|
Case Report
Pneumomediastinum: A severe complication of dermatomyositis
1 MBBS, Mackay Base Hospital, 475 Bridge Road, Mackay, Queensland, Australia
2 FRACP, Mackay Base Hospital, 475 Bridge Road, Mackay, Queensland, Australia
Address correspondence to:
Pranav Kumar
FRACP, Mackay Base Hospital, 475 Bridge Road, Mackay, Queensland, 4740,
Australia
Message to Corresponding Author
Article ID: 101247Z01SL2021
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Lee SZ, Syed MT, Kumar P. Pneumomediastinum: A severe complication of dermatomyositis. Int J Case Rep Images 2021;12:101247Z01SL2021.ABSTRACT
Dermatomyositis is an autoimmune disease that is considered a subset of idiopathic inflammatory myopathy. It is characterized by a skin rash with progressive muscle weakness. Pneumomediastinum is a rapidly progressive complication of dermatomyositis, which is hypothesized to be a result of ruptured subpleural cysts, pulmonary vasculopathy, and steroid-mediated weakening of alveolar walls. This complication is associated with a high mortality rate of 30%. Hence, it should be rapidly investigated and treated when there is a high clinical suspicion. In this article, we report a rare case of patient with dermatomyositis (DM) who developed pneumomediastinum and discussed a literature review of this occurrence.
Keywords: Dermatomyositis, Dyspnoea, Pneumomediastinum
Introduction
Dermatomyositis (DM) is an autoimmune inflammatory myopathy, characterized by muscle weakness in combination with a pruritic or burning rash predominantly present in sun-exposed areas of the body [1]. This report evaluates the presentation of pneumomediastinum (PnM) in a patient with DM. An extensive review of the literature reveals that this finding is not as uncommon as previously thought.
Case Report
A 48-year-old Filipino female was diagnosed with DM, after presenting with Gottron papules, a heliotrope rash, and muscle weakness. Laboratory studies and muscle biopsy were used to confirm her diagnosis. She was treated in the Philippines and was discharged on a drug regime which included prednisolone, hydroxychloroquine (HCQ) 200 mg daily, and mycophenolate mofetil (MMF) 500 mg daily.
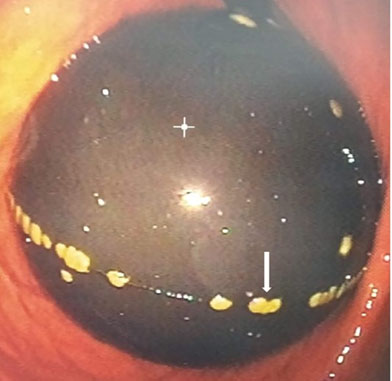
On her return to Australia, her general practitioner (GP) referred her to the outpatient department due to a poor clinical response to treatment after eight months. On outpatient review, she was found to have ongoing cutaneous manifestations and shortness of breath (SOB) (Figure 1A, Figure 1B, Figure 1C). She also had per rectum bleeding, a weight loss of 12 kg in the past one year, severe proximal muscle weakness limiting her mobility, and ongoing arthralgia primarily affecting her wrists, interphalangeal joints, and left knee. On physical examination, she had a blood pressure of 118/78 mmHg, a heart rate of 110 bpm, a respiratory rate of 16 bpm, oxygen saturation of 96% on room air, and was afebrile with a temperature of 37.1°C. She was cachectic with a typical heliotrope rash over her eyelids, Gottron papules localized to her hands and feet, and swollen metacarpophalangeal and carpometacarpal joints. Respiratory examination revealed bibasal, end inspiratory crepitations and coarse crackles. She had proximal weakness with muscle power graded 4/5 in the upper and lower extremities bilaterally.
Initial investigations included a comprehensive laboratory panel, computed tomography (CT) of her chest, abdomen and pelvis, a positron emission tomography (PET)-CT scan, gastroscopy, and colonoscopy. Her creatine kinase (CK) was normal (84 U/L). Her gamma-glutamyltransferase (GGT: 257 U/L), alanine transaminase (ALT: 127 U/L), and aspartate transaminase (AST: 108 U/L) were all elevated. Her autoimmune panel which consisted of rheumatoid factor, antinuclear antibodies, anti-dsDNA, anti-neutrophil cytoplasmic antibodies (ANCA) and extractable nuclear antigens (including anti-soluble liver antigen/liver-pancreas (SLA/LP), anti-LC-1, anti-gp210, anti-PML, anti-sp100, anti-3E, and anti-Mi-2) each returned negative results. Tests for hepatitis, HIV, tuberculosis, syphilis, G6PD deficiency and thyroid disease were also negative.
Imaging of the chest, abdomen, and pelvis revealed extensive PnM and multiple bilateral peripheral patches of consolidation (Figure 2A, Figure 2B, Figure 2C). In her previous chest radiograph five years prior, she was noted to have bilateral apical fibrosis so extensive progression was evident. Following CT, a PET-CT scan was arranged to exclude malignancy and it revealed mild fluorodeoxyglucose (FDG) uptake (SUVmax: 4) within the multifocal, predominantly peripheral, and subpleural pulmonary consolidation. Other findings on imaging included subcutaneous emphysema (SE), peribronchial thickening, and ground glass opacities with mild avidity. There was no dominant nodule or mass visible. There was also mildly increased multifocal FDG uptake throughout the proximal upper and lower limbs and larger muscle groups of the shoulder and pelvic girdles. There was no associated abdominal or pelvic mass or lymphadenopathy. Finally, the colonoscopy and gastroscopy were performed and did not reveal any concerning pathologies.
The patient was commenced on Prednisolone 50 mg once daily. A bronchoscopy was then performed, revealing a posterior tracheal wall disruption (Figure 2D) with bronchoalveolar lavage (BAL) showing bronchial epithelial cells, fungal elements, and alveolar macrophages. She was then prescribed Methylprednisolone IV 500 mg daily and antibiotics consisting of ceftriaxone and clarithromycin. Following clinical improvement on day four of admission, she was discharged from the hospital on Prednisolone 50 mg daily and MMF 1g BD, with outpatient follow-up arranged. Few days later, the patient was seen by her GP and was given antibiotics, which was not effective. She re-presented to the hospital one month later following a two-week history of increasing swelling in her neck and face associated with difficulty swallowing solids; she denied shortness of breath. The patient was arranged for an inter-hospital transfer to a tertiary center for persisting PnM confirmed on repeat chest X-ray (Figure 2E). A repeat bronchoscopy did show same anatomical defect in the tracheobronchial tree. A barium contrast swallow study revealed no concerning features and importantly, no sign of esophageal tear. As the patient showed clinical improvement, she was discharged home. At a one-month review, her chest radiograph (CXR) showed ongoing blunting of the left costophrenic angle, similar to her previous study, and a more prominent extensive SE overlying the neck and chest when compared to previous CXR, suggesting further deterioration. She was admitted to the hospital for observation and was managed conservatively with a constant input from cardio-thoracic team. One stable, she was discharged for outpatient follow-up.
Discussion
