|
Case Report
Chronic ectopic pregnancy after intrauterine insemination: A case report and review of the literature
1 UConn Health, Department of Obstetrics and Gynecology, Division of Reproductive Endocrinology and Infertility, Farmington, CT, USA
2 Center for Advanced Reproductive Services, Farmington, CT, USA
3 UConn Health, Department of Obstetrics and Gynecology, Division of Minimally Invasive Gynecologic Surgery, Farmington, CT, USA
4 UConn Health, Department of Obstetrics and Gynecology, Division of Reproductive Endocrinology and Infertility, Farmington, C, USA
Address correspondence to:
Melody So
MD, Center for Advanced Reproductive Services, Division of Reproductive Endocrinology and Infertility, University of Connecticut School of Medicine, 2 Batterson Park Road, Farmington, CT 06032,
USA
Message to Corresponding Author
Article ID: 100204Z08MS2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
So M, Koniares K, Nilsson W, Schmidt D, Luciano D. Chronic ectopic pregnancy after intrauterine insemination: A case report and review of the literature. J Case Rep Images Obstet Gynecol 2025;11(1):77–82.ABSTRACT
Introduction: Chronic ectopic pregnancy is a rare form of ectopic pregnancy characterized by repeated tubal injury and bleeding, leading to development of a chronic, inflammatory pelvic mass. Patients often present with mild, nonspecific symptoms, and a low beta-human chorionic gonadotropin (βhCG) level. Despite the established association between assisted reproductive technology and increased risk of ectopic pregnancy, this is the first reported case of a chronic ectopic pregnancy following intrauterine insemination.
Case Report: A 27-year-old primigravid female with primary infertility in the setting of polycystic ovary syndrome initially developed an ectopic pregnancy after underdoing ovulation induction and intrauterine insemination. After successful treatment with methotrexate, she was later found to have a complex pelvic mass despite a negative βhCG level, suggestive of chronic ectopic pregnancy. She underwent laparoscopic left salpingectomy with excision of adnexal mass and diagnosis was confirmed on intraoperative and histopathological findings.
Conclusion: Chronic ectopic pregnancy is a rare complication of assisted reproductive technology. Nonetheless, it should be considered in the setting of a new pelvic mass, negative pregnancy test, and recent history of ectopic pregnancy. Although conservative management is attractive, definitive treatment involves removal of the pelvic hematocele, effectively achieved through surgical intervention.
Keywords: Chronic ectopic pregnancy, Ectopic pregnancy, Intrauterine insemination, Methotrexate, Tubal pregnancy
Introduction
The incidence of ectopic pregnancies (EPs) is approximately 2% of all reported pregnancies, and of these, the majority are classified as acute. Chronic ectopic pregnancy (CEP) is a less common subtype of EP that differs from acute EP in clinical presentation as well as sonographic, intraoperative, and histopathologic findings. Chronic ectopic pregnancy is associated with high rates of misdiagnosis, which may be attributable to its unique pathophysiology [1]. Furthermore, there is currently no formal definition, diagnostic criteria, or treatment guideline for CEP. As such, the true incidence is difficult to ascertain but has been reported to be between 6% and 20% of all EPs [2],[3],[4],[5],[6].
Compared with acute EP, patients with CEP are more likely to be hemodynamically stable and clinically well-appearing on initial presentation and at time of diagnosis. The most common presenting symptoms of CEP include amenorrhea, abdominal pain, and abnormal uterine bleeding. Although these are also classic manifestations of acute EP, CEP develops in a more indolent manner and is generally associated with milder, subacute symptoms [2],[3],[4],[5],[7]. In one study, approximately 18% of patients with CEP were asymptomatic [3]. Although rare, CEP can present with acute rupture and hemodynamic decompensation, necessitating emergent surgical intervention [8]. Other patients with CEP, however, remain asymptomatic for months or years, or never develop symptoms to prompt further diagnostic workup [9],[10],[11].
