|
Review Article
The effects of androgen therapy in women with premature ovarian insufficiency: A systematic review
1 Department of Obstetrics and Gynecology, McGill University, Montreal, Quebec, Canada
2 Royal College of Surgeons, Ireland
3 SIMS IVF Centre, Ireland
4 Sidney Liswood Health Science Library, Mount Sinai Hospital, Toronto, Ontario, Canada
5 Department of Obstetrics and Gynecology, University of Toronto, Toronto, Ontario, Canada
6 Department of Obstetrics and Gynecology, Mount Sinai Hospital, University of Toronto, Toronto, Ontario, Canada
Address correspondence to:
Dina Mohamed
Royal Victoria Hospital, 1001 Decarie Blvd, Montreal, Quebec H4A 3J1,
Canada
Message to Corresponding Author
Article ID: 100211Z08DM2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Mohamed D, Shaltout N, Gomaa H, Philippopoulos E, Mejia-Gomez J, Wolfman W. The effects of androgen therapy in women with premature ovarian insufficiency: A systematic review. J Case Rep Images Obstet Gynecol 2025;11(2):8–17.ABSTRACT
Background: Premature ovarian insufficiency (POI) is a disorder of increasing occurrence and has multiple long-term impacts on affected females’ health; one of which is sexual dysfunction. Testosterone has been shown to improve hypoactive sexual desire disorder in postmenopausal women. Traditional hormone replacement therapy (estrogen and progesterone) is known for its beneficial effects in these cases, but there is still no scientific evidence to support the use of androgen in this situation.
Objective: Critically review and assess the literature on the use of androgen preparations in women with premature ovarian insufficiency (POI).
Search Strategy: Database search included randomized controlled trials, prospective comparative, observational, non-randomized studies, and case series of 10 or more subjects with no time limit restrictions.
Selection criteria: Eligible trials included females below the age of 40, with amenorrhea >4 months, FSH >40 IU/L on two occasions separated by six weeks.
Results: 539 abstracts were double screened, identifying 6 eligible studies; 3 randomized prospective double blinded studies, one randomized placebo controlled, one prospective observational, and one retrospective study. Discrepancies were arbitrated by a third reviewer. Androgen preparations included DHEA, danazol, and testosterone patch. Trials studied the impact of androgen on menstrual pattern, ovarian reserve markers, fertility outcomes, mood, self-esteem, and bone mineral density (BMD).
Conclusion: There was no significant effect of androgen supplementation on mood, self-esteem, or bone mineral density. Although one study demonstrated an increase in ovarian volume and antral follicular count, there was no significant change in ovarian reserve, ovarian function, fertility outcome or menstrual pattern.
Clinical Trial Registration: Trial was registered with open science framework (OSF), DOI: 10.17605/OSF. IO/2MYUV
Keywords: Androgen, Danazol, Dehydroepianderosterone acetate, Premature ovarian failure, Premature ovarian insufficiency, Testosterone
Introduction
