|
Case Report
Anti-GD1b and anti-GM1 positive, acute inflammatory demyelinating polyneuropathy associated with advanced ovarian carcinoma: A case report
1 Emergency Department, CHUV – Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland
2 Center of Gynecological Tumors, Oncology Department, CHUV – Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland
3 Director of Neuro-Oncology Unit, CHUV – Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland
4 Responsible of Center of Gynecological Tumors, Oncology Department, CHUV – Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland
Address correspondence to:
Aikaterini Liapi
Center of Gynecological Tumors, Oncology Department, CHUV – Centre Hospitalier Universitaire Vaudois, Lausanne, Rue du Bugnon 46, 1011, Lausanne,
Switzerland
Message to Corresponding Author
Article ID: 101195Z01JF2021
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Frutiger J, Liapi A, Hastir D, Hottinger A, Sarivalasis A. Anti-GD1b and anti-GM1 positive, acute inflammatory demyelinating polyneuropathy associated with advanced ovarian carcinoma: A case report. Int J Case Rep Images 2021;12:101195Z01JF2021.ABSTRACT
Introduction: Paraneoplastic neurological syndromes (PNSs) are mostly associated with non-epithelial ovarian cancers. We report the case of an advanced high-grade serous ovarian adenocarcinoma, diagnosed during the etiological investigation of a subacute motor-sensory axonal neuropathy variant (AMSAN) of Guillain-Barré syndrome (GBS). According to our literature review, only a single case-control study described a positive association between GBS and cancer.
Case Report: A 73-year-old woman was admitted to the emergency department for a progressive symmetrical distal tetraparesis, associated with hypoesthesia and abdominal pain. There was no sign of viral or bacterial infection. Over a period of 10 days, she presented a rapid neurological deterioration with nearly complete paralysis of all extremity, facial, and respiratory muscles. The laboratory testing ruled out active infection but the computed tomography (CT) scanner described diffuse peritoneal carcinomatosis, associated with bilateral ovarian tumors. Laparoscopic biopsies confirmed a high-grade serous ovarian cancer. Cerebrospinal fluid analysis confirmed the presence of elevated protein with a normal white blood cell count and anti-ganglioside autoantibodies anti-GD1b and anti-GM1. The patient received systemic chemotherapy together with plasmapheresis and intravenous immunoglobulins, and she experienced a rapid neurological recovery and long-term tumor remission.
Conclusion: Neurological paraneoplastic syndromes are rarely associated with epithelial ovarian cancer. Cerebellar ataxia is most frequently associated with this cancer type. Guillain-Barré syndrome can rarely be associated with high-grade serous ovarian cancer. It can delay the tumor diagnosis and treatment. In the case of our patient, early supportive and etiological treatment was of paramount importance for the complete recovery of our patient.
Keywords: Antiganglioside autoantibodies, Guillain-Barré, High-grade serous ovarian cancer, Paraneoplastic syndrome
Introduction
Paraneoplastic neurological syndromes (PNSs) derive from immune-mediated neuronal degeneration occurring in the absence of direct tumor invasion [1],[2]. Onco-neuronal antigens initiate the autoimmune response. These antigens either are released from the tumor, or are expressed as shared antigens on the surface of both nervous system and tumor cells. The resulting autoimmune response is mediated by activated T effector cells [3],[4]. Paraneoplastic neurological syndromes are diagnosed in 0.5–1% of all newly diagnosed cancer patients, including ovarian cancer patients [5],[6]. Although non-epithelial ovarian carcinomas are more frequently associated with this condition, PNSs can be the presenting symptom or a complication of high-grade serous ovarian carcinomas (HGSOC), irrespective of the tumor stage.
Paraneoplastic neurological syndromes occurrence is considered as a favorable factor for tumor prognosis and cancer treatment outcome [7]. This effect is believed to be driven by host immune response against tumor-host shared antigens.
Neurological symptoms are the main presenting symptoms in many cases of ovarian cancer associated with PNSs and they can often mask or delay the diagnosis of the underlying ovarian tumor [3].
We report the case of an anti-GD1b and anti-GM1 positive paraneoplastic Guillain-Barré syndrome associated with an underlying advanced high-grade serous ovarian adenocarcinoma.
Case Report
