|
Case Report
Pancreatic ductal adenocarcinoma (intestinal-type differentiation) presenting as ovarian metastases: A case report
1 Medical Student, University of Virginia School of Medicine, Charlottesville, Virginia, USA
2 Resident, Department of Obstetrics and Gynecology, University of Virginia, Charlottesville, Virginia, USA
3 Fellow, Department of Pathology, University of Virginia, Charlottesville, Virginia, USA
4 Associate Professor, Department of Pathology, University of Virginia, Charlottesville, Virginia, USA
5 Professor, Department of Obstetrics and Gynecology, University of Virginia, Charlottesville, Virginia, USA
Address correspondence to:
Emily Fronk
University of Virginia School of Medicine, 1215 Lee Street, Charlottesville, VA 22903,
USA
Message to Corresponding Author
Article ID: 100159Z08EF2023
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Fronk E, Welp A, Dibbern M, Mills A, Cantrell L. Pancreatic ductal adenocarcinoma (intestinal-type differentiation) presenting as ovarian metastases: A case report. J Case Rep Images Obstet Gynecol 2023;9(2):25–29.ABSTRACT
Introduction: Metastasis of pancreatic adenocarcinoma to the ovaries is uncommon and can clinically present similarly to primary ovarian disease, presenting challenges for diagnosis. Immunohistochemical staining is typically useful in distinguishing ovarian primaries from ovarian metastasis. A newer and rarer variant of pancreatic adenocarcinoma, that with intestinal-type differentiation, has not previously been reported as metastasizing to the ovary. Its diagnosis is particularly difficult due to the overlap in staining patterns with primary ovarian neoplasms.
Case Report: This case report presents the case of a 60-year-old female who presented with non-specific abdominal symptoms and was found to have a large adnexal mass. Imaging showed additional lesions in the peritoneum, pancreas, and liver. She underwent biopsy with morphology and immunohistochemical staining consistent with pancreatic ductal adenocarcinoma with intestinal-type differentiation. She was started on FOLFIRINOX therapy but ultimately opted to pursue comfort care due to disease progression.
Conclusion: Although rare, with this being the first report of this disease course to our knowledge, clinicians should be aware of this possibility, as it impacts choice of treatment.
Keywords: Immunohistochemical staining, Ovarian metastasis, Pancreatic adenocarcinoma
Introduction
Pancreatic cancer accounts for 2.7% of cancers in women and most often metastasizes through the lymphatic system [1],[2]. Common sites of distant spread include the liver, peritoneum, and lungs; however, metastases to the ovaries are seen about 4–6% of the time and are most commonly identified during autopsy [1],[3]. Metastatic disease is typically clinically identified if patients present with pelvic and gastrointestinal complaints, which is seen similarly in ovarian cancer. Given the key differences in the management of pancreatic versus ovarian cancer, coming to diagnostic clarity is critical. In this case report, we describe the presentation of a patient with an ovarian tumor, determined to be metastatic from pancreatic ductal adenocarcinoma with intestinal-type differentiation. To our knowledge, this is the first report of this specific type of pancreatic cancer metastasizing to the ovaries.
Case Report
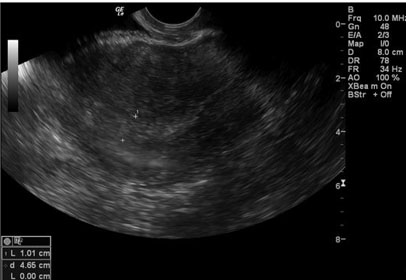
In June 2022, a 60-year-old postmenopausal nulligravid patient presented to the emergency department (ED) with complaints of progressive abdominal pain, bloating, nausea, early satiety, constipation, and weight loss over the previous 4–6 weeks. Her past medical history was notable for depression and she denied tobacco or alcohol use. She had a performance status of 1, a body mass index (BMI) of 21.5, and a malnutrition screening tool (MST) score of 1. She reported use of combined oral contraceptives for 5–7 years. Her family history was significant for a maternal grandmother with ovarian cancer and a paternal grandmother with pancreatic cancer. She had seen her primary care provider (PCP) at the onset of her abdominal pain two weeks earlier and had underwent a pelvic ultrasound demonstrating a 16 11 × 13 cm cystic mass. One day prior to her presentation to the ED, she underwent a non-contrast computed tomography (CT) scan, showing a 13.6 × 8.8 × 16.3 cm complex left adnexal mass with associated peritoneal carcinomatosis, ascites, and indeterminate pancreatic tail and hepatic lesions felt to be metastatic ovarian cancer. She was informed by her PCP that she would need a biopsy to confirm an underlying malignancy.
