|
Case Report
Primary omental ectopic pregnancy after frozen-thawed single embryo transfer: A case report and review of the literature
1 Fellow, The Center for Advanced Reproductive Services, Department of Obstetrics and Gynecology, University of Connecticut School of Medicine, Farmington, Connecticut, USA
2 Clinical Instructor, The Center for Advanced Reproductive Services, Department of Obstetrics and Gynecology, University of Connecticut School of Medicine, Farmington, Connecticut, USA
3 Clinical Professor, The Center for Advanced Reproductive Services, Department of Obstetrics and Gynecology, University of Connecticut School of Medicine, Farmington, Connecticut, USA
4 Assistant Clinical Professor, The Center for Advanced Reproductive Services, Department of Obstetrics and Gynecology, University of Connecticut School of Medicine, Farmington, Connecticut, USA
Address correspondence to:
Katherine Koniares
The Center for Advanced Reproductive Services, Division of Reproductive Endocrinology and Infertility, University of Connecticut School of Medicine, 2 Batterson Park Road, Farmington, Connecticut 06032,
USA
Message to Corresponding Author
Article ID: 100170Z08KK2024
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Koniares K, Godiwala P, Benadiva C, DiLuigi A. Primary omental ectopic pregnancy after frozen-thawed single embryo transfer: A case report and review of the literature. J Case Rep Images Obstet Gynecol 2024;10(1):23–28.ABSTRACT
Introduction: The objective of this case report is to present the first known case of a primary omental ectopic pregnancy following frozen-thawed embryo transfer.
Case Report: A 35-year-old G0 female with severe male factor infertility underwent a frozen-thawed single embryo transfer. Serial transvaginal ultrasounds and beta human chorionic gonadotropin levels led to the diagnosis of ectopic pregnancy. The patient did not respond to medical management with methotrexate. On laparoscopy, an ectopic pregnancy was diagnosed on the greater omentum. The pregnancy was resected with the LigaSure device. Pathology evaluation was consistent with an omental ectopic pregnancy.
Conclusion: Although rare, omental ectopic pregnancy should remain in the differential in the setting of a pregnancy of unknown location.
Keywords: Frozen embryo transfer, In vitro fertilization, Omental ectopic pregnancy
Introduction
The ectopic pregnancy rate following embryo transfer ranges from 1.4% to 2.2% [1],[2],[3]. Risk factors for ectopic pregnancy include assisted reproductive technology, previous ectopic pregnancy, history of tubal surgery or tubal disease, prior sexually transmitted infection, and pelvic adhesions [4]. The risk of heterotopic pregnancies following embryo transfer is estimated at 1% [5]. There are three published cases of heterotopic pregnancies following in vitro fertilization (IVF) and fresh embryo transfer (ET) with secondary implantation on the omentum [6],[7],[8].
Abdominal ectopic pregnancy can be classified as primary or secondary. Despite the rarity of omental ectopic pregnancies, Studdiford proposed the following diagnostic criteria in 1942 for a primary abdominal pregnancy: (1) bilateral fallopian tubes and ovaries are normal without evidence of remote or recent injury, (2) there is no evidence of a uteroperitoneal fistula, and (3) the pregnancy is exclusively related to the peritoneal surface and early enough in gestation to exclude the possibility of primary implantation in the fallopian tube with secondary implantation in the abdomen [9]. This is the first case, to our knowledge, of a primary omental ectopic pregnancy following IVF with frozen-thawed embryo transfer (FET).
Case Report
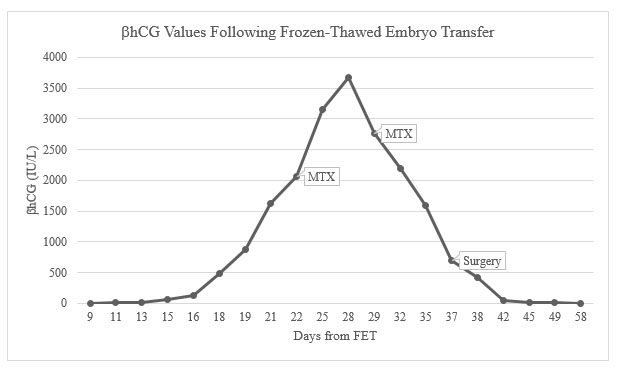