The pathogenesis of CEP is thought to involve small, repeated ruptures of a tubal pregnancy, which cause localized hemorrhages and subsequent breakdown of the tubal wall over a prolonged period. This ultimately leads to formation of a pelvic hematocele, or walled-off mass comprised of hematoma and degenerated chorionic villi with areas of necrosis, hemorrhage, and fibrosis on histopathology. Chronic inflammation leads to the classic intraoperative finding of dense adhesions formed between the hematocele and surrounding structures, including the omentum, ileum, cecum, sigmoid colon, and pelvic sidewall. As a result, lysis of adhesions and surgical removal can be quite challenging.
Histopathologic evaluation of resected CEP tissue reveals characteristic inactive, avital, or degenerated chorionic villi that have diminished or absent trophoblastic activity [2],[4]. Elsewhere this is described as necrotic, hyalinized ghost villi [11],[12]. As a result of trophoblastic quiescence, there is decreased hormone synthesis, which accounts for the characteristic finding of low or undetectable serum beta-human chorionic gonadotropin (βhCG) levels. Early retrospective studies reported a high incidence of negative pregnancy tests associated with CEP; however, these utilized low-sensitivity assays with higher detection thresholds [4],[7]. Today, more sensitive hCG immunoassays demonstrate the majority of patients with CEP have detectable serum βhCG levels, although often lower than levels seen with acute EP. Nonetheless, a diagnosis of CEP cannot be ruled out in the setting of undetectable βhCG titers [8],[13],[14].
Despite low levels of βhCG, a key sonographic feature of CEP is a complex echoic pelvic mass [3],[5],[7],[15]. This finding is attributed to varying components of fresh blood and organized hematoma. The most common location is the adnexa, often extending to the posterior cul-de-sac, although there are case reports of CEP in the cornua and ovary [16],[17]. Color Doppler flow may show enhanced peripheral vascularity, which can mistakenly suggest the diagnosis of tubo-ovarian abscess, endometrioma, complex ovarian cyst, leiomyoma, or pelvic malignancy, particularly in the context of a negative pregnancy test [18].
The mainstay of CEP treatment is surgical management. In the past, patients, including those who were hemodynamically stable, often underwent exploratory laparotomy. Today, however, laparoscopy with unilateral salpingectomy is the preferred approach [3]. Some patients may require unilateral salpingo-oophorectomy and, rarely, hysterectomy. As evidenced by several reports of concurrent appendectomy, more extensive procedures may be indicated depending on the degree of adhesions, structures involved, and extent of tissue damage discovered intraoperatively [4],[5].
Case Report
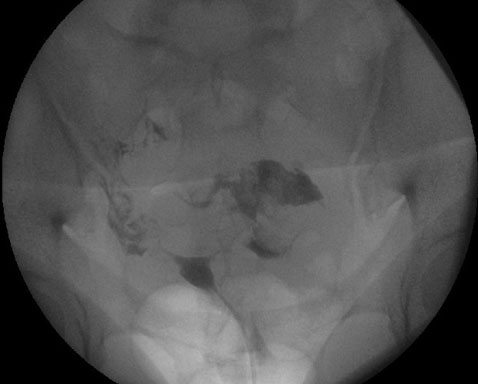
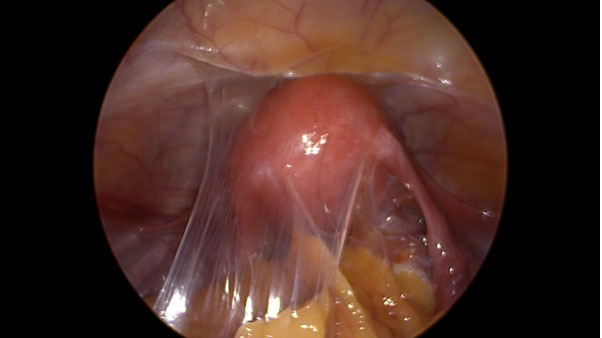
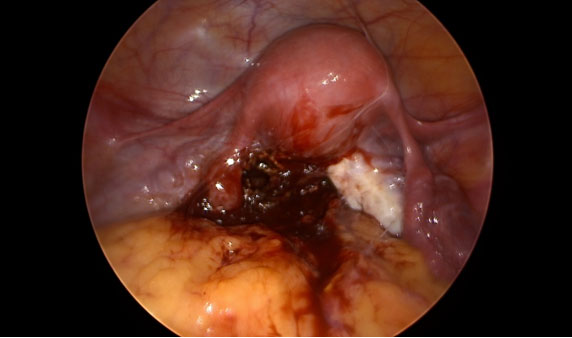
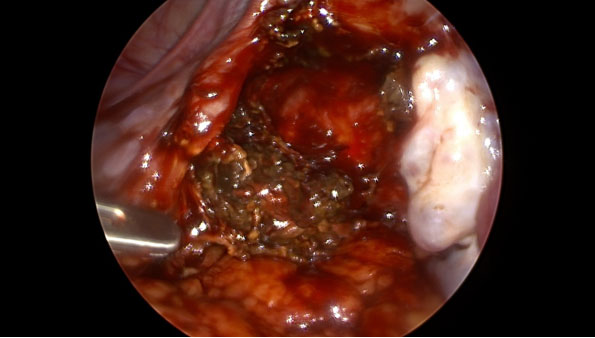
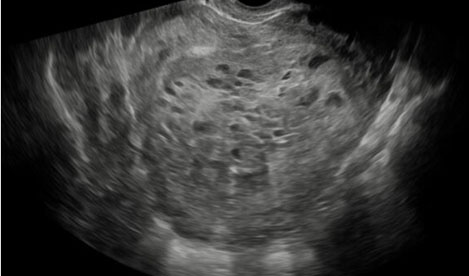
A 27-year-old nulliparous female with a history of primary infertility secondary to polycystic ovary syndrome (PCOS) underwent ovulation induction with clomiphene citrate and intrauterine insemination (IUI). Two weeks later, she had a βhCG level of 41.1 IU/L. Serial βhCG levels initially showed a small decline, followed by an appropriate rise. At approximately six weeks gestation, a small left adnexal ectopic pregnancy was visualized on transvaginal ultrasound with a corresponding βhCG level of 2,287 IU/L. Uterine aspiration was performed and showed no evidence of chorionic villi, confirming the diagnosis of ectopic pregnancy. The patient was asymptomatic and treated with intramuscular (IM) methotrexate (MTX) at a dose of 50 mg/m2. Monitoring of βhCG levels revealed a small rise from 4,003 to 4,324 IU/L between days four and seven of the MTX protocol; therefore, a second dose of MTX was administered. βhCG levels subsequently decreased appropriately and were undetectable (<1 IU/L) two months after treatment. The patient’s menses resumed and hysterosalpingography (HSG) was performed, revealing irregular filling of the