As women age, ovarian failure becomes a natural part of the process, typically occurring around the age of 51. However, 1–3.7% of women face premature ovarian insufficiency (POI) [1], characterized by a combination of oligomenorrhea/amenorrhea of more than four months duration associated with elevated gonadotropins (FSH > 40 IU/L) on at least two occasions measured four to six weeks apart. It occurs due to early depletion of ovarian follicular reserve, affecting women before the age of 40 [2]. This reduction in oocytes leads to hormonal changes with implications on fertility, menopausal symptoms, and long-term health, including cardiovascular risk, osteoporosis, and sexual desire and possible cognitive changes [3].
The etiology of POI is often idiopathic, but various causes such as genetic, iatrogenic, infectious metabolic, and autoimmune factors have been identified [1]. While oocyte depletion leads to infertility issues, and estrogen deficiency to the validated symptoms of menopause, possible androgen deficiency in POI women can impact vulvovaginal tissues, pelvic floor, bladder, urethra, and sexual functions [4],[5],[6],[7].
According to a recent systemic review, females with premature ovarian insufficiency suffer from lowered concentrations of testosterone, dehydroepiandrosterone sulfate, as well as androstenedione [8]. Exploring androgens as a therapeutic avenue for POI is crucial due to the current standard estrogen/hormone replacement therapy (HRT) regimen falling short in addressing the complex hormonal imbalances associated with POI. While estrogen replacement is a cornerstone in managing POI, androgens play a pivotal role in ovarian function, fertility, and overall well-being. Androgens, such as testosterone, have been shown to influence ovarian response, potentially improving ovarian reserve, and enhancing outcomes related to fertility [9]. Moreover, the conventional approach primarily focusing on estrogen may overlook the nuanced interplay of hormones in POI. A more comprehensive exploration of androgens may refine treatment strategies, offering a comprehensive approach that not only targets estrogen deficiency but also considers the significant contributions of androgens, possibly optimizing therapeutic outcomes for individuals with POI [10].
The primary objective of this systematic review is to review the literature about androgen therapies and premature ovarian insufficiency. By delving into the specificities of androgen interventions, and their outcomes, we aim to discern the varied impacts on hormonal profiles, ovarian function, and associated health parameters. This investigation is important for enhancing our understanding of the impact of the use of androgens in this population, with the goal of providing more effective management of POI.
Methodology and Search Strategy
Following the preferred reporting items for systematic reviews and meta-analyses (PRISMA) guidelines, this systematic review was conducted and registered with OSF under DOI 10.17605/OSF.IO/2MYUV. A comprehensive search, led by an information specialist (E.P.), covered databases such as Ovid Medline, PubMed, Cochrane Database for Systematic Reviews, Cochrane Central Database of Controlled Trials, Web of Science, and Google Scholar. The search included keywords and terms related to androgens, hormone replacement therapy, and primary ovarian insufficiency. No restrictions were placed on the search, and it was updated on January 15, 2023, to include new references.
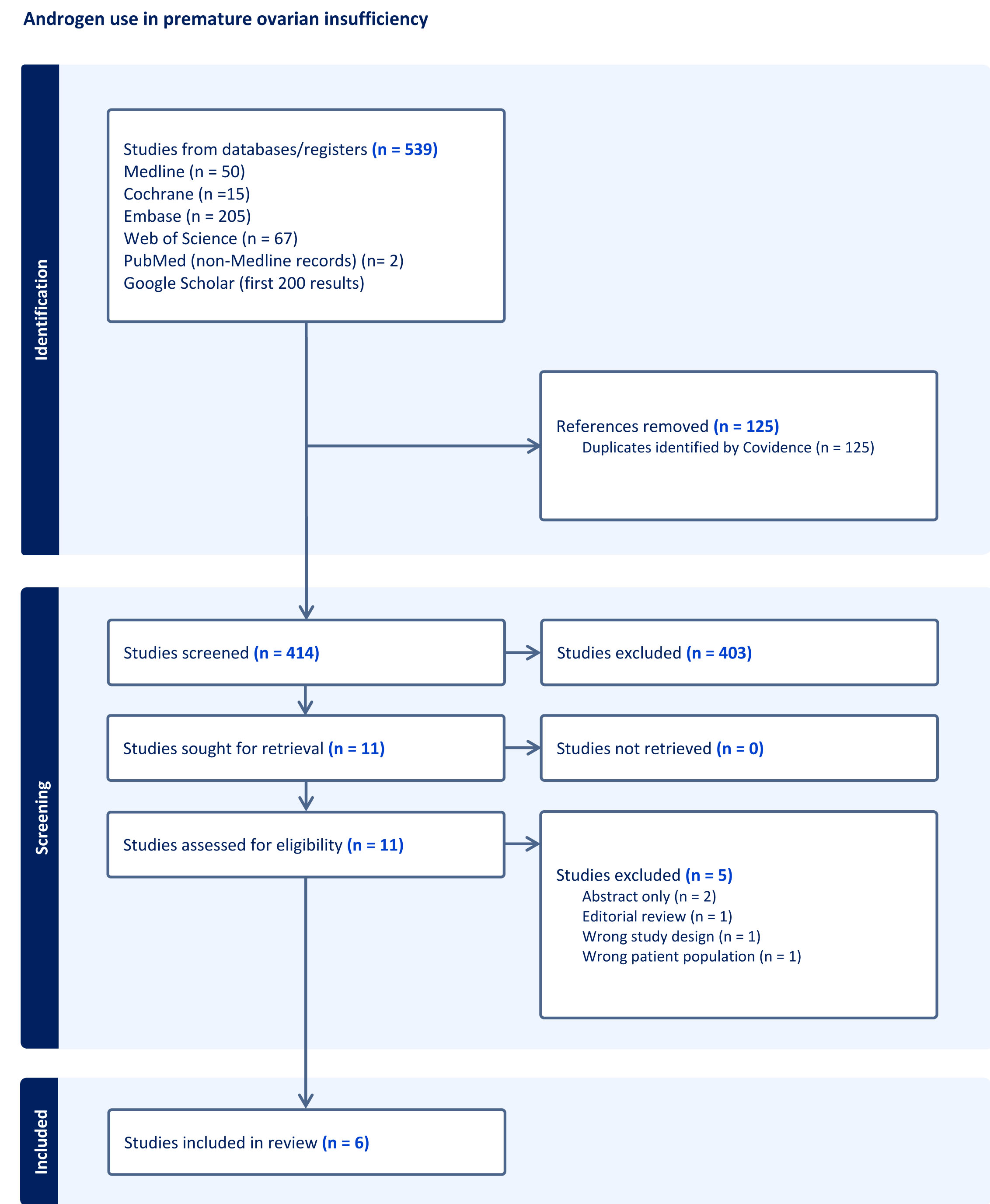
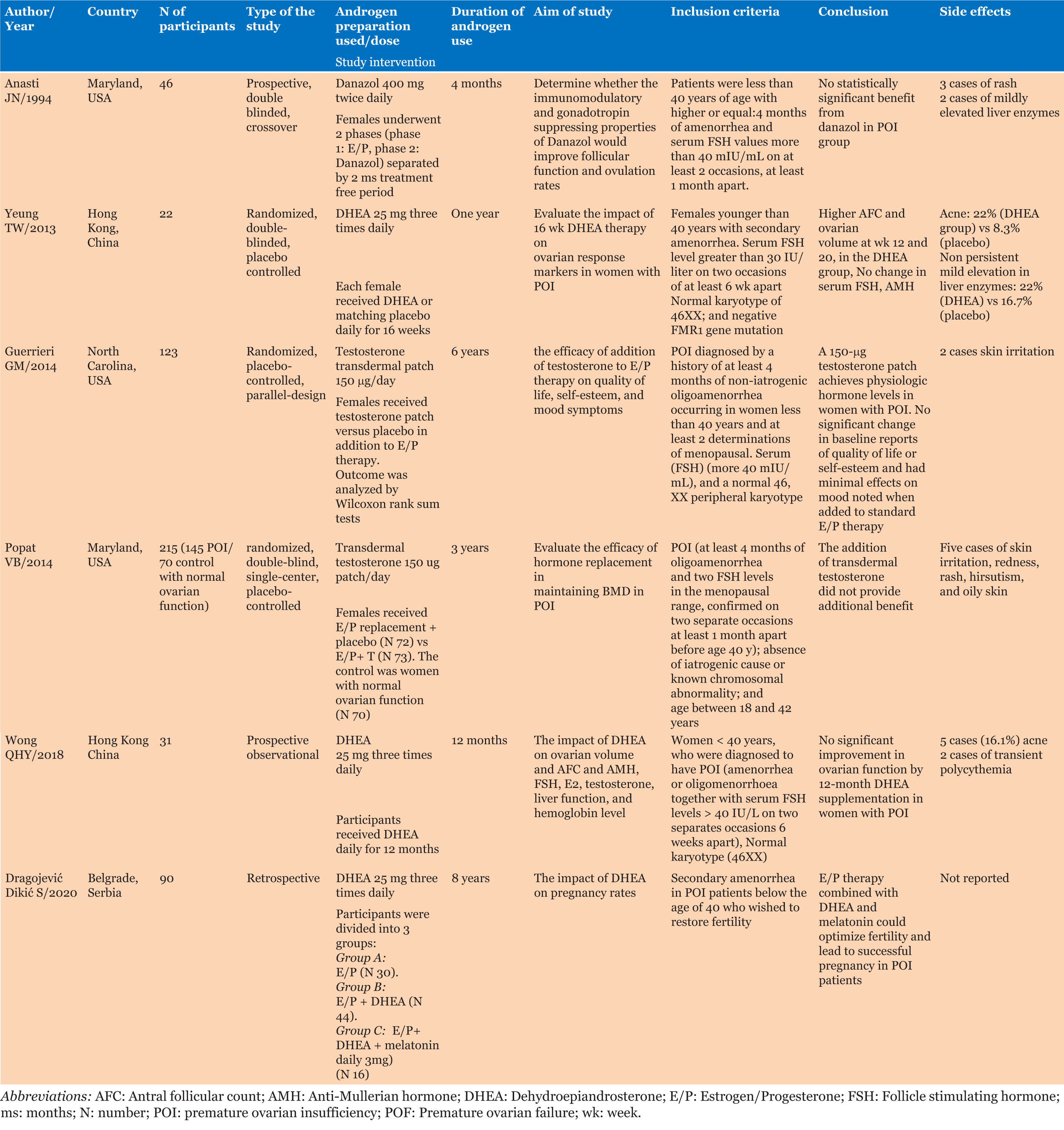
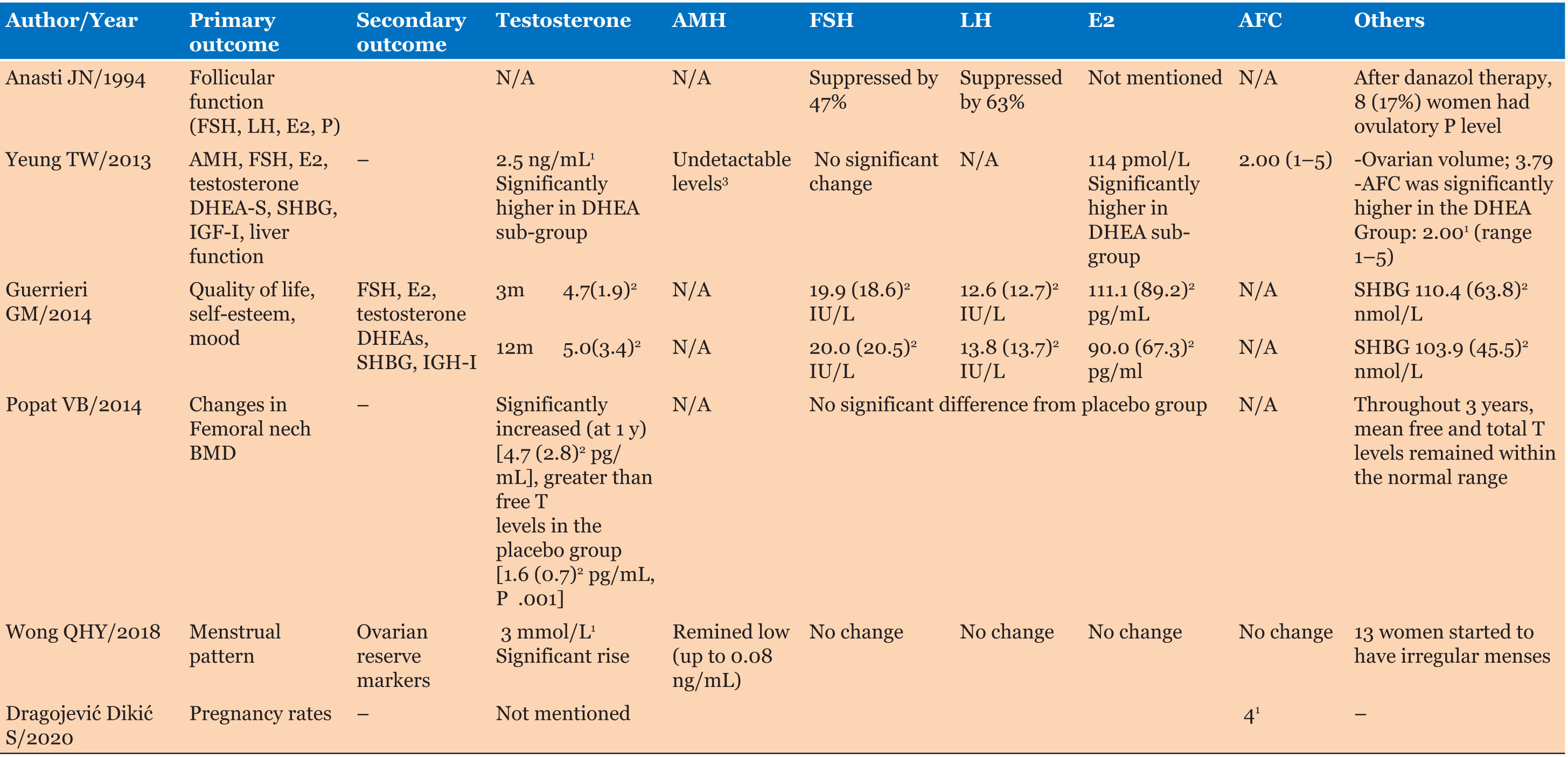