In the ED, tumor markers were collected and the gynecologic oncology service was consulted, with plan made for follow-up of her tumor markers and an outpatient abdominal and pelvic magnetic resonance imaging (MRI). Her tumor markers returned elevated, with a CEA of 26.2, CA19-9 of 8.060, and a CA-125 of 558. Other labs drawn at that time, including complete blood count (CBC) with differential and comprehensive metabolic panel (CMP) were within normal limits. The MRI confirmed the findings of her earlier CT and further elucidated the abdominal findings, demonstrating capsular metastatic deposits of the liver and a 4 cm intrapancreatic mass involving the pancreatic body and proximal pancreatic tail. The intrapancreatic mass was favored to represent an intrapancreatic metastatic deposit, given the extent of the pelvic disease and its similarities in signal to the pelvic and omental deposits. Figure 1 shows the patient’s CT and MRI scans. The patient was seen in Gynecology Oncology clinic a few days later, where she was counseled extensively regarding the differential diagnosis, including ovarian neoplasms versus metastatic cancers from another site. Subsequent biopsy of the abdominopelvic mass returned a final pathology of adenocarcinoma. On immunohistochemical staining (IHC), the tumor showed expression of CK7, CK20, and CDX-2 and lacked expression of PAX-8, suggestive of primary ovarian mucinous carcinoma, metastatic upper gastrointestinal carcinoma, or metastatic pancreatic adenocarcinoma.
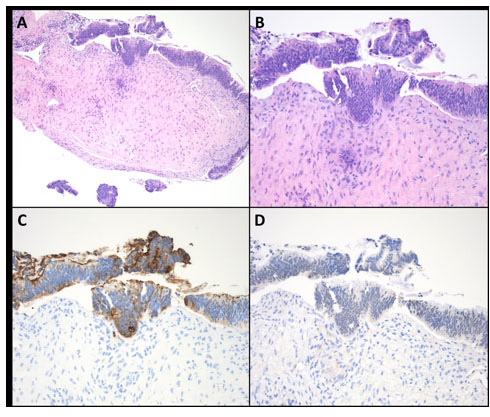
The patient was then preliminarily scheduled to start neoadjuvant chemotherapy. Her biopsy specimen underwent additional staining, which showed expression of CA19-9 and no expression of SATB2. Figure 2 shows the patient’s pelvic mass biopsy after the additional staining. The case was discussed at interdisciplinary tumor board and given the imaging findings, tumor histomorphology, and tumor immunoprofile, her primary tumor site was favored to be pancreatic ductal adenocarcinoma with intestinal-type differentiation. This diagnosis was made 31 days after her initial presentation to her PCP and 18 days after presentation to the ED. She transferred care to medical oncology, where she started on FOLFIRINOX therapy as standard of care for advanced pancreatic cancer. Her disease ultimately progressed, and the patient and her family opted to pursue comfort measures with home hospice by the end of July 2022, approximately six weeks after her initial presentation.
Discussion
Metastases to the ovary from non-gynecologic malignancies is estimated to occur 6–15% of the time, most commonly arising from colon, breast, appendix, and stomach primaries [3],[4],[5]. Metastatic disease to the ovaries can be particularly difficult to diagnose, as often the resulting ovarian tumors may strongly resemble ovarian primaries both clinically and pathologically [4],[5],[6]. Clinically, most patients, regardless of tumor origin, report non-specific gastrointestinal symptoms once the ovarian tumor has advanced to a large-enough tumor burden [7].
Pancreatic ductal adenocarcinoma with pancreatobiliary-type differentiation is the most common type of pancreatic cancer, accounting for more than 85% of pancreatic cancers [8]. Adenocarcinoma of intestinal-type differentiation is a distinct and rarer variant characterized by low-grade nuclear atypia with a morphology similar to intestinal metaplasia, which has been described in a variety of anatomical sites [9]. However, it has only been recognized as occurring in the pancreas within the past 15 years. There have been no reported cases in the literature of intestinal-type pancreatic cancer metastasizing to the ovary.