left fallopian tube (Figure 1) and transvaginal ultrasound demonstrated a complex left adnexal mass measuring 8 × 6 cm (Figure 2). Given the patient’s hemodynamic stability and asymptomatic presentation, she was expectantly managed with sonographic surveillance, which showed a persistent, albeit slightly smaller, left adnexal mass three weeks later. Tumor markers were negative for malignancy. The patient was referred to minimally invasive gynecologic surgery (MIGS) for management of suspected CEP and she underwent diagnostic laparoscopy, left salpingectomy with removal of ectopic pregnancy, lysis of adhesions, hysteroscopy, and dilation and curettage. Intraoperative findings revealed significant pelvic adhesions, a grossly dilated and ruptured left fallopian tube, as well as a pelvic mass occupying the posterior cul-de-sac that appeared to consist of blood clots, placental and decidual tissue (Figure 3, Figure 4, Figure 5). Histological examination of the fallopian tube showed decidual changes in the submucosa with evidence of extensive necrosis, hemorrhage, and chronic inflammation. These findings, in conjunction with the patient’s clinical presentation, are consistent with a diagnosis of CEP. The patient’s consent was provided for publication of this report.
Discussion
Incidence of Ectopic Pregnancy and Assisted Reproductive Technology
To our knowledge, this is the first documented case of CEP after ovulation induction and IUI. The literature review revealed only one other case of CEP following assisted reproductive technology (ART) in a patient who underwent in vitro fertilization (IVF) followed by transfer of two frozen embryos [16]. The dearth of reported CEPs following ART is unexpected given that ART utilization is a risk factor for EP, and patients seeking fertility treatment may be predisposed to additional risk factors for abnormal implantation, such as a history of tubal disease, tubal surgery, and/or age greater than 35 years [19].
Numerous studies have demonstrated significantly higher rates of EP associated with IVF compared with IUI [20]. With respect to IUI, stimulated cycles may increase the risk of EP compared with natural cycles [21]. A study by Cirillo et al. published in 2022 reported the incidence of EP among women undergoing IVF or IUI and determined 1.80% of all pregnancies were ectopic, 7.58% of which resulted from IUI and the remainder from IVF [20].
Pathogenesis of Chronic Ectopic Pregnancy
Review of the literature demonstrates a paucity of information and limited insight into the etiology of CEP. This case report brings attention to two salient assertions regarding the pathogenesis of CEP that have remained poorly understood: 1) CEP is a de novo process and 2) it is associated with methotrexate resistance [3],[14]. Due to its characteristic clinical course and histopathological findings, CEP has historically been described as an entity independent from acute EP [3],[14]. Furthermore, a clear distinction has been made between chronic and persistent EP, which specifically refers to residual active trophoblastic tissue following conservative surgical or medical management [19].
In a case report and systematic review published in 2019, Tempfer et al. suggested there may, in fact, be more overlap between CEPs and acute EPs that develop chronic features because of methotrexate resistance, delayed diagnosis, and/or delayed treatment. Our case report supports this theory: the patient was diagnosed with and successfully treated for an ectopic pregnancy following IUI, then was subsequently diagnosed with a CEP after an asymptomatic period with return of menses. Indeed, there are reports of patients who were successfully treated with medical management for a suspected EP and were later diagnosed with a CEP after surgical intervention [3],[14],[22],[23].
Despite being widely purported, a review of the literature showed a lack of robust evidence to support MTX resistance in CEP. Our case report, in addition to those described above, may lend support to the hypothesis that a feature of CEP is an element of MTX tolerance or resistance. Additionally, these cases share a common factor in that they were all initially suspected to be acute EPs and were managed as such. Clinically, this is consistent with the theory that CEP is not solely a de novo process, but also perhaps a late sequelae of acute EP following inadequate or ineffective treatment.