As shown in Figure 1, a comprehensive search yielded a total of 539 articles. Upon meticulous review of titles and abstracts, 11 studies underwent full-text assessment by D.M. and H.G. Subsequently, six articles met the inclusion criteria, comprising three randomized prospective double-blinded studies [11],[12],[13], one randomized placebo-controlled study [14], one prospective observational study [15], and one retrospective study [16]. A summary of the methodology of the studies included in this systematic review is shown in Table 1, while the impact of androgen therapy on hormonal profile and AFC was included in Table 2.
For the establishment of a POI diagnosis, the inclusion criteria mandated that females should be under 40 years old and experiencing amenorrhea for more than six months. One study reported that 22.6% of the recruited participants exhibited oligomenorrhea [15], while another study included four cases of primary amenorrhea [11]. The studies incorporated patients diagnosed with idiopathic POI while deliberately excluding cases of iatrogenic POI induced through surgical procedures or chemotherapy.
RESULTS
A total of 475 patients were recruited, with 356 females reaching the final analysis. Patients’ demographics were detailed, specifying previous hormone replacement therapy usage and criteria for POI diagnosis.
Androgen therapy used in the included studies had various indications including impact on; ovarian reserve [11],[12],[15],[16], self-esteem and mood changes [14], and bone mineral density [13]. In the realm of androgen preparations, the androgen preparations used were dehydroepiandrosterone (DHEA) dosed at 25 mg thrice daily, spanning durations from 16 weeks to 12 months [12],[15],[16], a testosterone transdermal patch, administered at as a daily 150 µg patch [13],[14], while a lone study employed danazol at 400 mg twice daily for four months [11] (Table 1).
Androgen was administrated either in isolation or in combination with other hormonal agents. Wong et al. and Yeung et al. compared DHEA to placebo [12],[15]. Popat et al. and Guerrieri et al. augmented hormone replacement therapy (estrogen and progesterone) with a testosterone patch [13],[14]. Anasti et al. sequenced hormone therapy, first using estrogen and progesterone replacement, followed by danazol [11]. Finally, Dragojevic Dikic et al. combined DHEA with estrogen and progesterone replacement therapy [16] (Table 1).
In view of the impact of androgen therapy on ovarian function (Table 2), three studies utilized androgen, in the form of DHEA, 25 mg thrice daily or danazol (400 mg twice daily), comparing it to placebo or estrogen/progesterone replacement therapy [11],[12],[16]. Although androgen did not significantly improve ovarian function, a small, randomized control trial conducted on 22 participants showed increased antral follicle count (AFC) at 12 weeks and ovarian volume after 20 weeks of DHEA administration with significant increase in median E2 levels (DHEA group) (114 pmol/L) compared to the placebo group (73 pmol/L), while they reported no significant change in FSH, LH, and AMH levels [12]. It was also noted that HRT (estradiol and progesterone) more significantly suppressed FSH and LH compared to danazol [11].