A 35-year-old G0 female with severe male factor infertility (total motile count <1 million sperm) underwent a natural cycle FET following a failed fresh ET. Previous hysterosalpingogram had demonstrated bilaterally patent fallopian tubes and a normal uterine cavity. A single high quality blastocyst was transferred six days after the patient’s peak luteinizing hormone (LH) surge in a natural cycle. Quantitative beta human chorionic gonadotropin (βhCG) was measured nine days after FET with a result of 2.2 IU/L. This was repeated two days later and demonstrated an appropriate percent rise to 4.4 IU/L. Serial βhCG levels were obtained every other day for one week and demonstrated an appropriate percent increase (Figure 1). At a βhCG of 124 IU/L and gestational age of five weeks and zero days, the patient was evaluated in the office with transvaginal ultrasound (TVUS) due to concern for an ectopic pregnancy in the setting of lower than expected absolute βhCG levels. The TVUS did not demonstrate an intrauterine pregnancy, adnexal mass or free fluid in the pelvis. The patient continued to be closely monitored with serial βhCG levels every 1–2 days. During this time, the patient denied abdominal pain, pelvic pain, and vaginal bleeding.
At a βhCG of 866 IU/L and a gestational age of five weeks and three days the patient underwent a repeat TVUS that again showed no evidence of an intrauterine pregnancy and no adnexal masses or free fluid in the cul-de-sac. The following day, the patient underwent another TVUS with the same findings followed by a manual vacuum aspiration to assess her endometrial tissue for the presence of chorionic villi. At a gestational age of five weeks and five days, the βhCG had increased to 2,057 IU/L and the pathology report demonstrated no evidence of chorionic villi in the endometrium. An ectopic pregnancy was diagnosed and the patient was counseled on treatment options including laparoscopic surgery or methotrexate (MTX). The patient elected to proceed with MTX therapy and received an intramuscular injection of MTX dosed at 50 mg/m2. Following MTX administration, the patient’s βhCG continued to rise with a peak of 3,670 IU/L and one week later the patient received a second dose of MTX. Transvaginal ultrasound at this time did not demonstrate an intrauterine gestational sac, adnexal mass, or free fluid, and the patient remained asymptomatic. Subsequently, the patient’s βhCG values began to decline appropriately.
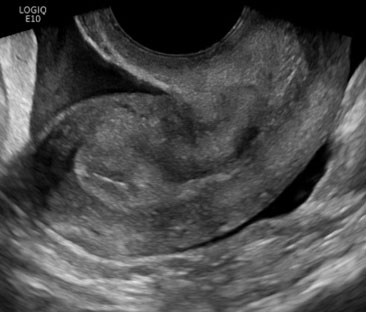
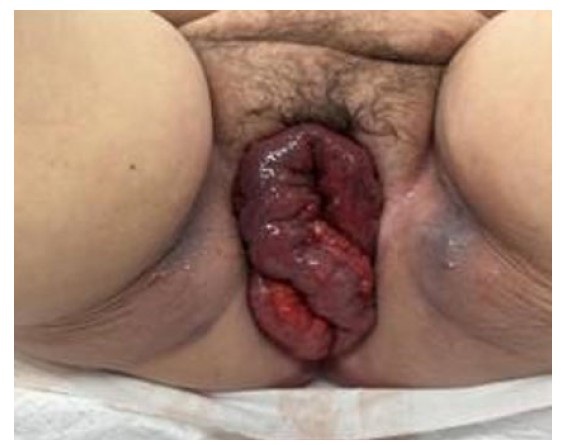
Approximately one week after receiving the second dose of MTX, at a gestational age of eight weeks and zero days, the patient called the office with sudden onset of severe bilateral lower quadrant abdominal pain associated with nausea and lightheadedness. The patient was instructed to present to the Emergency Department immediately. Physical exam demonstrated stable vital signs and a soft abdomen with diffuse tenderness to palpation and rebound without guarding. Laboratory results showed a hematocrit of 39.6% and βhCG of 688 IU/L. Transvaginal ultrasound revealed a 3.3 cm right ovarian hemorrhagic cyst and a large amount of free fluid in the pelvis and extending into the upper abdomen (Figure 2). At this time, the decision was made to proceed with a diagnostic laparoscopy.
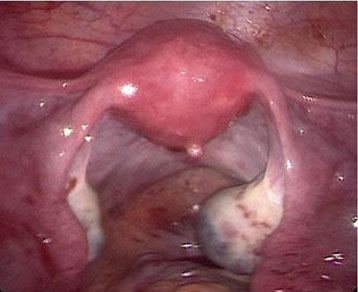
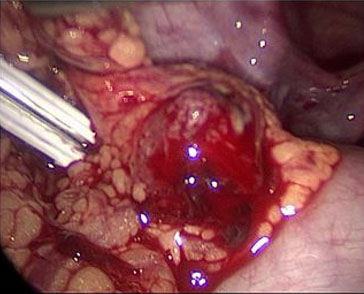
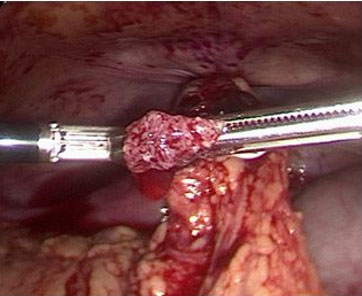
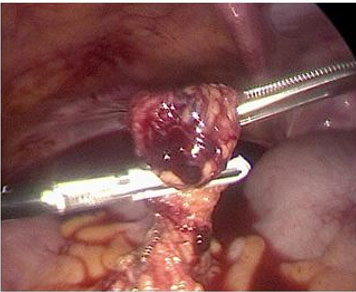
Upon entry into the abdomen, laparoscopy demonstrated 800 cc of hemoperitoneum in the pelvis and abdomen. After thorough suction and irrigation of the pelvic organs, normal appearing fallopian tubes and ovaries were observed bilaterally with no evidence of ectopic pregnancy (Figure 3). A right ovarian hemorrhagic cyst was seen laparoscopically, consistent with the TVUS performed in the Emergency Department. As no pelvic source of bleeding was identified, attention was turned to the remainder of the abdomen. An ectopic pregnancy in the greater omentum measuring approximately 1 cm with active bleeding from the placental bed was visualized (Figure 4). Upon surgical manipulation, chorionic villi aborted from the implantation site (Figure 5). The ectopic pregnancy was removed from the abdomen under direct visualization using graspers. The LigaSure device was then used to resect the bleeding placental bed from the remaining omentum (Figure 6). An Endo Catch bag was used to remove the omental placental bed from the abdomen. Visual inspection of the surgical specimens in the operating room was consistent with an ectopic pregnancy. Pathology evaluation was also consistent with an omental ectopic pregnancy. Following laparoscopic removal of the ectopic pregnancy, the patient’s βhCG levels were followed to zero over the course of three weeks. The patient gave permission for her case to be published in a deidentified article. The University of Connecticut Health Center Institutional Review Board does not require approval for case reports.
