|
Case Report
Preterm delivery in a 33-year-old pregnant woman with monkeypox virus infection
1 Resident Physician, OB-GYN, Downstate Health Sciences University, Brooklyn, NY, USA
2 Attending Physician, OB-GYN, Downstate Health Sciences University, Brooklyn, NY, USA
3 Attending Physician, OB-GYN, Kings County Hospital Center, Brooklyn, NY, USA
Address correspondence to:
Nawras Zayat
450 Clarkson Ave, Brooklyn, NY 11203,
USA
Message to Corresponding Author
Article ID: 100152Z08NZ2023
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Zayat N, Noumi R, Akkary W. Preterm delivery in a 33-year-old pregnant woman with monkeypox virus infection. J Case Rep Images Obstet Gynecol 2023;9(1):79–82.ABSTRACT
Introduction: Monkeypox is a viral illness caused by an enveloped double-stranded DNA virus that belongs to the genus orthopoxvirus in the Poxviridae family. Until now, available data on the perinatal outcomes in pregnant people with monkeypox infection acquired in pregnancy are still limited.
Case Report: We report a case of a 33-year-old pregnant woman with monkeypox virus infection confirmed by polymerase chain reaction (PCR) of skin lesions. She had a spontaneous preterm delivery at 32 weeks’ gestation. The monkeypox skin lesions occurred at 20 weeks’ gestation, cleared at 24 weeks’ gestation, and the infant was well at birth with no evidence of monkeypox infection.
Conclusion: This case report underlines the possible association between monkeypox virus infection acquired in pregnancy and preterm birth, which is a leading cause of neonatal morbidity and mortality.
Keywords: Human monkeypox, Pregnancy, Preterm delivery, Viruses, Zoonoses
Introduction
Monkeypox is a viral illness caused by an enveloped double-stranded DNA virus that belongs to the genus orthopoxvirus in the Poxviridae family. Until now, available data on the perinatal outcomes in pregnant people with monkeypox infection acquired in pregnancy are still limited.
Case Report
We report a 33-year-old pregnant woman, Para 1001 (prior normal spontaneous vaginal delivery at term in 2009), who was diagnosed with monkeypox virus infection via polymerase chain reaction (PCR) of lesions affecting her upper and lower extremities at 20 weeks’ gestation.
The diagnosis of monkeypox virus infection was made at 20 weeks’ gestation at a hospital affiliated with the State University of New York Downstate Health Sciences University (SUNY Brooklyn). The source of the infection was unknown. She was transferred to SUNY Brooklyn after the diagnosis was made, and at the time when she was seen at 24 weeks’ gestation, she had no remarkable medical history and the monkeypox skin lesions had already cleared.
At the referring hospital, the patient at 20 weeks’ gestation had a well-circumscribed and umbilicated maculopapular rash that appeared over her face and extremities including the palms of her hands and soles of her feet. There were less than 25 lesions over her body. The lesions were neither painful nor pruritic, and they were more concentrated on her face and extremities rather than on her trunk (Figure 1). The rash progressed to vesicles, pustules, and finally to crusts. She reported no prodromal symptoms such as fever, myalgias, lymphadenopathy, headaches, or fatigue. She did not have oral, vaginal, or perianal involvement and there was no associated lymphadenopathy. The patient underwent screening for rubella, measles, cytomegalovirus, toxoplasmosis, varicella zoster, gonorrhea, chlamydia, human immunodeficiency virus (HIV), herpes simplex virus, syphilis, and monkeypox. Results of laboratory tests were negative except for a positive polymerase chain reaction (PCR), which was collected from swabs of the lesions. The patient continued outpatient prenatal care with isolation precautions and clinical self-monitoring until all the lesions cleared.
It was unclear how the patient contracted the infection and there was no history of travel or close contact with an infected person. She had been sexually active with one male partner who did not experience similar rash. It was unknown if the partner had any sexually transmitted infections. The case was referred to the department of health, which is working with the Centers for Disease Control and Prevention (CDC), to investigate the etiology of the infection.
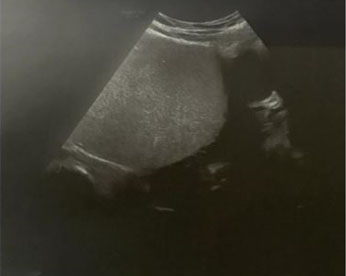
A fetal anatomical sonographic survey at 24 weeks’ gestation revealed no sonographic features suggestive of congenital monkeypox. The contour of the fetal calvarium appeared normal in shape without identifiable defects. Intracranial structures revealed a normal appearing choroid plexus and lateral ventricles, normal size and shape of the cerebellum and cisterna magna, normal thalami, and no deviation of the midline falx. The fetal spine appeared intact throughout its entire length. There was no evidence of cleft lip or facial anomalies or facial anomalies. Normal intra-abdominal anatomy was noted including stomach, liver, diaphragm, and an intact abdominal wall. Four symmetric cardiac chambers, left ventricular outflow tract, and right ventricular outflow tract were identified. Cardiac rhythm was regular. The placenta appeared normal with no evidence of placental calcifications. Interval sonographic fetal growth at 32 weeks’ gestation was appropriate with normal umbilical artery dopplers.
The patient was admitted to the hospital at 32 weeks’ gestation for management of progressive preterm labor. There were no rectovaginal lesions identified on examination. Additionally, there was no clinical evidence of other potential causes of preterm labor, including placental abruption, rupture of membranes, or intrauterine infection. The patient underwent cesarean delivery due to a transverse fetal lie in the setting of progressive preterm labor. She delivered a live vigorous female neonate weighing 1940 grams (~70th percentile), with a fronto-occipital head circumference measuring 30.5 cm (~78th percentile), and a length of 43 cm (~78th percentile). The APGAR scores were nine and nine at 1 and 5 minutes of life, respectively. There were no gross or histological signs of placental infection (necrotic villi, fibrin deposition, inflammatory infiltrates, or necrosis) or umbilical cord abnormalities. The newborn was admitted to the neonatal intensive care unit due to mild respiratory distress secondary to prematurity. Examination of the newborn was within normal limits without any symptoms or signs of congenital monkeypox infection. The neonatal skin, eyes, and mucous membranes were examined thoroughly with no evidence of lesions. Therefore, no active neonatal diagnostic or therapeutic interventions were indicated as per the recommendations of the physicians within SUNY Downstate Hospital’s Infectious Disease department. The newborn continued to develop appropriately and was successfully fed with breastmilk. There were no early neonatal abnormalities detected during the hospital course requiring continued hospitalization and the infant was discharged to home safely on day of life 15.