Two studies delved into the impact of androgen therapy, specifically DHEA (25 mg three times daily), on menstrual patterns [12],[15]. In one study, seven patients with oligomenorrhea and 24 with secondary amenorrhea were assessed. Among the oligomenorrheic patients, five maintained their menstrual patterns, while two transitioned to amenorrhea. In the group with secondary amenorrhea, 11 remained amenorrheic, while 13 experienced irregular menstruation, averaging 3.2 periods over 13 months [15]. Yeung et al. reported no significant disparity between the placebo and DHEA groups concerning the resurgence of irregular menstruation (25% in the placebo group versus 11% in the DHEA group) [12].
One of the retrospective studies examining the dynamics of fertility in POI, investigated the combined administration of dehydroepiandrosterone (DHEA) at a dose of 25 mg three times daily, and melatonin alongside estrogen-progesterone therapy. The addition of melatonin showed an increase in pregnancy rates when compared with other regimens, but the difference did not reach a statistically significant value [16].
In view of the repercussions of androgen deficiency on mood disturbances in POI, testosterone therapy was added to the conventional estrogen and progesterone, hormone replacement therapy (HRT). Notably, when compared to a placebo within the same HRT protocol, the addition of testosterone therapy did not yield any discernible alterations in baseline quality of life or mood reports [14]. Outcome measures for this trial included self- and rater-administered the Quality-of-Life Enjoyment and Satisfaction Questionnaire (Q-Les-Q) and the Rosenberg Self-Esteem Scale in which higher scores reflect better quality of life and self-esteem, respectively. They utilized the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID) to establish the presence or absence of Axis I psychiatric illness, as well as the self-administered Center for Epidemiologic Studies Depression Scale (CES-D) and the rater-administered Hamilton Rating Scale for Depression (HAM-D) to assess the severity of depressive symptom.
Popat et al. conducted a comprehensive study to assess the effect of androgen therapy on bone mineral density (BMD) three-year randomized trial which delved into the efficacy of a daily 150 micrograms testosterone patch as an augmentation to hormone replacement therapy (transdermal estradiol (100 mcg/d) and oral medroxyprogesterone acetate (10 mg/d) for 12 days per month). The findings revealed that while replacement therapy alone restored mean femoral neck BMD to normal levels in women with spontaneous POI, the addition of transdermal testosterone did not confer any additional advantage concerning BMD [13].
Surprisingly, in spite the fact that sexual problems are prevalent complaints expressed by females with idiopathic POI, there was no clinical trial delving into the impact of introducing androgen supplementation in those cases.
Among the six studies analyzed, four documented side effects associated with androgen therapy. Specifically, two studies employed DHEA, while the remaining two utilized a testosterone patch. The observed side effects encompassed a spectrum from skin irritation to elevated liver enzymes and transient polycythemia, as detailed in Table 1 [12],[13],[14],[15].
Discussion
