|
Case Series
Ultrasound findings in pregnancies affected by placental perivillous fibrin deposition: A case series
1 OBGYN Resident, Advocate Lutheran General Hospital, Park Ridge, IL 60068, USA
2 Maternal Fetal Medicine Physician, Advocate Lutheran General Hospital, Park Ridge, IL 60068, USA
3 Maternal Fetal Medicine Physician, Advocate Christ Medical Center, Oak Lawn, IL 60453, USA
Address correspondence to:
Olivia Garcia
DO, Obstetrics & Gynecology Department, Advocate Lutheran General Hospital, Park Ridge, IL 60068,
USA
Message to Corresponding Author
Article ID: 100178Z08OG2024
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Garcia O, Kase B, Harrison R, Holmgren C. Ultrasound findings in pregnancies affected by placental perivillous fibrin deposition: A case series. J Case Rep Images Obstet Gynecol 2024;10(2):13–17.ABSTRACT
Introduction: This article presents unique ultrasound findings in patients with perivillous fibrin deposition on placental pathology. Massive perivillous fibrin deposition (MPVFD), characterized by excessive deposition of fibrinoid material in the placenta, is a rare finding on pathologic investigation and has been associated with gestational morbidity and mortality.
Case Series: We present three cases with abnormal prenatal ultrasound findings that were formally diagnosed with excessive perivillous fibrin deposition by postnatal pathologic specimen review. Cases 1 and 2 are of the same patient following subsequent pregnancies. The first pregnancy resulted in new fetal growth restriction, necessitating delivery at 34 weeks. The second pregnancy then warranted close sonographic monitoring. This pregnancy was overall uncomplicated until placental cysts appeared at 31 weeks. She had antenatal testing until her planned delivery at 37 weeks. Placental abnormality was noted at 24 weeks in the third case. She ultimately was delivered at 35 weeks with fetal growth restriction, non-reassuring fetal status, and preeclampsia with severe features.
Conclusion: Placental perivillous fibrin deposition is clinically significant given risks for fetal growth restriction and preterm delivery for fetal indications. Therefore, when ultrasound findings are suggestive of this pathology, the obstetrician should be prompted to increase surveillance of the given pregnancy.
Keywords: Case series, Fetal growth restriction, Fibrin deposition, Obstetrics, Placental pathology, Pregnancy, Ultrasound
Introduction
Perivillous fibrin deposition is a pathologic finding in the placenta that involves the accumulation of fibrin around the chorionic villi. The presence of fibrin deposits in the perivillous space can impair maternal–fetal exchange, leading to compromised transfer of nutrients and oxygen to the developing fetus. Although uncommon, this discovery is of critical importance as it has been associated with significant gestational morbidity and mortality, namely recurrent pregnancy loss, preterm birth, fetal growth restriction, and stillbirth [1],[2]. Traditionally diagnosed histologically, these deposits appear as eosinophilic material surrounding the villous structures, often associated with maternal or fetal vascular lesions. Clinically, perivillous fibrin deposition is associated with fetal growth restriction, preterm birth, and stillbirth. Often the diagnosis is made retrospectively after pathologic evaluation of the placenta. Little has been described in the literature regarding the antenatal findings associated with this condition.
Case Series
Case 1
The patient is a 38-year-old G2 P1001 who presented for a level 2 ultrasound at 20 weeks gestation in her second pregnancy. Her first pregnancy was a term vaginal delivery complicated by small for gestational age infant (5 lb 9 oz) that developed hypoxic ischemic encephalopathy following delivery. The patient is otherwise healthy without any chronic medical issues or relevant family history of bleeding disorders, thrombosis or birth defects. She had low risk aneuploidy screening with cell free DNA testing, a normal alpha-fetoprotein (AFP) result and a normal 1-hour 50 g Glucola. The detailed anatomic ultrasound demonstrated a singleton intrauterine pregnancy with estimated fetal weight of 360 g (66 percentile) and normal fluid volume, placental appearance and fetal anatomy.
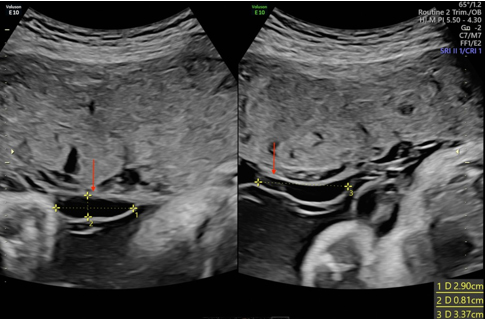
Given her history, the patient was advised to return for a follow-up ultrasound to assess fetal growth for which she presented at 30-week gestation. A follow-up ultrasound demonstrated estimated fetal weight (EFW) of 1494 g (30 percentile) with normal fluid volume. However, the appearance of the placenta was notable for thin-appearing membranous bands or cysts along the sub-amniotic edge of the placenta measuring 9.36 cm × 3.1 cm (Figure 1). Normal umbilical artery Doppler flow was noted.
The patient was admitted to the hospital for fetal monitoring and given betamethasone due to concern for possible preterm delivery, as at this time our team was concerned about significant placental dysfunction based on the ultrasound findings. Fetal heart rate monitoring was reactive and reassuring, and umbilical artery Dopplers remained normal. The patient was discharged and followed closely as an outpatient with twice weekly non-stress tests, umbilical artery Doppler assessments, and serial ultrasounds for fetal growth.
