|
Case Report
Case report of Hepatitis C treatment during pregnancy
1 Resident Physician within the UBC Department of Obstetrics and Gynaecology, Room C342, 4500 Oak Street, Vancouver, BC V6H 3N1, Canada
2 Reproductive Infectious Diseases Fellow, Faculty of Medicine, Obstetrics and Gynaecology, University of British Columbia, Room C342, 4500 Oak Street, Vancouver, BC V6H 3N1, Canada
3 Clinical Assistant Professor, Division of Gynaecologic Specialties, Faculty of Medicine, Obstetrics and Gynaecology, University of British Columbia, 915-750 West Broadway, Vancouver, BC V5Z 1H8, Canada
4 Clinical Associate Professor, Department of Gastroenterology, St. Paul’s Hospital, University of British Columbia, 770-1190 Hornby Street, Vancouver, BC V6Z 3C7, Canada
Address correspondence to:
Kirsten Bock
Room C342, 4500 Oak Street, Vancouver, BC V6H 3N1,
Canada
Message to Corresponding Author
Article ID: 100162Z08KB2023
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Bock K, Atkinson A, Elwood C, Ramji A. Case report of Hepatitis C treatment during pregnancy. J Case Rep Images Obstet Gynecol 2023;9(2):39–42.ABSTRACT
Introduction: Despite the increasing availability of Hepatitis C (HCV) anti-viral therapy for adults, there is minimal data regarding treatment safety and efficacy during pregnancy.
Case Report: A 33-year-old primiparous female underwent successful HCV treatment with direct-acting antivirals (DAAs) beginning at 26 weeks gestational age.
Conclusion: This case demonstrates the effective treatment of HCV during pregnancy. Formal trials are still required to assess the long-term safety and efficacy as treatment with DAAs for Hepatitis C is not yet established in pregnancy.
Keywords: HCV, Pregnancy, Reproductive infectious diseases
Introduction
Despite a vertical transmission rate of approximately 5% for Hepatitis C in pregnancy [1], there is limited information regarding interventions to reduce perinatal transmission. One interventional study in which DAA HCV treatment with sofosbuvir/ledipasvir was initiated in eight pregnant individuals, demonstrated similar pharmacokinetics in non-pregnant and pregnant participants, and there were no clinically significant safety concerns identified [2]. A 100% long-term viral suppression rate was achieved in all participants and there were no adverse effects on maternal or early infant outcomes. These findings also suggest that there is no need for dose adjustment in pregnant individuals. While pregnant individuals have been excluded from drug trials making it difficult to assess safety and efficacy in this population [3], given preliminary safety data and increased public awareness regarding treatment availability for HCV, research is needed to determine whether this treatment can be safely offered to reduce the risk of perinatal transmission.
Case Report
Mrs. L, a 33-year-old primiparous female, was diagnosed with HCV, genotype 1a, after abnormal liver enzyme tests were detected during investigations for hyperemesis gravidarum in the first trimester. Viral load at this time was 1.7 IU/mL. Mrs. L had no prior testing for HCV and risk factors identified were prior residence in a high-prevalence country with an occupation in healthcare. There was no family history of Hepatitis C. When diagnosed with HCV, Mrs. L sought Hepatology consultation for consideration of DAA therapy. Mrs. L was knowledgeable in regard to DAA therapy and its efficacy through her profession and had initial review by Hepatology at 14 weeks gestational age (GA). Based on her Fibrosis-4 Index (FIB 4) she was evaluated as not affected by cirrhosis. Mrs. L was referred to Reproductive Infectious Diseases clinic at 19+5 weeks GA for further evaluation and counseling regarding perinatal transmission and treatment options.
Mrs. L was otherwise healthy, with no significant past medical history. All other antenatal screening tests including Hepatitis B and human immunodeficiency virus (HIV) were negative. She was of Asian ethnicity and had a family history of type 2 diabetes mellitus and gestational diabetes. Her gestational diabetes screen at 28 weeks GA was positive and this was effectively managed with diet throughout pregnancy. She developed iron deficiency anemia in the second trimester which was treated with iron supplementation.
Mrs. L was counseled extensively regarding the lack of safety data of HCV DAA during pregnancy and unknown effects on the fetus. She expressed a firm desire to receive treatment for HCV and elected to proceed with DAA therapy. She was initiated on ledipasvir 90 mg/sofosbuvir 400 mg (Harvoni) at 26 weeks GA. This regimen was selected based on genotype and aforementioned research in pregnancy. Viral load did not influence our treatment selection.
Twenty-six weeks GA was chosen as a starting point for treatment as this allowed Mrs. L to complete a 12-week treatment course prior to 38 weeks GA. The aim was to complete treatment prior to delivery for the purpose of decreasing the likelihood of vertical transmission and avoiding treatment during the postpartum period. Patient tolerance of treatment was prioritized and nausea was managed proactively to maximize medication adherence. Mrs. L did not report any treatment side-effects including nausea and received DAA treatment without missed doses.