Discussion
While ectopic pregnancies comprise approximately 2% of all reported pregnancies, abdominal pregnancies comprise only 0.61–1.3% of ectopic pregnancies [3],[10],[11]. However, the mortality rate of abdominal ectopic pregnancies is 8-fold higher than that of tubal ectopic pregnancies, ranging from 0.5% to 18% depending on the gestational age at diagnosis and treatment method [11]. Additionally, heterotopic pregnancies have an incidence of 1/4,000 to 1/50,000 spontaneously conceived pregnancies and up to 1/100 pregnancies following fresh or frozen embryo transfer [5],[12].
A review of the literature was performed to identify all reported cases of omental ectopic pregnancy following IVF with fresh ET or FET. The literature search was performed in PubMed, Ovid, and SCOPUS using the search terms “omental pregnancy” or “omental ectopic” and “IVF” or “in vitro fertilization” without language or study design restriction through January 2023. After removal of duplicates, 22 articles were screened, and 20 were excluded due to abdominal implantation at a site other than the omentum. Reference lists of the 20 remaining articles were then reviewed for other pertinent publications.
Although omental ectopic pregnancies have been reported following natural conceptions, this is the first report, to our knowledge, of a primary omental ectopic pregnancy following IVF and FET. There are, however, three published reports of heterotopic pregnancies involving an omental ectopic pregnancy following fresh ET of multiple embryos [8],[13]. In 2003, Cormio et al. published a report of a 30-year-old G2P0 status post bilateral salpingectomy for bilateral tubal ectopic pregnancies who underwent fresh transfer of four embryos [6]. First trimester ultrasound demonstrated a heterotopic pregnancy and an 8 cm amniotic sac was removed from the omentum at laparotomy. A second case of a heterotopic pregnancy following fresh ET involving secondary implantation on the omentum was reported by Ali et al. in 2006 [7]. In this case, a 35-year-old G0 with bilateral tubal occlusion underwent IVF followed by fresh ET of a single embryo and was subsequently diagnosed with simultaneous right tubal and omental ectopic pregnancies, both of which were managed surgically. In 2022, Karampas et al. reported a third case of an omental heterotopic pregnancy involving a 32-year-old G0 with a history of bilateral salpingectomy who underwent fresh ET of two embryos [8]. The patient became acutely hemodynamically unstable at six weeks gestational age and a ruptured omental ectopic pregnancy was removed during laparoscopy, followed by cesarean delivery at 37 weeks of her intrauterine pregnancy.
These three heterotopic pregnancies resulted from fresh rather than frozen embryo transfers and involved secondary omental pregnancies that could have originated from spontaneous conceptions. In two of the heterotopic cases following IVF with fresh ET, primary implantation was in the uterine fundus while in one case primary implantation was in the fallopian tube. Additionally, all of these cases were of patients with tubal factor infertility, which increased their risk of ectopic pregnancy. In contrast, the patient in our case of primary omental pregnancy had severe male factor infertility, making spontaneous conception highly unlikely, and no known risk factors for ectopic pregnancy.