While IHC has traditionally been the tool of choice in differentiating between primary ovarian cancer and ovarian metastases, differentiating ovarian primaries from intestinal-type tumors presents a diagnostic challenge due to the significant overlap in markers. Table 1 addresses the stains commonly used to differentiate colorectal, non-mucinous ovarian, mucinous ovarian, and pancreaticobiliary tumors. CK7 and CK20 are stains commonly used in combination to determine the primary site of metastatic carcinoma and are particularly useful in differentiating between metastatic colorectal carcinomas and ovarian carcinomas [6],[10],[11]. Colorectal carcinomas are generally CK7-negative while both mucinous and non-mucinous primary ovarian carcinomas are CK7-positive [6],[10],[11]. Distinguishing primary mucinous ovarian carcinoma from metastatic mucinous carcinoma involving the ovary can be especially challenging, as both tumor types can show expression of CK20. In these instances, incorporation of imaging and clinical findings is necessary for definitive localization of the primary neoplasm.
Pancreatic cancer with intestinal-type differentiation has been found to most often be CK7-positive and CK20-positive, while that with pancreatobiliary-type differentiation is most often CK7-positive and CK20-negative [12],[13]. CA 19-9 is positive in both subtypes of pancreatic cancers [8]. In spite of its more ubiquitous use as a serum marker for pancreatic cancer, CA19-9 expression is also seen colorectal, gastric, and mucinous ovarian carcinomas [6]. Therefore, the utility of CA 19-9 IHC staining is in its use as part of an IHC panel, rather than as an independent marker of tumor site of origin.
CDX-2 is a gene essential for the development of the large and small intestine, making it ubiquitously positive in both colorectal carcinoma and pancreatic cancer with intestinal-type differentiation [10],[13]. It is also positive in up to 93% of mucinous ovarian carcinomas but is rarely positive in non-mucinous ovarian carcinomas or pancreatic adenocarcinoma with pancreatobiliary-type differentiation [6],[8],[10].
PAX-8 is a transcription factor involved in the development of the Mullerian [11]. In non-mucinous ovarian carcinomas, the epithelium acquires PAX-8 after malignant transformation, thus staining positive for PAX-8 almost ubiquitously [11]. However, mucinous ovarian carcinomas only acquire PAX-8 in up to half of cases [11]. SATB2 has a narrow expression profile in the lower gastrointestinal (GI) tract, thus making it a relatively specific marker for colorectal or appendiceal carcinoma [14]. It is negative in both mucinous and non-mucinous primary ovarian carcinomas and both sub-types of pancreatic ductal adenocarcinoma [6],[11].
A literature review was undertaken to identify prevalence of and treatment options for pancreatic adenocarcinoma with intestinal-type differentiation with ovarian metastases. It was conducted via PubMed using the following terms: pancreas cancer AND intestinal type AND ovarian metastasis. Eight papers were identified; however, upon further review, none reported a case of intestinal-type metastasis to the ovaries. Further searches were attempted with various modifications of the terms, yet the authors were unable to find a paper describing this specific disease process. To our knowledge, this is the first report of pancreatic adenocarcinoma with intestinal-type differentiation metastasizing to the ovaries.
Conclusion
This case report describes the presentation of a patient with pancreatic adenocarcinoma with intestinal-type differentiation that had metastasized to the ovaries. A review of the literature demonstrates that this is the only reported case published thus far, underscoring the rarity of our patient’s presentation. Therefore, this case is presented to raise awareness of this unique disease subtype among clinicians. Due to the initial symptoms that our patient presented with, as well as the complexity of diagnosis, clinicians, specifically gynecologists, need to be aware of the possibility for pancreatic cancer to present in this manner. Although rare, maintaining pancreatic adenocarcinoma with intestinal-type differentiation on the differential for a pelvic mass with metastatic lesions is essential for ensuring that patients receive the correct treatment.
REFERENCES
1.
Zhou X, Cao H, Zhao B, et al. Pancreatic cancer with ovarian metastasis: Clinical features, diagnosis and management. Research Square 2020. [CrossRef]