Treatment of Chronic Ectopic Pregnancy
In the context of a subfertile population, the potential for conservative management whether medically with IM MTX or surgically with salpingostomy, remains desirable. However, there were very few cases noted in the literature of CEP treated initially with these methods. One case report published by Santos et al. in 2021 describes a patient who underwent medical management with a fixed multiple-dose MTX protocol for suspected CEP [24]. Despite normalization of hCG levels, persistence of the mass was noted on ultrasound surveillance. Thus, there remains a risk of acute rupture and continued inflammation after medical management.
Following conservative surgical management, the American College of Obstetricians and Gynecologists (ACOG) emphasizes the importance of close follow-up with serial βhCG monitoring to ensure resolution of any ectopic trophoblastic tissue that may persist after tube-sparing surgery [19]. However, given that CEP may present with undetectable βhCG levels, post-treatment surveillance may not be feasible, thus posing a significant barrier to this treatment approach.
Similarly, both ACOG and the American Society for Reproductive Medicine recommend against routine post-treatment sonographic surveillance due to lack of clinical utility. Following MTX administration for acute EPs, sonographic findings may include fluctuations in size, shape, and echogenicity of the ectopic mass as well peripheral vascularity surrounding the EP [25]. More commonly, an interval increase in size is observed, which has been found to have no association with ultimate success of treatment [26]. Although no large-scale studies have evaluated sonographic patterns of CEPs, the case study by Santos et al. suggests they may exhibit a similar pattern of progression as acute EPs. Furthermore, resolution of the ectopic pregnancy mass typically lags behind normalization of βhCG levels, from several weeks to months [25],[26].
Currently, there does not seem to be sufficient evidence to support the safety or efficacy of conservative management of CEP. CEP can often present with undetectable βhCG levels and treatment success of EP, with primary medical management, is universally defined as resolution of serum βhCG levels without the need for surgical intervention [19]. In the setting of CEP, the adnexal mass, representing the pelvic hematocele, may persist for much longer. Therefore, it is reasonable to infer that sonographic findings are also not an optimal indicator of treatment success. Perhaps it would be more appropriate to have different definitions of treatment success for acute versus chronic EP.
Lastly, there is currently not enough evidence to understand, or reliably predict, the short- and long-term sequelae of expectantly managing a persistent CEP despite undetectable βhCG levels. Given that chronic inflammation is the main driver of its pathogenesis, expectant management may increase potential for intra-abdominal adhesion development that could impose further harm to fertility.
Conclusion
Although less prevalent than acute ectopic pregnancy, CEP remains a possibility and should be considered in the differential diagnosis of a premenopausal patient presenting with mild, nonspecific symptoms, a new pelvic mass, and low or undetectable βhCG levels. This case report and review clarifies that patients who have recently undergone treatment of an ectopic pregnancy should not be excluded. Adequate understanding of the pathogenesis and pathophysiology of CEP is important for timely diagnosis and appropriate management. Currently, there is insufficient evidence to support a recommendation for conservative medical management of CEP. Definitive treatment, therefore, necessitates surgical intervention.
REFERENCES
1.
O'Neill D, Pounds R, Vella J, Singh K, Yap J. The diagnostic conundrum of chronic ectopic pregnancy: A case report. Case Rep Womens Health 2018;20:e00086. [CrossRef]
[Pubmed]