Serum βhCG is secreted by placental syncytiotrophoblasts and is used as a marker for early pregnancy development [14]. Early in pregnancy, serum βhCG values increase in a curvilinear manner until they plateau at approximately 100,000 IU/L, around 10–11 weeks gestation [14],[15]. Barnhart et al. performed a retrospective cohort study of 285 women from diverse ethnic backgrounds who presented with first trimester vaginal bleeding and pain in the setting of pregnancy of unknown location who were ultimately diagnosed with a viable intrauterine pregnancy [15]. This study demonstrated that the minimum (1st percentile) rise in βhCG over 48 hours in viable intrauterine pregnancies is 49%, 40%, and 33% for initial βhCG values of <1,500, 1,500–3,000, and >3,000 IU/L, respectively [15]. This suggests that the traditional teaching that βhCG values should double every 48 hours in early pregnancy may be too strict [16]. Based on these criteria, our case of a primary omental ectopic pregnancy did meet the minimum expected rise in βhCG for a viable intrauterine pregnancy.
Compared to percent increases in βhCG, there is less data on absolute values for serum βhCG following implantation. The initial βhCG value for our case of an omental ectopic pregnancy was 2.2 IU/L and the repeat value two days later was 4.4 IU/L. By percent increase criteria, this βhCG trend exceeded the minimum increase of 49% for initial βhCG values <1,500 IU/L for an intrauterine pregnancy [15]. This patient’s βhCG value continued to increase in a pattern that surpassed the minimum percent increase criteria. However, by a gestational age of five weeks and zero days, or 16 days after FET, the patient’s βhCG was subjectively low at 124 IU/L. A retrospective study by Homan et al. concluded that a βhCG between 25 and 50 IU/L 16 days following ovulation in IVF pregnancies portends a <35% chance of ongoing pregnancy while a βhCG level >500 IU/L is associated with a >95% chance of ongoing pregnancy [17]. Meanwhile, an analysis of 417 pregnancies after fresh ET demonstrated that a βhCG > 55 IU/L measured 12 days after ET was associated with a 90% chance of a viable pregnancy [18]. Although our patient did not have a βhCG drawn 12 days after FET, her βhCG values 11 and 13 days after FET were 4.4 IU/L and 13.2 IU/L, respectively. Both of these levels are below the threshold of 55 IU/L. Furthermore, the importance of serially following βhCG levels between 1.0 and 5.0 IU/L was demonstrated in a retrospective cohort study by Maslow et al. in which six patients with initial βhCG values <5.0 IU/L, which were considered negative pregnancy tests, went on to develop ectopic pregnancies, five of which were ultimately managed with surgery [19].
It is our practice to monitor the percent increase in βhCG between days 9 and 11 following FET. When the percent rise exceeds minimum criteria for an intrauterine pregnancy, the βhCG level is repeated one week later. However, when the percent rise is below the threshold for normal or is subjectively low, βhCG levels are repeated every 1–2 days and the patient is closely monitored with serial TVUS. This case of an omental ectopic pregnancy demonstrates that despite βhCG values exceeding the minimal percent rise for a viable intrauterine pregnancy, low absolute βhCG levels should raise suspicion of abnormal implantation.
Conclusion
This is the first reported case of a primary omental ectopic pregnancy following a FET. Prior reports of omental ectopic pregnancies following ET were secondary omental pregnancies and could have resulted from a spontaneous conception. This case also highlights the need to maintain a high suspicion of an abnormal pregnancy when absolute βhCG levels are low despite an appropriate percent rise over 48 hours. Although omental ectopic pregnancies are rare, they must remain in the differential diagnosis when a patient is suspected to have an ectopic pregnancy and no adnexal masses are seen on TVUS even in the absence of risk factors, especially given the diagnostic challenges in identification.
REFERENCES
1.
Li Z, Sullivan EA, Chapman M, Farquhar C, Wang YA. Risk of ectopic pregnancy lowest with transfer of single frozen blastocyst. Hum Reprod 2015;30(9):2048–54. [CrossRef]
[Pubmed]