A 73-year-old woman was admitted to the emergency department for a progressive symmetrical distal tetraparesis, associated with hypoesthesia and abdominal pain. Symptoms were evolving over a period of two weeks. She presented no signs of viral nor bacterial infection.
The neurological examination revealed a proximal-predominant symmetrical paresis of all limbs, corresponding to Medical Research Council grade 3/5 for proximal muscles, 4/5 for distal muscles. No fasciculation was noted. On cranial nerve exam, the patient presented a slight facial peripheral paresis, House-Brackmann score grade II. There were postural abnormalities more present on the lower limbs than on the upper limbs. The upper limbs showed no signs of kinetic ataxia, while the lower limbs examination was impossible. There was reduced pallesthesia at 6/8 in the upper limbs and 0/8 at the malleolar level of both lower limbs. Other discriminative general senses were impaired only in the lower limb extremities.
Symmetric areflexia was also present with absent deep tendon reflexes. The neurological distribution was suggestive of an acute inflammatory demyelinating polyneuropathy (AIDP) or GBS (Table 1).
During the first days of her hospitalization, she presented a rapid neurological deterioration with progressive centrally spreading paresis and signs of respiratory failure with resting dyspnea and a restrictive pattern with reduced vital capacity in the spirometry.
The diagnosis of GBS was confirmed by the lumbar puncture. In the cerebrospinal fluid of the patient a normal cell count with no pathogens on Gram staining and an albuminocytologic dissociation was also noticed, with hyperproteinorachia at 776 mg/L but with absence of hypercellulorachia.
In both the cerebrospinal fluid (CSF) and serum of the patient anti-ganglioside antibodies (GD1b, GM1) were detected. The lumbar puncture revealed an elevated CSF protein (776 mg/L) with a normal CSF white blood cell count, also known as albuminocytologic dissociation. Antineuronal antibodies were negative.
The magnetic resonance imaging (MRI) of the brain and spine was unremarkable. Nerve conductions studies showed a sensitive and motor demyelination pattern, compatible with GBS.
The medical history, clinical examination, and laboratory values ruled out active infection from common GBS-associated pathogens.
The patient also complained of abdominal pain unrelated to stool or food intake. Over her hospitalization, she developed persistent constipation requiring laxative treatment and manual extraction of stools. The abdominal symptoms were initially attributed to the opioid treatment and the enteric dysautonomia, a well-described component of the GBS. The persistence of abdominal symptoms and the intensity of the acute abdominal pain episodes, without signs of ascites or bowel obstruction, prompted an abdominal, contrast enhanced, CT-scanner.
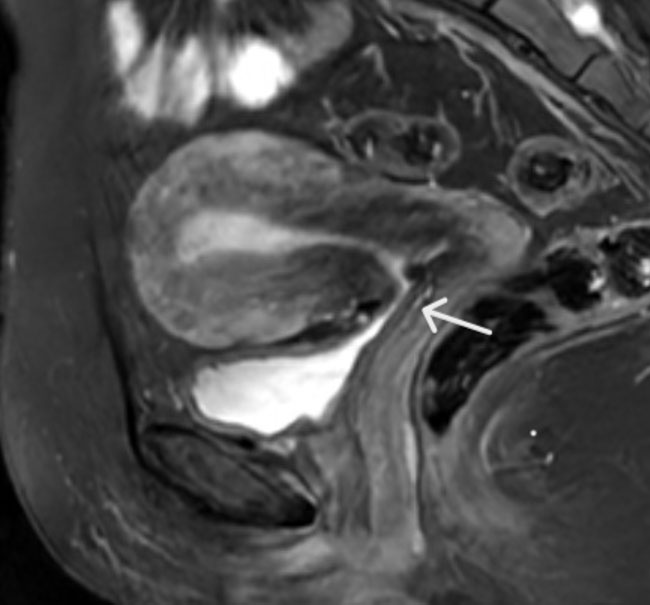
The CT-scanner described diffuse peritoneal carcinomatosis associated with bilateral ovarian tumors. For the radiological staging of the disease a whole body, positron emission tomography scan was performed. An intense fluorodeoxyglucose (FDG) uptake on both ovaries and diffuse peritoneal localization without extra peritoneal spread was documented (Figure 1 and Figure 2).
The CA 125, CA 19-9, and CEA values were 188 kU/L (UNL<35), 3 kU/L (<37), and 1.3 μg/L (<5), respectively.
The surgical staging with multiple biopsies was carried out by laparoscopy. Primary cytoreduction was not possible due to the poor performance status of the patient. The biopsies revealed a HGSOC, harboring the pathognomonic p53 mutation (Figure 3). The disease was staged AJCC/TNM cT3c cN0 cM0, FIGO IIIC. Her cancer was not associated with a germline BRCA 1 or BRCA 2 mutation.
The patient was treated for her GBS with plasmapheresis and five days of intravenous immunoglobulin 2 g/kg. Physiotherapy and occupational therapy were also prescribed.
The detection of the underlying advanced HGSOC prompted a weekly carboplatinum AUC2 and paclitaxel 60 mg/m2, intravenous (IV) chemotherapy, adapted to her performance status, which was initiated three weeks after the administration of immunoglobulins. Her neurological condition rapidly improved with a complete recovery in 10 days. The patient underwent a macroscopically complete interval debulking surgery after three, three-weekly, cycles of chemotherapy. Her treatment was completed with three more cycles of the same regimen, for a total of six cycles. Of note, no peripheral sensory, taxane-induced, neuropathy was observed on treatment (Figure 4).
Discussion