Dermatomyositis is an autoimmune condition that involves rash and muscle weakness with an estimated annual incidence of 1.9–7.7 cases per million people and a prevalence of 20 cases per million people with a predilection toward females (2:1) [2]. A five-point criterion was first conceptualized in 1975 by Bohan and Peter, who combined clinical findings, investigation results, and pathological features in order to diagnose this. The requirements include the presence of typical DM rashes, symmetrical proximal muscle weakness, elevated serum levels of muscle-associated enzymes, muscle biopsy showing evidence of myositis and myopathic changes on electromyography. Definite DM was defined as the presence of a rash with three other features; probable DM was defined as the presence of a rash with two other features; and possible DM was defined as the presence of rash with one other feature (Table 1) [3].
Dermatomyositis is sometimes associated with respiratory disease, such as bronchiolitis obliterans, organizing pneumonia, interstitial pneumonitis, and diffuse alveolar damage, in up to 50% of patients [4]. The authors would like to further add that spontaneous PnM is a rare but distinctive complication of DM. The first reported case of PnM in a patient with DM was in 1986 [5]. To date, there are 63 reported cases of PnM in the context of DM, presenting at a mean age of 40.7 years old, with an age range between 10 and 74 years (Table 2) [1],[4],[5],[6],[7],[8],[9],[10],[11],[12],[13]. Dermatomyositis can happen in any age groups, however, previous case reports suggest that are more common in the adult population with only 3 children. In these 63 cases, 36 were male and 27 were female. Pneumomediastinum in DM has been associated with a poor prognosis, especially when the patient has a normal CK level. Previous research reveals that the mortality rate of PnM in DM patients may reach as high as 50%, a rate which is directly related to the severity of the patient’s interstitial lung disease (ILD) [6],[7]. In our literature review of 63 patients, 20 patients (31.7%) had died, with the deaths mostly related to rapidly progressive ILD.
There are three hypotheses surrounding the pathogenesis of PnM in DM. The first of these is the rupture of subpleural cysts secondary to raised intra-alveolar pressure in ILD patients with previously damaged alveoli. Secondly, pulmonary vasculopathy causing disruption of the mucosal barrier and rupture of airway lesions has been proposed. Finally, the use of glucocorticoids in the treatment of ILD resulting in alveolar wall weakening and thus increasing the risk of rupture has also been suggested [4],[8],[9]. Known risk factors for the development of PnM in DM include the presence of ILD, cutaneous vasculopathy, hoarseness of the voice, laryngeal lesions, previous steroid use, younger age, and normal CK levels [8],[10].
Previously reported clinical features of DM-specific patients developing PnM include SOB, DM-specific dermatological features, no or little evidence of muscular involvement, the presence of cutaneous vasculopathy, normal or slightly elevated levels of CK, ILD and a history of systemic glucocorticoid use [13]. The differential diagnoses for dyspnoea in patients with DM should include: pulmonary infections, aspiration pneumonia from esophageal dysfunction, concomitant ILD or drug-mediated ILD, pneumothorax, and hypoventilation from weakened respiratory muscle activity [8].
There are four categories of investigations that can be performed to assess this. These include laboratory studies, imaging, electromyography, and histology. In laboratory testing, the two foci are the assessment of muscle-associated enzyme levels, which include CK, aldolase, lactate dehydrogenase (LDH) and AST, as well as an autoimmune screen [3]. Of the muscle enzyme levels, the most sensitive and specific is CK [3],[14]. Autoimmune assessment of DM is complex and fraught with low sensitivities [15]. Such tests should be subclassified into DM-specific autoantibodies and DM-associated autoantibodies. There are six subtypes of DM-specific autoantibodies, namely, anti-aminoacyl transfer RNA synthetase (ARS) (including Anti-Jo-1) anti-DNA helicase (anti-Mi2); anti-melanoma differentiation-associated gene 5 (MDA5); anti-transcription intermediary factor (TIF-1γ); anti-nuclear matrix protein-2 (NXP-2); and anti-small ubiquitin-like modifier activating enzyme (SAE) [15]. DM-associated antibodies include anti-Ku, which is involved in DNA repair, antinuclear antibody (ANA), and anti-SSA/Ro, which are antibodies for ribonucleoprotein complexes with small cytoplasmic RNAs (hY-RNA) [15],[16].
In terms of imaging modalities, chest radiograph (CXR) has a low sensitivity for early detection but is useful as a baseline assessment of the lungs and to assess for significant ILD. When diagnosed, serial CXR should be conducted to not only assess the progression of the disease, but also to determine the presence of complications, such as spontaneous pneumothorax (PTX), PnM, SE, and infection [17]. High-resolution CT (HRCT) of the chest can provide a better assessment of ILD, including findings of irregular linear opacities, consolidation, ground glass opacities, pleural effusion, and honeycombing, as well as providing information about the location and extent of PTX, PnM, and SE, when CXR is inconclusive [18]. The utility of bronchoscopy includes assessment of the site and size of laryngeal lesions, bronchial wall necrosis and the ability to perform a histological assessment when lung biopsy is performed [6],[10],[19]. Histological findings may include nonspecific interstitial pneumonia, organizing pneumonia, diffuse alveolar damage, and usual interstitial pneumonia [1]. Bronchoalveolar lavage plays a supportive role, as it may provide some information regarding disease progression, however, there is no characteristic BAL cell profile for parenchymal involvement in DM [20].
The management of DM with ILD is complex and involves a multi-disciplinary approach. With regard to dermatological presentation, non-pharmacological management includes avoidance of sunlight and using protective clothing. For extensive erythematous lesions and muscle weakness, steroids are titrated to CK levels. The addition of immunosuppressive agents and anti-pruritic agents are given as per treating rheumatologist or dermatologist recommendations, with due consideration of patient’s tolerability and medication side effects [21]. The most effective treatment for ILD has yet to be decided. Currently, patients are treated with corticosteroids as first-line management. However, since high dose steroids alone are associated with poorer prognosis, patients are often given one or more immunosuppressive agents including azathioprine (AZA), cyclophosphamide (CYC), cyclosporine (CS), mycophenolate mofetil (MMF), and/or intravenous immunoglobulins (IVIG) [13],[21].
Conclusion
In conclusion, PnM is a rapid progressive complication of DM with concomitant ILD. A respiratory physician should regularly follow up patients with PnM to ensure that the condition can be monitored and respiratory function optimized.
REFERENCES
1.
Kaya AG, Çiledağ A, Küçükşahin O, Kumbasar ÖÖ, Atasoy Ç. A case of amyopathic dermatomyositis with pneumomediastinum and subcutaneous emphysema. Case Rep Rheumatol 2015;2015:813902. [CrossRef]
[Pubmed]

