|
Case Report
Prenatal diagnosis of a fetal inferior vena cava thrombus
1 Medical Student, Medical College of Wisconsin, Milwaukee, Wisconsin, United States
2 Physician Fellow, Division of Maternal Fetal Medicine, Department of Obstetrics and Gynecology, Medical College of Wisconsin, Milwaukee, Wisconsin, United States
3 Sonographer, Maternal Fetal Care Center, Froedtert Hospital, Milwaukee, Wisconsin, United States
4 Attending Physician, Division of Maternal Fetal Medicine, Department of Obstetrics and Gynecology, Medical College of Wisconsin, Milwaukee, Wisconsin, United States
Address correspondence to:
Emily Fahl
Medical College of Wisconsin, 8701 West Watertown Plank Road, Milwaukee, Wisconsin 53226,
United States
Message to Corresponding Author
Article ID: 100114Z08EF2022
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Fahl E, Livergood MC, Penzkover D, Peterson E. Prenatal diagnosis of a fetal inferior vena cava thrombus. J Case Rep Images Obstet Gynecol 2022;8: 100114Z08EF2022.ABSTRACT
Introduction: Diagnosis and management of fetal thrombosis during pregnancy is rare. Few cases of prenatally diagnosed inferior vena cava thrombi are reported and often occur with renal vein thrombi. Several maternal and fetal risk factors have been identified, including thrombophilias.
Case Report: Patient is a 39-year-old G6P2123 with heterozygous Factor V Leiden with a prenatally diagnosed fetal inferior vena cava thrombus at 34 weeks of gestation during antenatal ultrasound. Her peripartum course was uncomplicated. On day 1 of life, neonatal ultrasound and computed tomography confirmed the presence of thrombus. On day 7, the neonate underwent a thrombectomy for definitive management due to risk of embolization.
Conclusion: While ultrasound is not routinely used for screening for fetal thromboses, when thrombosis is diagnosed, antenatal management is complex, weighing fetal and maternal risks and benefits. Thrombectomy as definitive thrombus management is an appropriate treatment when risk of embolization exists. Imaging modalities are useful for both diagnosis and management of fetal thrombi.
Keywords: Fetal thrombus, Fetal ultrasound, Prenatal diagnosis, Prenatal ultrasound
Introduction
While Factor V Leiden (FVL) is a common thrombophilia for pregnant patients, diagnosis and management of a fetal thrombosis during pregnancy is rare. Despite the increased risk of venous thromboembolism (VTE) in the pregnant patient due to increased clotting potential in normal pregnancy as well as increased risk for patients with FVL, it is unclear if inherited thrombophilias cause adverse pregnancy outcomes such as fetal loss, fetal growth restriction, preeclampsia, or placental abruption [1]. Cases of fetal thrombosis diagnosed in the prenatal period are limited and the risk of a VTE in the fetus is undetermined. When neonates are diagnosed with a thrombus, it is unknown if these clots began developing in utero. While neonates have been described as hypercoagulable, especially in stressful conditions, the coagulation system in the fetus is less well known, but it is believed that it develops toward that of a healthy newborn [2],[3].
This case report aims to explain the diagnosis and management of prenatally diagnosed fetal thrombosis of the inferior vena cava (IVC) as well as to review the current literature.
Case Report
Our patient is a 39-year-old female who presented with an obstetric history of six total pregnancies, including the pregnancy discussed in this case, two prior term deliveries, one prior preterm delivery, two prior spontaneous abortions, with three living children (gravida (G) 6, para (P) 2123). Her medical history was significant for heterozygous FVL. At the age of 28, our patient had a deep vein thrombosis (DVT). In her preceding pregnancy three years prior, delivery was necessitated due to placental abruption at 33 weeks of gestation. Due to a chronic subchorionic hemorrhage, she was not on anticoagulation during that pregnancy. On day 14 of life, that neonate had a pulmonary embolism (PE). This prompted her husband to be tested for thrombophilia and he was found to also be heterozygous for FVL. The newborn consequently tested as homozygous FVL.