The most common PNSs associated with ovarian tumors are subacute cerebellar degeneration and subacute sensory neuropathy [2],[4],[8],[9],[10],[11],[12],[13],[14],[15],[16],[17],[18],[19]. Paraneoplastic GBS has been infrequently associated with ovarian cancers [20]. The initial diagnosis of GBS relies on clinical presentation, and it is supported by CSF and electro-neurophysiological abnormalities [9],[10],[21],[22]. In the case of this patient, we concluded to a paraneoplastic inflammatory demyelinating polyneuropathy syndrome, associated with the underlying HGSOC. The chronological association of the symptoms, the absence of a concurrent or passed infection, the presence of pathologic electromyoneurography (EMNG) and positive anti-GM1 and anti-GD1b antibodies in the CSF and serum of the patient and the rapid complete recovery after the initiation of chemotherapy, confirmed our hypothesis [10],[23].
Early diagnosis and a high degree of clinical awareness are of paramount importance in PNS management. Indeed, PNS-associated neurological symptoms almost invariably predate symptoms associated with the primary tumor [1],[2]. Delays in the treatment can negatively affect the prognosis and the potential of neurological recovery. Furthermore, early detection of PNS can guide subsequent diagnostic tests especially according to the detected autoantibody.
Epithelial carcinomas are often associated with anti-Yo, anti-Ri, and anti-amphiphysin while non-epithelial tumors with anti- N-methyl-D-aspartate receptor (NMDAR) [2],[4],[24]. The most common ovarian tumors associated PNSs are subacute cerebellar degeneration and subacute sensory neuropathy.
Paraneoplastic GBS is rarely associated with epithelial ovarian cancers. According to our research, only a single case-control study described a positive association between GBS and cancer [25]. Guillain-Barré syndrome is mostly associated with lymphomas [26],[27]. Solid tumors associated with GBS include small cell lung cancer, breast cancer, renal cancer, hepatocellular carcinoma, tongue carcinoma, esophageal carcinoma, and the ovarian dysgerminoma. Thus, many tend to question the GBS as a plausible PNS in epithelial ovarian cancer. The underlying immunological mechanism remains elusive. In the case of our patient anti-GM1 and the anti-GD1b antibodies were positive and, along with the electroneuromyography (ENMG) findings, helped confirm the diagnosis of AMSAN of Guillain-Barré syndrome.
Anti-ganglioside antibodies are positive in 36% of GBS patients. They are detected early on in the evolution of disease.
Our case indicates that ovarian carcinoma can be associated with both central and peripheral PNS and illustrates the treatment requirement for such a complicated condition. Beside the standard therapy of GBS with plasmapheresis or administration of intravenous immune globulin (IVIG), an emergency oncological systemic treatment is also required. The oncologic treatment should be adapted to the clinical condition of the patient but should be administered at effective dosage. This treatment approach provides higher chances of neurological recovery and treatment of the underlying ovarian carcinoma. Nevertheless, some studies suggest patients with cancer PNSs to have worse neurologic outcomes [28],[29]. The patient presented a transient worsening of tetraparesis, probably due to taxane chemotherapy and corticosteroids administration, with subsequent, slowly progressive improvement and complete neurological recovery, following immunoglobulin treatment and intensive physiotherapy and ergotherapy. Of note, following her multimodal oncological treatment she is disease free, two years from her diagnosis. The observed long-term oncological remission could confirm the observation that neoplasms presenting with PNS may be associated with innate anti-tumor immune response [30],[31]. Early onset treatment of the underlying tumor together with specific neurological and supportive treatment is paramount to obtain the best possible outcome.
Conclusion
Neurological paraneoplastic syndromes are rarely associated with epithelial ovarian cancer. Cerebellar ataxia is most frequently associated with this cancer type. Guillain-Barré syndrome (GBS) can rarely be associated with high-grade serous ovarian cancer. It can delay the tumor diagnosis and treatment. In the case of our patient, early supportive and etiological treatment was of paramount importance for the complete recovery of our patient.
REFERENCES
1.
Rees JH. Paraneoplastic syndromes: When to suspect, how to confirm, and how to manage. J Neurol Neurosurg Psychiatry 2004;75 Suppl 2(Suppl 2):ii43–50. [CrossRef]
[Pubmed]