The postpartum course of the mother was uncomplicated, and the patient was discharged home on postoperative day two.
Discussion
The existing literature on perinatal outcomes following monkeypox infection in pregnancy is limited [1], and most cases to date have been reported by local news media rather than medical or public health publications [2],[3]. As a result, more research must be performed on the clinical characteristics of monkeypox infection in pregnancy, which would address the knowledge gaps related to the effects of monkeypox infection on maternal, fetal, and neonatal outcomes.
The World Health Organization (WHO) reported that transmission of monkeypox virus infection from the mother to the fetus can occur via the placenta (which can lead to congenital monkeypox) [4]. Spontaneous pregnancy loss and stillbirth have been reported in cases of confirmed monkeypox infection during pregnancy [1],[5].
Furthermore, exposure to anogenital monkeypox during vaginal delivery may pose risk of viral transmission to the fetus or neonate [1]; as a result, cesarean delivery would be the most reasonable mode of delivery in pregnant patients with monkeypox infection [6].
Among the cases in pregnancy that have been reported, the outcomes were one case of a mild disease with delivery of a neonate at term with no clinical features of monkeypox infection [7]. Two moderate-to-severe cases of maternal infection had spontaneous first-trimester miscarriages. One moderate case of maternal monkeypox infection had pregnancy loss at 18 weeks’ gestation. In this case, the stillborn fetus had a maculopapular rash, hepatomegaly, and hydrops with a high viral load detected in fetal tissue, umbilical cord, and placenta, confirming vertical transmission of monkeypox [7]. In addition, there was one case of maternal infection at 24 weeks’ gestation complicated by preterm delivery at 30 weeks’ gestation. The premature infant had a generalized skin rash at birth suggestive of monkeypox [7]. Even though the neonate in our case had no evidence of monkeypox infection, it remains unknown whether our patient’s history of maternal monkeypox infection led to the development of preterm labor.
Findings from a recent systematic review that included 7 pregnant people with monkeypox infection showed that monkeypox acquired in pregnancy is associated with a high risk (~ 62%) of vertical transmission [5]. In our case, there was no clinical evidence of congenital monkeypox infection; however, our assessment was based on the absence of the macroscopic signs of the disease rather than on the virological analysis of fetal tissues.
Due to the increased risk of perinatal loss and severe disease during pregnancy, close maternal and fetal surveillance is important [1],[5]. Additionally, it is vital to rule out common etiologies of vesiculopustular rash in pregnancy using polymerase chain reaction (PCR) tests, including varicella zoster, herpes simplex, and secondary syphilis [6]. The decision to treat and monitor a pregnant person as an outpatient or in the inpatient setting should be individualized and treatment should be offered to pregnant patients when indicated [1]. Serial obstetrical ultrasound assessment for signs of congenital monkeypox infection should be performed in pregnant people with monkeypox infection acquired in pregnancy given the risk of vertical transfusion to the fetus [6]. If treatment is indicated, tecovirimat is the first-line antiviral for pregnant patients [1]. It is unknown if treatment with tecovirimat during pregnancy prevents congenital monkeypox infection [1]. Cidofovir and brincidofovir should be avoided in pregnant people, especially in the first trimester, due to evidence of teratogenicity in animal studies [1],[5].
The two available vaccines that have demonstrated efficacy against monkeypox are JYNNEOS and ACAM2000 [8]. When a pregnant person is diagnosed with monkeypox infection via PCR analysis, careful assessment should be performed to identify patients with severe disease [5]. Those patients with severe disease should be considered for Tecovirimat and vaccinia immune globulin [5],[8]. For the current outbreak, pregnant patients, who are exposed to monkeypox can receive the JYNNEOS vaccination [8]. Postexposure vaccination is recommended within four days from the date of exposure for the best chance to prevent onset of the disease [8]. The use of ACAM2000 vaccine is contraindicated in pregnant people because of the risk of pregnancy loss, congenital defects, and vaccinia virus infection in fetuses and newborns and the availability of a nonreplicating viral vaccine [8].
Conclusion
This case report underlines the possible association between monkeypox virus infection acquired in pregnancy and preterm birth, which is a leading cause of neonatal morbidity and mortality. It is imperative that health care providers stay abreast of the principles of obstetrical care for pregnant women with monkeypox infection. To better understand the actual burden of the disease, continuous research on the biology, epidemiology, clinical manifestations, diagnosis, and treatment is needed. In addition, a worldwide registry of monkeypox infection cases acquired during pregnancy is important to increase education and awareness of this infectious disease.
REFERENCES
1.
2.
Khalil A, Samara A, O’Brien P, et al. Monkeypox in pregnancy: Update on current outbreak. Lancet Infect Dis 2022;22(11):1534–5. [CrossRef]
[Pubmed]