Premature ovarian insufficiency (POI) is an increasingly prevalent disorder with profound implications for the mental and physical health of affected females. Women with POI face potential complications such as weight gain and dyslipidemia, which may escalate to type 2 diabetes and early cardiovascular disease [17],[18]. Additionally, long-term health concerns include accelerated bone loss leading to osteoporosis and fractures as well as cardiovascular diseases, which contribute directly to early mortality [17],[18]. While the estrogen deficiency in POI is well-established, the impact of androgen reduction remains underexplored [19].
During the premenopausal period, testosterone is considered the primary circulating androgen, with 50% contributed by the ovary and the rest resulting from peripheral conversion of pro-hormones from the adrenals and ovary [8]. While hormone replacement therapy traditionally features estrogen and progesterone, androgen replacement remains less prioritized in managing POI, leaving the optimal testosterone dose for physiological replacement unknown [8]. Consequently, the role of androgens in POI hormone replacement therapy, a critical aspect, has yet to be fully elucidated [8]. This review aimed to synthesize the best available evidence to guide clinicians in optimizing androgen use in POI cases.
Dehydroepiandrosterone was hoped to be a potential ally in reproductive health, with its proposed roles in minimizing follicular atresia and augmenting gonadotrophin effects on folliculogenesis. This is achieved through increased follicular insulin-like growth factor-1 (IGF-1) and heightened FSH receptor expression in granulosa cells [20],[21],[22]. Additionally, it plays a pivotal role as a prohormone in ovarian follicular steroidogenesis [22]. This explains the focus of previous research on androgen influence on restoring ovarian function, improving ovarian reserve (AMH, AFC, FSH, and estradiol), and exploring subsequent effects on pregnancy outcomes [12],[15],[16].
In 2021 a cross-sectional study was conducted to evaluate the prevalence, and severity of menopausal symptoms in 293 women with premature ovarian insufficiency (POI). The most prevalent symptoms were mood swings (73.4%), insomnia (58.7%), sexual problems (58.7%), and fatigue (57.3%) [23]. According to survey-based study, women with POI suffer from diminished general and sexual well-being and showed less satisfaction with their sexual lives compared to control women [24].
While numerous trials have investigated the effects of adding androgens to hormone replacement therapy, particularly in postmenopausal and surgically menopausal females, such research on sexual function in premature ovarian insufficiency (POI), is still lacking. In 2019, Davis et al. published a Global Consensus Position Statement, highlighting testosterone therapy as evidence-based solely for treating Hypoactive Sexual Desire Disorder/Dysfunction (HSDD) in postmenopausal females. However, this consensus did not address women with POI, despite evidence indicating lower circulating levels of testosterone in these individuals [7]. Given testosterone’s efficacy in postmenopausal females with HSDD, a logical approach would be to trial testosterone therapy in women with POI experiencing HSDD despite adequate estrogen replacement therapy (ERT) [8]. Notably, randomized trials evaluating androgen therapies in POI women with HSDD are lacking, with DHEA, danazol, and testosterone patches being the only studied forms [11],[12],[13],[14],[15],[16].