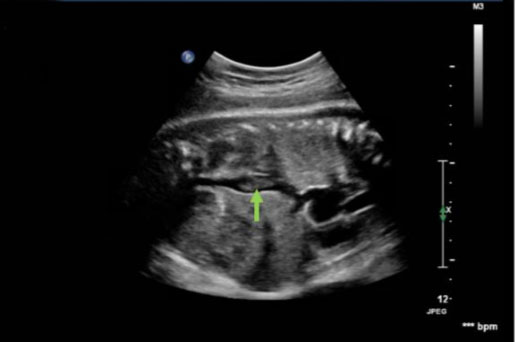
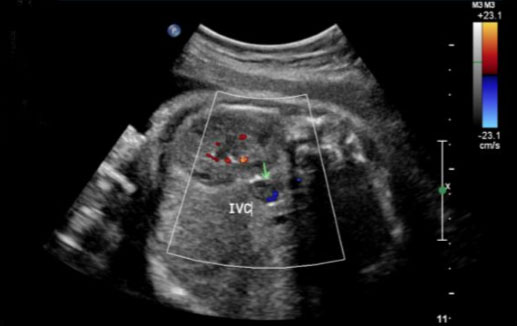
During this pregnancy, our patient was on 40 mg subcutaneous low molecular weight heparin (LMWH) daily for VTE prophylaxis due to her history of DVT and low-risk thrombophilia as well as 81 mg aspirin daily. Her first and second trimesters were uncomplicated. The patient began antenatal testing at 32 weeks of gestation, consisting of fetal heart rate monitoring and ultrasounds. Weekly biophysical profiles (BPP) were reassuring. At 32 weeks of gestation, growth ultrasound was normal, showing an estimated fetal weight of 2688 g. At 34 weeks and 3 days of gestation, her BPP was normal; however, the ultrasound showed a finding concerning for a fetal thrombosis in the IVC (Figure 1, Figure 2, Figure 3).
The patient was admitted to the hospital, given a dose of betamethasone, placed on continuous fetal monitoring, and her LMWH was held. A fetal echocardiogram was then conducted and showed an echobright mass in the fetal IVC, concerning for possible organized thrombus measuring 1.09 cm × 0.94 cm. The clot measured about the size of the pulmonary valve and was thought to be unlikely to cross the pulmonary valve if it embolized. There were no cardiac structural abnormalities identified. Approximately 24 hours after her first dose of betamethasone, fetal monitoring showed late decelerations, indicating uteroplacental insufficiency. The patient was counseled that the benefit of delivery outweighed the risk of continuing the pregnancy and the patient consequently underwent a repeat cesarean section (CS).
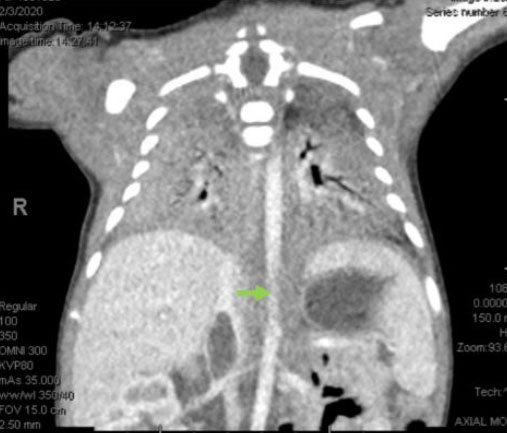
There were no maternal complications with delivery. The neonate weighed 2.825 kg and APGARs were 9 at 1 minute and 9 at 5 minutes; however, required 6 liters of supplemental oxygen through continuous positive airway pressure and then nasal canula. Placental pathology demonstrated a 381 g placenta with a three-vessel umbilical cord with no significant lesions. It also showed fetal membranes with thrombosis of maternal decidual capillaries and atherosis and fibrinoid necrosis of maternal decidual arterioles, consistent with maternal vascular malperfusion. There was also fibrinoid change of the fetal vessel, rare thrombi in the fetal vessel, and villitis of uncertain etiology. A disseminated intravascular coagulation panel showed a normal prothrombin time of 16.9 seconds, an internal normalized ratio of 1.32, and a partial thromboplastin time of 43.9 seconds. Both the D-dimer and fibrinogen were abnormal, with an elevated D-dimer at 9.72 ug/mL and a low fibrinogen at 123 mg/dL [4]. On the day of delivery, an abdominal ultrasound showed a nonocclusive thrombus measuring 2.2 cm by 1.2 cm by 0.8 cm within the suprarenal IVC with a mildly hypoechoic center and peripheral hyperechogenicity. The remaining portions of the IVC, renal veins, and common iliac veins were patent and free of thrombus. A head ultrasound showed no evidence of an intraventricular hemorrhage and renal vein Doppler showed patency of both renal veins. A therapeutic heparin drip was started and prostaglandin (PGE) was given to maintain a patent ductus arteriosus in event of a pulmonary embolism (PE).