2.
Levy NB, Goldberger SB, Batchelder CS. Chronic ectopic pregnancy. A survey of 54 cases. S Afr Med J 1984;65(18):727–9.
[Pubmed]

3.
Tempfer CB, Dogan A, Tischoff I, Hilal Z, Rezniczek GA. Chronic ectopic pregnancy: Case report and systematic review of the literature. Arch Gynecol Obstet 2019;300(3):651–60. [CrossRef]
[Pubmed]

4.
Cole T, Corlett RC Jr. Chronic ectopic pregnancy. Obstet Gynecol 1982;59(1):63–8.
[Pubmed]

5.
Uğur M, Turan C, Vicdan K, Ekici E, Oğuz O, Gökmen O. Chronic ectopic pregnancy: A clinical analysis of 62 cases. Aust N Z J Obstet Gynaecol 1996;36(2):186–9. [CrossRef]
[Pubmed]

6.
7.
Bedi DG, Fagan CJ, Nocera RM. Chronic ectopic pregnancy. J Ultrasound Med 1984;3(8):347–52. [CrossRef]
[Pubmed]

8.
Brennan DF, Kwatra S, Kelly M, Dunn M. Chronic ectopic pregnancy—Two cases of acute rupture despite negative beta hCG. J Emerg Med 2000;19(3):249–54. [CrossRef]
[Pubmed]

9.
Curry NS, Blackwood GA, Tsai CC. Diagnosis of chronic ectopic gestation by hysterosalpingography. Abdom Imaging 1999;24(1):98–9. [CrossRef]
[Pubmed]

10.
11.
Avery DM, Silverman JF, Mazur MT. Retained fetal bones in chronic tubal pregnancy. Am J Obstet Gynecol 1984;149(7):794–5. [CrossRef]
[Pubmed]

12.
Abramov Y, Nadjari M, Shushan A, Prus D, Anteby SO. Doppler findings in chronic ectopic pregnancy: Case report. Ultrasound Obstet Gynecol 1997;9(5):344–6. [CrossRef]
[Pubmed]

13.
Porpora MG, Alò PL, Cosmi EV. Unsuspected chronic ectopic pregnancy in a patient with chronic pelvic pain. Int J Gynaecol Obstet 1999;64(2):187–8. [CrossRef]
[Pubmed]

14.
Dunn RC, Taskin O. Chronic ectopic pregnancy after clinically successful methotrexate treatment of ectopic pregnancy. Int J Gynaecol Obstet 1995;51(3):247–9. [CrossRef]
[Pubmed]

16.
17.
Su CC, Tzeng CC, Huang KF. Chronic ovarian pregnancy mimicking an ovarian tumor diagnosed by peritoneal washing cytology: A case report. Acta Cytol 2009;53(2):195–7. [CrossRef]
[Pubmed]

18.
Omih E, Makris A, Choy C, Bhuskute N. Chronic ectopic pregnancy mimicking pelvic malignancy. BMJ Case Rep 2021;14(3):e233534. [CrossRef]
[Pubmed]

19.
Committee on Practice Bulletins—Gynecology. ACOG Practice Bulletin No. 191: Tubal ectopic pregnancy. Obstet Gynecol 2018;131(2):e65–77. [CrossRef]
[Pubmed]

20.
21.
Bu Z, Xiong Y, Wang K, Sun Y. Risk factors for ectopic pregnancy in assisted reproductive technology: A 6-year, single-center study. Fertil Steril 2016;106(1):90–4. [CrossRef]
[Pubmed]

22.
Kasaven LS, Shah A, Sadoon S. Chronic tubal ectopic pregnancy following clinically successful methotrexate treatment for an acute ectopic: A review of the literature. J Obstet Gynaecol 2019;39(4):562–3. [CrossRef]
[Pubmed]

23.
24.
25.
Revzin MV, Pellerito JS, Moshiri M, Katz DS, Nezami N, Kennedy A. Use of methotrexate in gynecologic and obstetric practice: What the radiologist needs to know. Radiographics 2021;41(6):1819–38. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Melody So - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Katherine Koniares - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Wesley Nilsson - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
David Schmidt - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Danielle Luciano - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Melody So et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.