Clinicians encounter several challenges when prescribing androgens for POI women. The scarcity of trials addressing androgen use in this population, coupled with controversial results regarding baseline testosterone levels in POI cases compared to non-menopausal controls, contributes to the lack of definitive evidence [8],[25],[26],[27],[28],[29]. Moreover, despite positive study outcomes for postmenopausal women testosterone therapy lacks FDA approval for all women with HSDD, necessitating informed consent for its off-label use [30]. Patients must also consider the economic costs of using an off-label medication. Notably, while there are over 30 FDA-approved testosterone preparations for male hypogonadism, there are no approved formulations for females [30]. Determining the most beneficial dosage for premenopausal women, estimated at 1/10th the recommended starting dose in men, further complicates prescription practices. For instance, the recommended dose of testosterone gel 1% for male hypogonadism would be used as one tube daily for men but differs significantly from the recommended regimen for females which recommends using a dose of one tube or packet every 10 days to be equivalent to about 5 mg/day (0.5 mL) [30]. Finally, clinicians must consider practical aspects, such as using resealable tubes to prevent evaporation, as compounded testosterone for HSDD is discouraged due to the lack of quality surveillance [31],[32],[33].
Various androgen formulations, including buccal, nasal, subdermal, transdermal, and intramuscular options, are available. Transdermal formulations (patch, gel, cream, spray) emerge as the preferred choice for women due to their ease of administration and the ability to titrate to physiological levels [30].
The literature review reveals an overlap between premature ovarian insufficiency and poor ovarian response (POR), emphasizing the need for precise terminology in future studies [34]. In a study by Malik et al. in 2015, patients included under the term “premature ovarian aging” exhibited characteristics of poor ovarian response rather than premature ovarian insufficiency, highlighting the importance of clear definitions in research [35].
This review has some limitations including: the limited number of studies available on the selected topic. The various androgen preparations used by each study made it difficult to compare between them or perform a meta-analysis on the results, and finally, simply the paucity of studies that demonstrates the impact of testosterone on HSDD in women with idiopathic POI. On the other hand, the strength of the study is related to the fact that it uncovers how scarce the existing data is regarding the topic of concern.
Conclusion
Based on the limited evidence presented by the included studies, there is no significant effect of androgen supplementation on mood, self-esteem, or bone mineral density. Regarding ovarian function, there has been an increase in ovarian volume and antral follicular count, with no significant change in ovarian reserve, ovarian function, fertility outcome or menstrual pattern. There is no current data to support the use of testosterone for HSDD in women with POI. Long-term trials are strongly recommended.
REFERENCES
1.
Meskhi A, Seif MW. Premature ovarian failure. Curr Opin Obstet Gynecol 2006;18(4):418–26. [CrossRef]
[Pubmed]