3.
Centers for Disease Control and Prevention. Mpox infections after vaccination. [Available at: https://www.cdc.gov/poxvirus/mpox/index.html]

4.
World Health Organization. Mpox (monkeypox). 2023. [Available at: https://www.who.int/news-room/fact-sheets/detail/monkeypox]

5.
D’Antonio F, Pagani G, Buca D, Khalil A. Monkeypox infection in pregnancy: A systematic review and metaanalysis. Am J Obstet Gynecol MFM 2023;5(1):100747. [CrossRef]
[Pubmed]

6.
Dashraath P, Nielsen-Saines K, Rimoin A, Mattar CNZ, Panchaud A, Baud D. Monkeypox in pregnancy: Virology, clinical presentation, and obstetric management. Am J Obstet Gynecol 2022;227(6):849–61.e7. [CrossRef]
[Pubmed]

7.
Mbala PK, Huggins JW, Riu-Rovira T, et al. Maternal and fetal outcomes among pregnant women with human monkeypox infection in the democratic republic of congo. J Infect Dis 2017;216(7):824–8. [CrossRef]
[Pubmed]

8.
Centers for Disease Control and Prevention. Vaccination administration considerations for specific populations. 2022. [Available at: https://www.cdc.gov/poxvirus/mpox/interim-considerations/special-populations.html]

SUPPORTING INFORMATION
Acknowledgments
We would like to express our deepest appreciation to the patient who kindly allowed us to share her story in this article so that it may inform future obstetrical care. We acknowledge the valuable input provided by Dr. Mudar Dalloul. Their insightful suggestions greatly enhanced the quality of this manuscript.
Author ContributionsNawras Zayat - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Rami Noumi - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Wissam Akkary - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2023 Nawras Zayat et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.