2.
Graus F, Delattre JY, Antoine JC, et al. Recommended diagnostic criteria for paraneoplastic neurological syndromes. J Neurol Neurosurg Psychiatry 2004;75(8):1135–40. [CrossRef]
[Pubmed]

3.
Maverakis E, Goodarzi H, Wehrli LN, Ono Y, Garcia MS. The etiology of paraneoplastic autoimmunity. Clin Rev Allergy Immunol 2012;42(2):135–44. [CrossRef]
[Pubmed]

4.
Titulaer MJ, Soffietti R, Dalmau J, et al. Screening for tumours in paraneoplastic syndromes: Report of an EFNS task force. Eur J Neurol 2011;18(1):19–e3. [CrossRef]
[Pubmed]

5.
Zaborowski MP, Spaczynski M, Nowak-Markwitz E, Michalak S. Paraneoplastic neurological syndromes associated with ovarian tumors. J Cancer Res Clin Oncol 2015;141(1):99–108. [CrossRef]
[Pubmed]

6.
Giometto B, Grisold W, Vitaliani R, et al. Paraneoplastic neurologic syndrome in the PNS Euronetwork database: A European study from 20 centers. Arch Neurol 2010;67(3):330–5. [CrossRef]
[Pubmed]

7.
Darnell RB, Posner JB. Paraneoplastic syndromes involving the nervous system. N Engl J Med 2003;349(16):1543–54. [CrossRef]
[Pubmed]

8.
Yuki N, Hartung HP. Guillain-Barré syndrome. N Engl J Med 2012;366(24):2294–304. [CrossRef]
[Pubmed]

9.
Saeed ML, Kaleem Baloch B, Mahmud SN, et al. Role of anti-ganglioside antibodies in the diagnosis of Guillain-Barré syndrome as an alternate investigation. Cureus 2019;11(5):e4625.
[Pubmed]

10.
Sekiguchi Y, Uncini A, Yuki N, et al. Antiganglioside antibodies are associated with axonal Guillain-Barré syndrome: A Japanese-Italian collaborative study. J Neurol Neurosurg Psychiatry 2012;83(1):23–8. [CrossRef]
[Pubmed]

11.
Hadden RD, Karch H, Hartung HP, et al. Preceding infections, immune factors, and outcome in Guillain-Barré syndrome. Neurology 2001;56(6):758–65. [CrossRef]
[Pubmed]

12.
Jacobs BC, Rothbarth PH, van der Meché FG, et al. The spectrum of antecedent infections in Guillain-Barré syndrome: A case-control study. Neurology 1998;51(4):1110–5. [CrossRef]
[Pubmed]

13.
Burns TM, Lawn ND, Low PA, Camilleri M, Wijdicks EF. Adynamic ileus in severe Guillain-Barré syndrome. Muscle Nerve 2001;24(7):963–5. [CrossRef]
[Pubmed]

14.
Appropriate number of plasma exchanges in Guillain-Barré syndrome. The French Cooperative Group on Plasma Exchange in Guillain-Barré Syndrome. Ann Neurol 1997;41(3):298–306. [CrossRef]
[Pubmed]

15.
Tanyi JL, Marsh EB, Dalmau J, Chu CS. Reversible paraneoplastic encephalitis in three patients with ovarian neoplasms. Acta Obstet Gynecol Scand 2012;91(5):630–4. [CrossRef]
[Pubmed]

