover, it can be accomplished as outpatient procedure. The effectiveness of this technique was demonstrated by the early loss of embryonic cardiac activity and the early increase of beta-hCG (due to trophoblast cells necrosis) followed by its fast and progressive fall [6] [13]. In current literature, only one report describes a hysteroscopy-guided MTX administration for treatment of viable CSP. After systemic MTX failure and under hysteroscopy guidance the Authors injected MTX within the gestational sac, obtaining an embryo's demise but observing a persistent increase of beta-hCG levels [16]. Although no guidelines provided preferential options in CSP management, the recent literature suggests a combined sequential approach. The first measure is aimed to pregnancy termination by MTX administration or by acute reduction of placental blood supply by uterine artery embolization. Subsequently, an appropriate timing for surgical pregnancy removal is advised [7][8][9] [17]. Based on literature data, the hysteroscopic CSP removal appears to be the safest surgical therapy. Hysteroscopy allows a precise assessment of pregnancy topography and placental implantation, a bleeding control by use of coagulating current and a selective pregnancy removal under vision [3] [8][11][12][16][17]. As previously reported, based on the significant decrease of beta-hCG levels after MTX administration and demonstrating the pregnancy termination, we safely removed the CSP by resectoscope, accordingly [11][12][16] . | ||||||
|
Conclusion
| ||||||
|
A successful and never described technique to manage a viable CSP by the selective administration of MTX under hysteroscopy guidance, within the placental intervillous space of the ectopic implantation, has been reported. An eye-driven MTX administration within the target tissue, leading to high drug cell concentrations, may optimize its cytotoxicity. When hysteroscopy facilities are available, this technique can be considered as an option for CSP termination. | ||||||
|
Acknowledgements
| ||||||
|
We are thankful to Mrs. Elena Bosoni for language revision of the Manuscript. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Giancarlo Garuti – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Stefania Calabrese – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Lorenzo Quirino – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Marco Di Mario – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2016 Giancarlo Garutid et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|