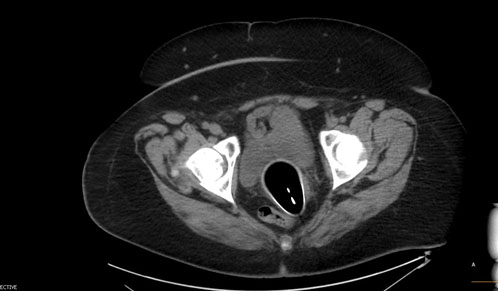
On post-op day 1, the mother restarted her LMWH and was recovering well. On day 1 of life, the neonate underwent an ultrasound showing a similar appearing focal non-occlusive clot. On day 2 of life, there were no intracardiac clots visualized on the echocardiogram and PGE was stopped. On post-op day 3, the mother was discharged home. On day 3 of life, a computed tomography (CT) angiograph was performed and it was determined that the clot seemed to be recently formed, was deforming the IVC, and was not well attached to the vessel wall because there was blood flow around it, thus raising concern for embolization risk (Figure 4).
The decision was made to undergo a thrombectomy as definitive treatment on day 7 of life when the neonate’s brain would be more mature and there would be a decreased risk of intraventricular hemorrhage from bypass. The procedure was successful. The pathology report of the thrombus demonstrated a histiocytic/macrophage reaction and focal calcification. Of note, the neonate was found to be heterozygous FVL. The mother and infant were healthy at her six week postpartum visit.
Discussion
In a review of the literature, reported cases of prenatally diagnosed thrombosis have variable presentations and etiologies. Majority of the cases include thrombosis of the inferior vena cava and/or renal vein thrombosis and less commonly intracranial thrombosis or portal vein thrombosis. The maternal risk factors associated with fetal thromboses include, but are not limited to, diabetes (type I, type II, or gestational), hypertension, preeclampsia, systemic lupus erythematosus, antiphospholipid syndrome, thrombophilias, dehydration, acute pyelonephritis, acute gastroenteritis, cytomegaly virus infection, and corticosteroid use [2],[3],[5],[6]. The fetal risk factors associated with thromboses include anatomical malformations or variations, thrombophilias, and hypoxemia. Presentation of the thromboses also differ; some are found incidentally on ultrasound, as in our case, while others present as hydrops fetalis, polyhydramnios, ascites, pleural effusions, cardiomegaly, reduced fetal movement, or fetal distress on fetal heart monitoring [3],[5],[6]. Majority of the thrombi were found in the third trimester. Thromboses diagnosed in the postpartum neonatal period are more common and better documented; however, it is unknown how many thromboses started developing in the prenatal period [2],[3],[6],[7].