At 34 weeks gestation, the follow-up ultrasound demonstrated fetal growth restriction with EFW of 1974 g (7 percentile overall and AC 8 percentile). Amniotic fluid volume and umbilical artery Doppler measurements were normal. A large septate cystic area was again seen, now measuring 13.96 cm × 7.21 cm (Figure 2).
The patient was admitted to the hospital for planned induction of labor given the increasing size of the placental cystic area and new onset fetal growth restriction. At the time of admission, she was found to be 3 cm dilated, so Pitocin was initiated per hospital protocol for induction of labor. The patient was also given ampicillin as the Group B Streptococcus (GBS) status was unknown. She progressed in labor and delivered a 1930-g infant with APGARs of 9 and 9. Placental delivery required manual extraction as well as curettage. The cystic area seen on ultrasound was noted to rupture during delivery with about 300 cc of blood-tinged fluid. The infant was in the neonatal intensive care for 10 days and subsequently discharged in good condition.
Placental pathology was performed with the following findings: small for gestational age, fragmented placenta (281.2 g; expected, 389 g) with massive perivillous fibrin deposition/maternal floor infarct, cystification and villous fibrosis. Membranes were also noted to have associated decidual necrosis; otherwise, no pathological change. Umbilical cord with no pathological change.
Case 2
The same patient in Case 1 became pregnant again two years following the delivery noted above, now 40 years old and a G3P1102. In the interim, she underwent evaluation for possible causes of the above growth restriction and placental pathology and was found to have negative findings for antiphospholipid syndrome, normal thyroid function, and normal lipid panel assessment. Viable intrauterine pregnancy was established with ultrasound, and she was started on a daily low dose aspirin and low molecular weight heparin. The patient was also started on hydroxychloroquine, but this was discontinued at 16 weeks gestation secondary to maternal side effects. Aneuploidy screening and AFP testing were declined.
At 20 weeks, the patient had a detailed anatomic ultrasound with estimated fetal weight of 361 g (70 percentile), normal fluid volume and normal appearing fetal anatomy. The anterior placenta appeared normal.
The patient had serial ultrasounds for fetal growth every three weeks and non-stress testing initiated at 30 weeks for the remainder of the pregnancy. Ultrasound demonstrated normal fetal growth and placental appearance until 31 weeks when two small anechoic cysts were noted in the sub-amniotic space measuring 1.1 cm and 0.7 cm in largest diameter, respectively. A follow-up ultrasound was performed one week later which now revealed three cystic structures increasing in size (Figure 3 and Figure 4). Ultrasound for growth at 34 weeks demonstrated an EFW of 2443 g (61 percentile) and normal fluid volume. The placental findings continued to demonstrate enlarging cystic areas. The patient was followed closely, with reassuring fetal monitoring with the recommendation for delivery at 37 weeks.
The patient was admitted to the hospital for induction of labor at 37 weeks gestation. She had a vaginal delivery of a 2830 g infant with APGARs of 8 and 9. The placenta delivered spontaneously and was sent to pathology. The infant was discharged home with mom in good condition.
Placental pathology was performed and demonstrated a “histologically mature singleton placenta, 546 g, with three-vessel umbilical cord, massive perivillous fibrin deposition, and few subamniotic cysts, negative for acute chorioamnionitis.”
Case 3
The patient is a 34-year-old G4 P0030 who presented for prenatal care at 10 weeks. She had a low-risk cell free DNA screening, but an elevated AFP of 6.49 multiple of the median (MoM). The patient met with Maternal-Fetal Medicine (MFM) and had a 17 week ultrasound that demonstrated normal growth and no concern for neural tube defect or abdominal wall defect. There was a moderate sized maternal right adnexal mass noted suggestive of a dermoid cyst.
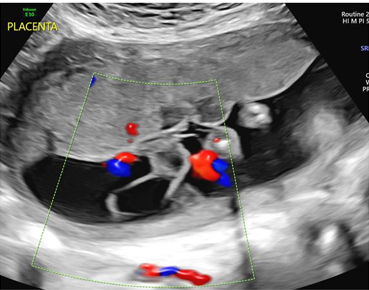
Detailed anatomy ultrasound performed at 20 weeks demonstrated EFW 321 g (41 percentile), normal fluid volume and an anterior placenta. Fetal anatomic survey appeared normal. However, there were two cystic structures noted on fetal side of placenta measuring 2.1 cm and 4.2 cm in largest dimension, respectively; the latter cyst with septations (Figure 5). No internal color flow observed. She was scheduled for a follow-up ultrasound in four weeks. This follow-up ultrasound at 24-weeks then revealed new onset fetal growth restriction (EFW 536 g, 6 percentile, normal fluid and UA Dopplers) and persistence of the aforementioned cystic structures. Her care was transferred to MFM and she was scheduled for weekly testing with ultrasound. Antiphospholipid antibody testing was also performed and was negative.