2.
Mamas L, Mamas E. Premature ovarian failure and dehydroepiandrosterone. Fertil Steril 2009;91(2):644–6. [CrossRef]
[Pubmed]

3.
van Kasteren YM, Schoemaker J. Premature ovarian failure: A systematic review on therapeutic interventions to restore ovarian function and achieve pregnancy. Hum Reprod Update 1999;5(5):483–92. [CrossRef]
[Pubmed]

4.
McCoy NL, Davidson JM. A longitudinal study of the effects of menopause on sexuality. Maturitas 1985;7(3):203–10. [CrossRef]
[Pubmed]

5.
6.
Alder EM, Cook A, Davidson D, West C, Bancroft J. Hormones, mood and sexuality in lactating women. Br J Psychiatry 1986;148:74–9. [CrossRef]
[Pubmed]

7.
Davis SR, Davison SL, Donath S, Bell RJ. Circulating androgen levels and self-reported sexual function in women. JAMA 2005;294(1):91–6. [CrossRef]
[Pubmed]

8.
Soman M, Huang LC, Cai WH, Xu JB, Chen JY, He RK, et al. Serum androgen profiles in women with premature ovarian insufficiency: A systematic review and meta-analysis. Menopause 2019;26(1):78–93. [CrossRef]
[Pubmed]

9.
Janse F, Tanahatoe SJ, Eijkemans MJC, Fauser BCJM. Testosterone concentrations, using different assays, in different types of ovarian insufficiency: A systematic review and meta-analysis. Hum Reprod Update 2012;18(4):405–19. [CrossRef]
[Pubmed]

10.
Panay N, Anderson RA, Nappi RE, Vincent AJ, Vujovic S, Webber L, Wolfman W. Premature ovarian insufficiency: An International Menopause Society White Paper. Climacteric 2020;23(5):426–46. [CrossRef]
[Pubmed]

11.
Anasti JN, Kimzey LM, Defensor RA, White B, Nelson LM. A controlled study of danazol for the treatment of karyotypically normal spontaneous premature ovarian failure. Fertil Steril 1994;62(4):726–30. [CrossRef]
[Pubmed]

12.
Yeung TWY, Li RHW, Lee VCY, Ho PC, Ng EHY. A randomized double-blinded placebo-controlled trial on the effect of dehydroepiandrosterone for 16 weeks on ovarian response markers in women with primary ovarian insufficiency. J Clin Endocrinol Metab 2013;98(1):380–8. [CrossRef]
[Pubmed]

13.
Popat VB, Calis KA, Kalantaridou SN, Vanderhoof VH, Koziol D, Troendle JF, et al. Bone mineral density in young women with primary ovarian insufficiency: Results of a three-year randomized controlled trial of physiological transdermal estradiol and testosterone replacement. J Clin Endocrinol Metab 2014;99(9):3418–26. [CrossRef]
[Pubmed]

14.
Guerrieri GM, Martinez PE, Klug SP, Haq NA, Vanderhoof VH, Koziol DE, et al. Effects of physiologic testosterone therapy on quality of life, self-esteem, and mood in women with primary ovarian insufficiency. Menopause 2014;21(9):952–61. [CrossRef]
[Pubmed]

15.
Wong QHY, Yeung TWY, Yung SSF, Ko JKY, Li HWR, Ng EHY. The effect of 12-month dehydroepiandrosterone supplementation on the menstrual pattern, ovarian reserve markers, and safety profile in women with premature ovarian insufficiency. J Assist Reprod Genet 2018;35(5):857–62. [CrossRef]
[Pubmed]

16.
Dragojević Dikić S, Vasiljević M, Jovanović A, Dikić S, Jurišić A, Srbinović L, et al. Premature ovarian insufficiency – novel hormonal approaches in optimizing fertility. Gynecol Endocrinol 2020;36(2):162–5. [CrossRef]
[Pubmed]

17.
Casson PR, Lindsay MS, Pisarska MD, Carson SA, Buster JE. Dehydroepiandrosterone supplementation augments ovarian stimulation in poor responders: A case series. Hum Reprod 2000;15(10):2129–32. [CrossRef]
[Pubmed]

18.
Shuster LT, Rhodes DJ, Gostout BS, Grossardt BR, Rocca WA. Premature menopause or early menopause: Long-term health consequences. Maturitas 2010;65(2):161–6. [CrossRef]
[Pubmed]