2.
Deeb A, Haque SU, Olowokure O. Pulmonary metastases in pancreatic cancer, is there a survival influence? J Gastrointest Oncol 2015;6(3):E48–51. [CrossRef]
[Pubmed]

3.
Wang SD, Zhu L, Wu HW, Dai MH, Zhao YP. Pancreatic cancer with ovarian metastases: A case report and review of the literature. World J Clin Cases 2020;8(21):5380–8. [CrossRef]
[Pubmed]

4.
Hart WR. Diagnostic challenge of secondary (metastatic) ovarian tumors simulating primary endometrioid and mucinous neoplasms. Pathol Int 2005;55(5):231–43. [CrossRef]
[Pubmed]

5.
Ackroyd SA, Goetsch L, Brown J, Houck K, Wang C, Hernandez E. Pancreaticobiliary metastasis presenting as primary mucinous ovarian neoplasm: A systematic literature review. Gynecol Oncol Rep 2019;28:109–15. [CrossRef]
[Pubmed]

6.
Babaier A, Ghatage P. Mucinous cancer of the ovary: Overview and current status. Diagnostics 2020;10(1):52. [CrossRef]
[Pubmed]

7.
Kir G, Gurbuz A, Karateke A, Kir M. Clinicopathologic and immunohistochemical profile of ovarian metastases from colorectal carcinoma. World J Gastrointest Surg 2010;2(4):109–16. [CrossRef]
[Pubmed]

8.
Mostafa ME, Erbarut-Seven I, Pehlivanoglu B, Adsay V. Pathologic classification of “pancreatic cancers”: Current concepts and challenges. Chin Clin Oncol 2017;6(6):59–9. [CrossRef]
[Pubmed]

9.
Ushiku T, Arnason T, Ban S, et al. Very well-differentiated gastric carcinoma of intestinal type: Analysis of diagnostic criteria. Mod Pathol 2013;26(12):1620–31. [CrossRef]
[Pubmed]

10.
Baker PM, Oliva E. Immunohistochemistry as a tool in the differential diagnosis of ovarian tumors: An update. Int J Gynecol Pathol 2005;24(1):39–55.
[Pubmed]

11.
Strickland S, Wasserman JK, Giassi A, Djordjevic B, Parra-Herran C. Immunohistochemistry in the diagnosis of mucinous neoplasms involving the ovary: The added value of SATB2 and biomarker discovery through protein expression database mining. Int J Gynecol Pathol 2016;35(3):191–208. [CrossRef]
[Pubmed]

12.
Albores-Saavedra J, Simpson K, Dancer YJ, Hruban R. Intestinal type adenocarcinoma: A previously unrecognized histologic variant of ductal carcinoma of the pancreas. Ann Diagn Pathol 2007;11(1):3–9. [CrossRef]
[Pubmed]

13.
Fernández Moro C, Fernandez-Woodbridge A, Alistair D’souza M, et al. Correction: Immunohistochemical typing of adenocarcinomas of the pancreatobiliary system improves diagnosis and prognostic stratification. PLoS One 2017;12(1):e0171283. [CrossRef]
[Pubmed]

14.
Berg KB, Schaeffer DF. SATB2 as an immunohistochemical marker for colorectal adenocarcinoma: A concise review of benefits and pitfalls. Arch Pathol Lab Med 2017;141(10):1428–33. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Emily Fronk - Conception of the work, Design of the work, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Annalyn Welp - Conception of the work, Design of the work, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Megan Dibbern - Conception of the work, Design of the work, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Anne Mills - Conception of the work, Design of the work, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Leigh Cantrell - Conception of the work, Design of the work, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2023 Emily Fronk et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.