2.
Clayton HB, Schieve LA, Peterson HB, Jamieson DJ, Reynolds MA, Wright VC. Ectopic pregnancy risk with assisted reproductive technology procedures. Obstet Gynecol 2006;107(3):595–604. [CrossRef]
[Pubmed]

3.
Bouyer J, Coste J, Fernandez H, Pouly JL, Job-Spira N. Sites of ectopic pregnancy: A 10 year population-based study of 1800 cases. Hum Reprod 2002;17(12):3224–30. [CrossRef]
[Pubmed]

4.
Practice Committee of American Society for Reproductive Medicine. Medical treatment of ectopic pregnancy: A committee opinion. Fertil Steril 2013;100(3):638–44. [CrossRef]
[Pubmed]

5.
Barrenetxea G, Barinaga-Rementeria L, Lopez de Larruzea A, Agirregoikoa JA, Mandiola M, Carbonero K. Heterotopic pregnancy: Two cases and a comparative review. Fertil Steril 2007;87(2):417. e9–15. [CrossRef]
[Pubmed]

6.
Cormio G, Santamato S, Putignano G, Bettocchi S, Pascazio F. Concomitant abdominal and intrauterine pregnancy after in vitro fertilization in a woman with bilateral salpingectomy. A case report. J Reprod Med 2003;48(9):747–9.
[Pubmed]

7.
Ali CR, Fitzgerald C. Omental and peritoneal secondary trophoblastic implantation – An unusual complication after IVF. Reprod Biomed Online 2006;12(6):776–8. [CrossRef]
[Pubmed]

8.
Karampas G, Zouridis A, Deligeoroglou E, et al. Heterotopic pregnancy after bilateral salpingectomy, IVF and multiple embryos transfer. A case report and systematic review of the literature. J Obstet Gynaecol 2022;42(5):809–15. [CrossRef]
[Pubmed]

9.
Scadron EN. Primary peritoneal pregnancy. Am J Obstet Gynecol 1957;73(3):686–9. [CrossRef]
[Pubmed]

10.
Varma R, Gupta J. Tubal ectopic pregnancy. BMJ Clin Evid 2012;2012:1406.
[Pubmed]

11.
Tang P, Li X, Li W, Li Y, Zhang Y, Yang Y. The trend of the distribution of ectopic pregnancy sites and the clinical characteristics of caesarean scar pregnancy. Reprod Health 2022;19(1):182. [CrossRef]
[Pubmed]

12.
Maymon R, Shulman A. Controversies and problems in the current management of tubal pregnancy. Hum Reprod Update 1996;2(6):541–51. [CrossRef]
[Pubmed]

13.
Yoder N, Tal R, Martin JR. Abdominal ectopic pregnancy after in vitro fertilization and single embryo transfer: A case report and systematic review. Reprod Biol Endocrinol 2016;14(1):69. [CrossRef]
[Pubmed]

14.
Morse CB, Barnhart KT, Senapati S, et al. Association of the very early rise of human chorionic gonadotropin with adverse outcomes in singleton pregnancies after in vitro fertilization. Fertil Steril 2016;105(5):1208–14.e3. [CrossRef]
[Pubmed]

15.
Barnhart KT, Guo W, Cary MS, et al. Differences in serum human chorionic gonadotropin rise in early pregnancy by race and value at presentation. Obstet Gynecol 2016;128(3):504–11. [CrossRef]
[Pubmed]

16.
Wu Y, Liu H. Possibility of live birth in patients with low serum β-hCG 14 days after blastocyst transfer. J Ovarian Res 2020;13(1):132. [CrossRef]
[Pubmed]

17.
Homan G, Brown S, Moran J, Homan S, Kerin J. Human chorionic gonadotropin as a predictor of outcome in assisted reproductive technology pregnancies. Fertil Steril 2000;73(2):270–4. [CrossRef]
[Pubmed]

18.
Bjercke S, Tanbo T, Dale PO, Mørkrid L, Abyholm T. Human chorionic gonadotrophin concentrations in early pregnancy after in-vitro fertilization. Hum Reprod 1999;14(6):1642–6. [CrossRef]
[Pubmed]

19.
Maslow BSL, Bartolucci A, Sueldo C, Engmann L, Benadiva C, Nulsen JC 3rd. Occult abnormal pregnancies after first post-embryo transfer serum beta-human chorionic gonadotropin levels of 1.0-5.0 mIU/mL. Fertil Steril 2016;105(4):938–945.e1. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Katherine Koniares - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Prachi Godiwala - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Claudio Benadiva - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Andrea DiLuigi - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2024 Katherine Koniares et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.