We found few other reported cases of prenatally diagnosed IVC thromboses. In a case reported by Boussicault et al., a fetal IVC thrombus was found incidentally on ultrasound at 31 weeks of gestation and managed expectantly. After delivery via cesarean section (CS) at 39 weeks of gestation, the infant was healthy and all labs were normal. Computed tomography angiography was done on day 12 of life and showed a calcified thrombus. The neonate was observed and there was no anticoagulation, thrombolysis, or surgery used for treatment [8]. Another case reported by Bakkum and co-authors discussed a 45 year old G7P5 who was taking losartan and found to have oligohydramnios at 27 weeks of gestation. After stopping the losartan, she underwent a transabdominal amnioinfusion to achieve a normal amniotic fluid index. At 32 weeks of gestation, a thrombus in the IVC was found, possibly suggesting a hypercoagulable state after withdrawal of losartan. After expectant management followed by labor induction and delivery at 38 weeks of gestation, the infant was healthy and asymptomatic, and no further treatment was pursued [9]. Weissman-Brenner et al. described a 32 weeks of gestation ultrasound showing hydrops fetalis in a 43-year-old G4P2 with gestational diabetes. After a report of decreased fetal movement followed by a diagnosis of preeclampsia and late decelerations on fetal monitoring, a CS was performed. In the first day of life, the infant was found to have an IVC thrombus on an abdominal ultrasound, thrombocytopenia, and decreased anti-thrombin III levels. The neonate received a platelet transfusion and was placed on LMWH for 14 days. Ultrasound on day 19 of life showed complete resolution of the thrombus [5].
In several instances, a renal vein thrombus was found at the same time as IVC thrombus, however, most often postpartum with the suspicion of the clot forming prenatally. In a case reported by Weber et al., a 32-year-old G4P1 with a dichorionic-diamniotic twin pregnancy had an ultrasound at 29 weeks of gestation that showed one fetus with bilateral enlarged kidneys, mild ascites, and mild pleural effusions. The patient underwent a CS and delivered two males. On day 1 of life, a bilateral renal vein thrombus expanding into the IVC was confirmed in one neonate via ultrasound and the infant was thrombocytopenic. The neonate was then started on tissue plasminogen activator and unfractionated heparin (UFH). Workup revealed a heterozygous FVL mutation in the infant, like our case, however it was de novo [6]. Similarly, Smorgick and colleagues reported a case in a healthy 25-year-old female at 27 weeks of gestation who presented with decreased fetal movement. An ultrasound showed polyhydramnios and a thrombus in the fetal bladder. Due to late decelerations and abnormal venous Doppler waveforms, the infant was delivered by CS, was anemic and thrombocytopenic, and ultrasound on day 1 of life showed a left renal vein thrombus. The neonate died on day 10 of life and on postmortem examination, there were bilateral renal vein thrombi extending into the IVC. It was assumed that these thrombi formed prenatally and that the clot in the bladder was thought to have originated in the renal veins [3].
Although still rare, prenatally diagnosed renal vein thromboses are the most common fetal thrombosis. A case report and literature analysis by Moaddab and co-authors explored 23 cases of prenatally diagnosed renal vein thromboses. The most common ultrasound finding was an enlarged kidney; polyhydramnios or hydrops fetalis were also common presentations. One case had associated maternal thrombophilias including deficiencies of protein C, protein S, and antithrombin III. Another case had one infant with FVL and methylene tetrahydrofolate reductase gene mutation. A different case had a paternal 4G PA1 gene mutation. Five of the cases received thrombolytic or anticoagulation treatment after birth and the others were observed and provided supportive treatment [2]. In a review conducted by Winyard et al., 44% of perinatal renal venous thromboses were associated with thrombotic abnormalities, FVL being the most common [7].