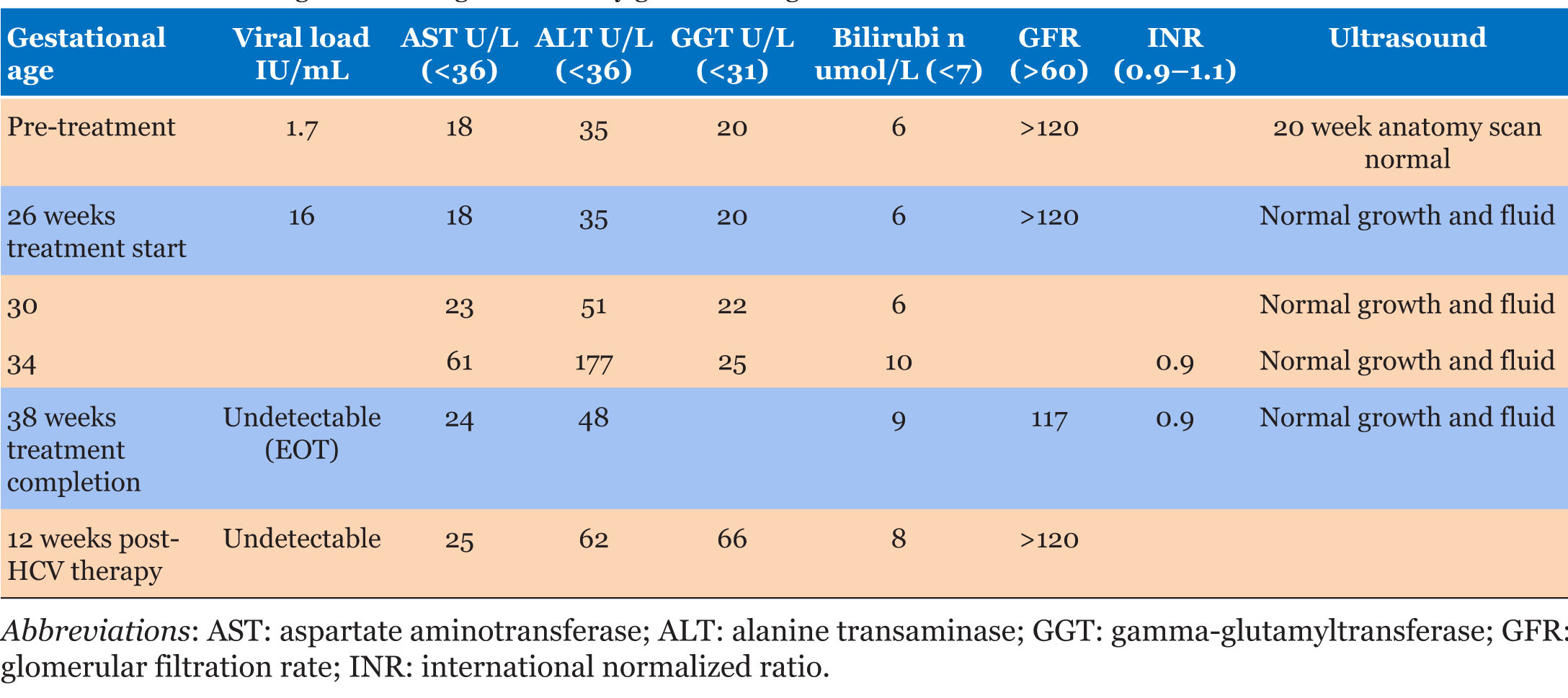
Close monitoring of liver enzymes and function, renal function as well as fetal growth was performed. The initial anatomy scan prior to starting therapy demonstrated normal fetal anatomy and appropriate growth for gestational age. Repeat fetal growth scans were performed at 24, 30, 34, and 38 weeks GA, all of which were normal with appropriate growth and fluid.
First-trimester liver enzyme abnormalities resolved spontaneously prior to commencement of HCV treatment. Subsequently Mrs. L did have a mild increase in alanine transaminase (ALT), gamma-glutamyltransferase (GGT), and bilirubin as shown in Table 1 at 34 weeks GA. There were no other signs of preeclampsia or cholestasis. The manufacturer monograph for Harvoni does not report any known cases of increased liver enzymes or acute hepatitis related to the drug. The Harvoni monograph reports cases of Hepatitis B virus (HBV) reactivation in the setting of HCV treatment but repeat HBV testing was negative. The etiology of the ALT elevation was unclear. Repeat work-up for other causes of liver disease including autoimmune hepatitis were unremarkable. The bilirubin was only mildly elevated and as such DAA therapy was continued with Mrs. L’s understanding and consent.
Mrs. L completed DAA treatment at 38 weeks GA. Her HCV RNA at the end-of therapy (EOT) was undetectable though she continued to have a mildly elevated ALT and normal bilirubin. At 39+3 weeks GA the patient developed headache with vision changes and proteinuria without hypertension. Induction of labor with prostaglandins was therefore recommended for possible evolving preeclampsia. During induction of labor, Mrs. L had spontaneous rupture of membranes followed by uterine tachysystole and abnormal fetal heart rate patterns for which she underwent emergency cesarean section at 39+4 weeks GA. She delivered a healthy infant weighing 3.7 kg. The baby did not require any intervention following delivery and was cleared for discharge following pediatric review on day 1 of life.
Mrs. L’s postpartum course was complicated by anemia with a hemoglobin of 63 g/L which required transfusion of 2 units of packed red blood cells. She was discharged from hospital on postoperative day 2, with no medical concerns on follow-up at 6 weeks postpartum. Her HCV RNA was undetectable at EOT as well as at 4 and 12 weeks post-therapy-indicative of a sustained viral response (SVR 4 and 12). Liver enzyme tests continued to be elevated with elevated ALT. At five months postpartum transient elastography diagnosed stage 0-1 fibrosis, with minimal hepatic steatosis. As repeat work-up for other etiologies for liver disease was unremarkable, the liver dysfunction was ascribed to steatosis with no further intervention planned. Mrs. L’s infant received negative testing for HCV at five months of age.
Discussion
Perinatal transmission continues to be an ongoing cause for new HCV infections worldwide. With an estimated 290,000 people worldwide dying from cirrhosis and hepatocellular cancer resulting from long-term infection with HCV in 2019, reduction of HCV globally has been highlighted by the World Health Organization (WHO) as a priority area. The WHO has set a goal of reducing new HCV infections by 80% by 2030 [4]. Few countries are on track to achieve this, but those healthcare systems that are, have incorporated universal treatment and wider screening in their strategies [5],[6]. Screening for chronic infection as part of routine antenatal care provides an opportunity to screen patients for HCV and engage those who test positive. Diagnosis of HCV at this time can come as a shock to many and the potential for transmission to their baby can cause significant stress and undue feelings of guilt. For this reason, patients may increasingly seek treatment in an attempt to prevent perinatal transmission. There is the potential for treatment during pregnancy to provide an opportunity for closely monitored care for the pregnant person during antenatal visits and for a reduction in the incidence of disease by reducing perinatal transmission.
Treatment for HCV also has a very high likelihood of cure for the patient which is known to improve overall mortality and morbidity [7].
To date a phase I pharmacokinetic trial of HCV treatment in pregnancy has been undertaken with reassuring results [2], and while awaiting further phases II and III trials, individual situations may arise whereby patients request treatment during pregnancy.
Conclusion
This case demonstrates the potential for HCV therapy in pregnancy. Direct-acting antivirals have been shown to be highly effective in non-pregnant patients and achieved sustained virologic response during follow-up for our patient. Patients need to be consented to therapy particularly as the safety profile of DAA for HCV in pregnancy has not been established. In this case, HCV RNA was undetectable at five months for the infant, indicative of no perinatal transmission of HCV.
REFERENCES
1.
Benova L, Mohamoud YA, Calvert C, Abu-Raddad LJ. Vertical transmission of hepatitis C virus: Systematic review and meta-analysis. Clin Infect Dis 2014;59(6):765–73. [CrossRef]
[Pubmed]