19.
Muka T, Oliver-Williams C, Kunutsor S, Laven JSE, Fauser BCJM, Chowdhury R, et al. Association of age at onset of menopause and time since onset of menopause with cardiovascular outcomes, intermediate vascular traits, and all-cause mortality: A systematic review and meta-analysis. JAMA Cardiol 2016;1(7):767–76. [CrossRef]
[Pubmed]

20.
Davis SR. Androgens in premenopausal women and women with premature ovarian insufficiency. Climacteric 2021;24(5):459–65. [CrossRef]
[Pubmed]

21.
Barad DH, Gleicher N. Increased oocyte production after treatment with dehydroepiandrosterone. Fertil Steril 2005;84(3):756. [CrossRef]
[Pubmed]

22.
Vendola KA, Zhou J, Adesanya OO, Weil SJ, Bondy CA. Androgens stimulate early stages of follicular growth in the primate ovary. J Clin Invest. 1998;101(12):2622–9. [CrossRef]
[Pubmed]

23.
Huang Y, Qi T, Ma L, Li D, Li C, Lan Y, et al. Menopausal symptoms in women with premature ovarian insufficiency: Prevalence, severity, and associated factors. Menopause 2021;28(5):529–37. [CrossRef]
[Pubmed]

24.
van der Stege JG, Groen H, van Zadelhoff SJN, Lambalk CB, Braat DDM, van Kasteren YM, et al. Decreased androgen concentrations and diminished general and sexual well-being in women with premature ovarian failure. Menopause 2008;15(1):23–31. [CrossRef]
[Pubmed]

25.
Kalantaridou SN, Calis KA, Vanderhoof VH, Bakalov VK, Corrigan EC, Troendle JF, et al. Testosterone deficiency in young women with 46,XX spontaneous premature ovarian failure. Fertil Steril 2006;86(5):1475–82. [CrossRef]
[Pubmed]

26.
Doldi N, Belvisi L, Bassan M, Fusi FM, Ferrari A. Premature ovarian failure: Steroid synthesis and autoimmunity. Gynecol Endocrinol 1998;12(1):23–8. [CrossRef]
[Pubmed]

27.
Elias AN, Pandian MR, Rojas FJ. Serum levels of androstenedione, testosterone and dehydroepiandrosterone sulfate in patients with premature ovarian failure to age-matched menstruating controls. Gynecol Obstet Invest 1997;43(1):47–8. [CrossRef]
[Pubmed]

28.
Hartmann BW, Kirchengast S, Albrecht A, Laml T, Söregi G, Huber JC. Androgen serum levels in women with premature ovarian failure compared to fertile and menopausal controls. Gynecol Obstet Invest 1997;44(2):127–31. [CrossRef]
[Pubmed]

29.
Kaufman FR, Donnell GN, Lobo RA. Ovarian androgen secretion in patients with galactosemia and premature ovarian failure. Fertil Steril 1987;47(6):1033–4. [CrossRef]
[Pubmed]

30.
Parish SJ, Simon JA, Davis SR, Giraldi A, Goldstein I, Goldstein SW, et al. International Society for the Study of Women’s Sexual Health Clinical Practice Guideline for the Use of Systemic Testosterone for Hypoactive Sexual Desire Disorder in Women. J Sex Med 2021;18(5):849–67. [CrossRef]
[Pubmed]

31.
Davis SR, Baber R, Panay N, Bitzer J, Perez SC, Islam RM, et al. Global consensus position statement on the use of testosterone therapy for women. J Clin Endocrinol Metab 2019;104(10):4660–6. [CrossRef]
[Pubmed]

32.
Benetti-Pinto CL, Bedone AJ, Magna LA. Evaluation of serum androgen levels in women with premature ovarian failure. Fertil Steril 2005;83(2):508–10. [CrossRef]
[Pubmed]

33.
National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Sciences Policy; Committee on the Clinical Utility of Treating Patients with Compounded Bioidentical Hormone Replacement Therapy. The clinical utility of compounded bioidentical hormone therapy: A review of safety, effectiveness, and use. Jackson LM, Parker RM, Mattison DR, editors. Washington (DC): National Academies Press (US); 2020. [CrossRef]
[Pubmed]

34.
Grober ED, Garbens A, Božović A, Kulasingam V, Fanipour M, Diamandis EP. Accuracy of testosterone concentrations in compounded testosterone products. J Sex Med 2015;12(6):1381–8. [CrossRef]
[Pubmed]

35.
Malik N, Kriplani A, Agarwal N, Bhatla N, Kachhawa G, Yadav RK. Dehydroepiandrosterone as an adjunct to gonadotropins in infertile Indian women with premature ovarian aging: A pilot study. J Hum Reprod Sci 2015;8(3):135–41. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Dina Mohamed - Conception of the work, Design of the work, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Nada Shaltout - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Hala Gomaa - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Eleni Philippopoulos - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Javier Mejia-Gomez - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Wendy Wolfman - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Dina Mohamed et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.