As our case and the previous cases suggest, the cause of fetal thromboses is likely multifactorial. There are also several approaches to management, ultimately decided by what is safest for the pregnant patient and the affected fetus. Interestingly, in our case, delivery was necessitated due to non-reassuring fetal testing, and this had been described as an indication for delivery in other cases with similar outcomes. Similar to our case with a diagnosis of a thrombus and delivery during the third trimester at 34 weeks, all other cases had diagnoses and deliveries during the third trimester. Diagnosis most likely occurs in the third trimester due to indicated ultrasound imaging conducted during that period, with the thrombi diagnosed incidentally or as a result of abnormal imaging findings. Timing of delivery is determined by the risks and benefits to the infant and the pregnant patient. All the cases we discussed managed the thrombi in the neonatal period. Majority of the cases were managed conservatively through observation or through medication, including thrombolytics or anticoagulants. In our particular case, thrombectomy was pursued due to the threat of embolization of the thrombus; the benefits of this treatment outweighed the risk of embolization. Thus, consideration of this risk based on size and attachment of the thrombus is necessary when determining treatment plans of fetal thrombi. Of the thrombi that were truly diagnosed prenatally, ultrasound was the main method of diagnosis, highlighting that thrombi can be visualized on prenatal ultrasound. Both ultrasound and CT were used in the neonatal period to monitor progression or changes of the thrombi. Therefore, fetal and neonatal imaging, namely ultrasound and CT, have utility in both diagnosis and management of thrombi.
Conclusion
Our case demonstrates inherited thrombophilias, namely Factor V Leiden, as an association with fetal thrombosis. Although the thrombus was found incidentally on ultrasound, delivery was necessitated by fetal distress, as seen in other cases. This case was uniquely treated by thrombectomy in the neonatal period due to risk of embolization, emphasizing the importance of size and location of the thrombus and weighing the benefits and risks of the procedure. Both prenatal and neonatal monitoring were facilitated by the use of imaging, highlighting the importance of utilizing these tools for both diagnosis and management of thrombi.
REFERENCES
1.
American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins–Obstetrics. ACOG practice bulletin no. 197: Inherited thrombophilias in pregnancy. Obstet Gynecol 2018;132(1):e18–34. [CrossRef]
[Pubmed]

2.
Moaddab A, Shamshirsaz AA, Ruano R, Salmanian B, Lee W, Belfort MA, Espinoza J. Prenatal diagnosis of renal vein thrombosis: A case report and literature review. Fetal Diagn Ther 2016;39(3):228–33. [CrossRef]
[Pubmed]

3.
Smorgick N, Herman A, Wiener Y, Halperin R, Sherman D. Prenatal thrombosis of the inferior vena cava and the renal veins. Prenat Diagn 2007;27(7):603–7. [CrossRef]
[Pubmed]

5.
Weissmann-Brenner A, Ferber A, O’Reilly-Green C, Avila C, Grassi A, Divon MY. Inferior vena cava thrombosis presenting as non-immune hydrops in the fetus of a woman with diabetes. Ultrasound Obstet Gynecol 2004;23(2):194–7. [CrossRef]
[Pubmed]

6.
Weber SK, Müller A, Geipel A, Berg C, Gembruch U. Prenatal renal vein thrombosis in dichorionic twin pregnancy. Arch Gynecol Obstet 2014;290(3):587–90. [CrossRef]
[Pubmed]

7.
Winyard PJD, Bharucha T, De Bruyn R, et al. Perinatal renal venous thrombosis: Presenting renal length predicts outcome. Arch Dis Child Fetal Neonatal Ed 2006;91(4):F273–8. [CrossRef]
[Pubmed]

8.
Boussicault G, Sentilhes L, Cipierre C, Lépinard C, Bouderlique C. Successful outcome of an idiopathic thrombosis of the fetal inferior vena cava. Ultrasound Obstet Gynecol 2009;33(5):611–2. [CrossRef]
[Pubmed]

9.
Bakkum JN, Brost BC, Johansen KL, Johnston BW, Watson WJ. In utero losartan withdrawal and subsequent development of fetal inferior vena cava thrombosis. Obstet Gynecol 2006;108(3 Pt 2):739–40. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Emily Fahl - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Mary Christine Livergood - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Deborah Penzkover - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Erika Peterson - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2022 Emily Fahl et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.