Serial ultrasound for growth was performed every two weeks and noted persistent fetal growth restriction. Antenatal testing continued to be reassuring. The placental cysts increased in size with measurements at 29 weeks of (1) 5.16 cm × 2.78 cm and (2) 9.96 cm × 3.68 cm (Figure 6) with a 1.6 cm hyperechoic structure within the previously mentioned simple cystic structures.
At 35 weeks, the patient presented for antenatal testing with new onset elevated blood pressure measurements with readings in the 150s/100s mmHg. She was sent to obstetric (OB) triage for evaluation where she had persistent severe range blood pressures and intermittent fetal heart rate decelerations. A decision was made to proceed with cesarean delivery at this time secondary to the fetal heart rate tracing. The patient was started on magnesium sulfate for seizure prophylaxis and taken to the operating room with delivery of a female infant weighing 1830 g with APGARs of 6, 8, and 8. Per the operative report, the amniotic fluid was “dark and chocolate colored.” The patient also had a right oophorectomy for the previously noted adnexal mass.
Placental pathology resulted with blue gray appearance, extensive subchorionic fibrin on the fetal side. The maternal surface consisted of red-brown, spongy cut surfaces with diffuse fibrin trabeculation, compromising approximately 80–90% of the placental volume.
Discussion
Massive perivillous fibrin deposition (MPVFD) in the placenta was first reported in 1967 and refers to the accumulation of fibrinoid material around the chorionic villi within the intervillous space [3]. This phenomenon is often associated with abnormal placental development and can have significant implications for both maternal and fetal health [4] .
Normally, fibrin is involved in the physiologic processes of blood clotting and wound healing. However, when it accumulates excessively in the placenta, it can hinder proper nutrient and oxygen exchange between the maternal blood and fetal vessels [5]. This excess fibrin within the placenta may compromise its function, potentially leading to inadequate fetal growth and development. The reported risk of fetal death in MPVFD ranges from 15 to 80%, preterm delivery is seen in 50 to 70% of cases, and fetal growth restriction noted in 30–50% of cases [6]. Causes of poor perinatal outcomes are still poorly understood, although several avenues have been explored. Therefore, understanding and monitoring suspected perivillous fibrin deposition antenatally are crucial aspects of assessing placental health and ensuring optimal outcomes for both the mother and the developing fetus.
As mentioned above, the underlying mechanisms for perivillous fibrin deposition are poorly studied and there is no biomarker available for the diagnosis of this condition. Current theories suggest an autoimmune cause; MPVFD has been found in association with antiphospholipid syndrome and polymyositis [7],[8]. It has also been reported that MPVFD may be associated with an imbalance of angiogenic/anti-angiogenic factors [9]. In addition to uncertainty about the underlying cause, there is no available evidence regarding the optimal therapeutic approach to manage MPVFD. There are very limited data regarding protocols or therapies to prevent this condition. Prior published cases have utilized the administration of aspirin, heparin, hydroxychloroquine, and intravenous immunoglobulins with varying degrees of success reported.
Because MPVFD is diagnosed based on histological examination of the placenta, antenatal detection is often limited and found later in pregnancy. There is no specific diagnostic marker for this condition, although some case studies demonstrated an association between elevated AFP in the second trimester and MPVFD [10]. Ultrasound suspicion for this condition is generally limited to non-specific findings of fetal growth restriction, abnormal umbilical artery Doppler waveforms, and oligohydramnios [11],[12]. However, we report the presence of unique ultrasound findings of large sub-amniotic placental cysts that correlate with the histological findings of MPVFD and the clinical findings associated with this histopathologic diagnosis. To our knowledge, these findings have not been reported previously.
Conclusion
The described findings on prenatal ultrasound are associated with pathologically identified MPVFD and should prompt obstetricians to increase surveillance of the pregnancy including more frequent ultrasound assessment and antenatal testing, counsel the patient regarding the potential risks, consider delivery at an earlier gestational age, and obtain a thorough pathologic evaluation of the placenta after delivery.
REFERENCES
1.
Katzman PJ, Genest DR. Maternal floor infarction and massive perivillous fibrin deposition: Histological definitions, association with intrauterine fetal growth restriction, and risk of recurrence. Pediatr Dev Pathol 2002;5(2):159–64. [CrossRef]
[Pubmed]