16.
Jacobs BC, O'Hanlon GM, Bullens RWM, Veitch J, Plomp JJ, Willison HJ. Immunoglobulins inhibit pathophysiological effects of anti-GQ1b-positive sera at motor nerve terminals through inhibition of antibody binding. Brain 2003;126(Pt 10):2220–34. [CrossRef]
[Pubmed]

17.
Graham RC, Hughes RAC, White CM. A prospective study of physiotherapist prescribed community based exercise in inflammatory peripheral neuropathy. J Neurol 2007;254(2):228–35. [CrossRef]
[Pubmed]

18.
Peterson K, Rosenblum MK, Kotanides H, Posner JB. Paraneoplastic cerebellar degeneration. I. A clinical analysis of 55 anti-Yo antibody-positive patients. Neurology 1992;42(10):1931–7. [CrossRef]
[Pubmed]

19.
Dalmau J, Rosenfeld MR. Paraneoplastic syndromes of the CNS. Lancet Neurol 2008;7(4):327–40. [CrossRef]
[Pubmed]

20.
Vigliani MC, Magistrello M, Polo P, et al. Risk of cancer in patients with Guillain-Barré syndrome (GBS). A population-based study. J Neurol 2004;251(3):321–6. [CrossRef]
[Pubmed]

21.
Asbury AK, Cornblath DR. Assessment of current diagnostic criteria for Guillain-Barré syndrome. Ann Neurol 1990;27 Suppl:S21–4. [CrossRef]
[Pubmed]

22.
van den Berg B, Walgaard C, Drenthen J, Fokke C, Jacobs BC, van Doorn PA. Guillain-Barré syndrome: Pathogenesis, diagnosis, treatment and prognosis. Nat Rev Neurol 2014;10(8):469–82. [CrossRef]
[Pubmed]

23.
Nishimoto Y, Odaka M, Hirata K, Yuki N. Usefulness of anti-GQ1b IgG antibody testing in Fisher syndrome compared with cerebrospinal fluid examination. J Neuroimmunol 2004;148(1–2):200–5. [CrossRef]
[Pubmed]

24.
Zuliani L, Graus F, Giometto B, Bien C, Vincent A. Central nervous system neuronal surface antibody associated syndromes: Review and guidelines for recognition. J Neurol Neurosurg Psychiatry 2012;83(6):638–45. [CrossRef]
[Pubmed]

25.
Kim MH, Hwang MS, Park YK, et al. Paraneoplastic Guillain-Barré syndrome in small cell lung cancer. Case Rep Oncol 2015;8(2):295–300. [CrossRef]
[Pubmed]

26.
Bishay RH, Paton J, Abraham V. Variant Guillain-Barré syndrome in a patient with non-Hodgkin's lymphoma. Case Rep Hematol 2015;2015:979237. [CrossRef]
[Pubmed]

27.
Anderson D, Beecher G, Steve TA, Jen H, Camicioli R, Zochodne DW. Neurological nuance: Hodgkin lymphoma presenting with Guillain-Barré syndrome. Muscle Nerve 2017;55(4):601–4. [CrossRef]
[Pubmed]

28.
Cao Y, Abbas J, Wu X, Dooley J, van Amburg AL. Anti-Yo positive paraneoplastic cerebellar degeneration associated with ovarian carcinoma: Case report and review of the literature. Gynecol Oncol 1999;75(1):178–83. [CrossRef]
[Pubmed]

29.
Rojas I, Graus F, Keime-Guibert F, et al. Long-term clinical outcome of paraneoplastic cerebellar degeneration and anti-Yo antibodies. Neurology 2000;55(5):713–5. [CrossRef]
[Pubmed]

30.
Liapi A, Sarivalasis A. Paraneoplastic cerebellar ataxia can affect prognosis in high-grade serous ovarian cancer: A case report. Case Rep Oncol 2020;13(2):1006–12. [CrossRef]
[Pubmed]

31.
Vogrig A, Muñiz-Castrillo S, Desestret V, Joubert B, Honnorat J. Pathophysiology of paraneoplastic and autoimmune encephalitis: Genes, infections, and checkpoint inhibitors. Ther Adv Neurol Disord 2020;13:1756286420932797. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Julie Frutiger - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Aikaterini Liapi - Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Delfyne Hastir - Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Andreas Hottinger - Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Apostolos Sarivalasis - Conception of the work, Design of the work, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionDr. Apostolos Sarivalasis is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2021 Julie Frutiger et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.