2.
Chappell CA, Scarsi KK, Kirby BJ, et al. Ledipasvir plus sofosbuvir in pregnant women with hepatitis C virus infection: A phase 1 pharmacokinetic study. Lancet Microbe 2020;1(5):e200–8. [CrossRef]
[Pubmed]

3.
Jhaveri R, Yee LM, Antala S, Murphy M, Grobman WA, Shah SK. Responsible inclusion of pregnant individuals in eradicating HCV. Hepatology 2021;74(3):1645–51. [CrossRef]
[Pubmed]

4.
World Health Organization. Global health sector strategy on viral hepatitis 2016–2021. Towards ending viral hepatitis. World Health Organization. 2016. [Available at: https://apps.who.int/iris/handle/10665/246177]

5.
Pedrana A, Munari S, Stoové M, Doyle J, Hellard M. The phases of hepatitis C elimination: Achieving WHO elimination targets. Lancet Gastroenterol Hepatol 2021;6(1):6–8. [CrossRef]
[Pubmed]

6.
Razavi H, Sanchez Gonzalez Y, Yuen C, Cornberg M. Global timing of hepatitis C virus elimination in high-income countries. Liver Int 2020;40(3):522–9. [CrossRef]
[Pubmed]

7.
Shoukry NH, Feld JJ, Grebely J. Hepatitis C: A Canadian perspective. Can Liver J 2018;1(2):1–3.
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Kirsten Bock - Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Andrea Atkinson - Conception of the work, Design of the work, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Chelsea Elwood - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Alnoor Ramji - Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2023 Kirsten Bock et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.