2.
Faye-Petersen OM, Ernst LM. Maternal floor infarction and massive perivillous fibrin deposition. Surg Pathol Clin 2013;6(1):101–14. [CrossRef]
[Pubmed]

3.
Fox H. Perivillous fibrin deposition in the human placenta. Am J Obstet Gynecol 1967;98(2):245–51. [CrossRef]
[Pubmed]

4.
Chen A, Roberts DJ. Placental pathologic lesions with a significant recurrence risk – What not to miss! APMIS 2018;126(7):589–601. [CrossRef]
[Pubmed]

5.
Qi M, En Chang KT, Quan Lian DW, Khoo CK, Tan KH. Placental massive perivillous fibrinoid deposition is associated with adverse pregnancy outcomes: A clinicopathological study of 12 cases. Case Reports in Perinatal Medicine 2016;5(1):35–9. [CrossRef]

6.
Cheloufi M, Coulomb A, Abisror N, et al. Massive perivillous fibrin deposition: Diagnosis, obstetrical features, and treatment. Eur J Obstet Gynecol Reprod Biol 2024;292:125–32. [CrossRef]
[Pubmed]

7.
Spinillo A, Gardella B, Muscettola G, Cesari S, Fiandrino G, Tzialla C. The impact of placental massive perivillous fibrin deposition on neonatal outcome in pregnancies complicated by fetal growth restriction. Placenta 2019;87:46–52. [CrossRef]
[Pubmed]

8.
Hung NA, Jackson C, Nicholson M, Highton J. Pregnancy-related polymyositis and massive perivillous fibrin deposition in the placenta: Are they pathogenetically related? Arthritis Rheum 2006;55(1):154–6. [CrossRef]
[Pubmed]

9.
Whitten AE, Romero R, Korzeniewski SJ, et al. Evidence of an imbalance of angiogenic/antiangiogenic factors in massive perivillous fibrin deposition (maternal floor infarction): A placental lesion associated with recurrent miscarriage and fetal death. Am J Obstet Gynecol 2013;208(4):310.e1–e11. [CrossRef]
[Pubmed]

10.
Devisme L, Chauvière C, Franquet-Ansart H, et al. Perinatal outcome of placental massive perivillous fibrin deposition: A case-control study. Prenat Diagn 2017;37(4):323–8. [CrossRef]
[Pubmed]

11.
Chang P, Millar D, Tsang P, Lim K, Houlihan E, Stephenson M. Intravenous immunoglobulin in antiphospholipid syndrome and maternal floor infarction when standard treatment fails: A case report. Am J Perinatol 2006;23(2):125–9. [CrossRef]
[Pubmed]

12.
Lampi K, Papadogiannakis N, Sirotkina M, Pettersson K, Ajne G. Massive perivillous fibrin deposition of the placenta and pregnancy outcome: A retrospective observational study. Placenta 2022;117:213–8. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Olivia Garcia - Conception of the work, Design of the work, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Benjamin Kase - Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Rachel Harrison - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Calla Holmgren - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2024 Olivia Garcia et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.