2.
Anquetil C, Benveniste O. Dermatomyositis. 2021. [Available at: https://www.orpha.net/consor/cgi-bin/OC_Exp.php?lng=en&Expert=221]

3.
Kaya AG, Çileda? A, Küçük?ahin O, Kumbasar ÖÖ, Atasoy Ç. A case of amyopathic dermatomyositis with pneumomediastinum and subcutaneous emphysema. Case Rep Rheumatol 2015;2015:813902. [CrossRef]
[Pubmed]

4.
Kuroda T, Morikawa H, Satou T, et al. A case of dermatomyositis complicated with pneumomediastinum successfully treated with cyclosporin A. Clin Rheumatol 2003;22(1):45–8. [CrossRef]
[Pubmed]

5.
Bradley JD. Spontaneous pneumomediastinum in adult dermatomyositis. Ann Rheum Dis 1986;45(9):780–2. [CrossRef]
[Pubmed]

6.
Le Goff B, Chérin P, Cantagrel A, et al. Pneumomediastinum in interstitial lung disease associated with dermatomyositis and polymyositis. Arthritis Rheum 2009;61(1):108–18. [CrossRef]
[Pubmed]

7.
Olivo Pallo PA, Shinjo SK. Spontaneous pneumomediastinum in dermatomyositis: A case series and literature review. MedicalExpress (São Paulo, online) 2018;5:mo18009. [CrossRef]

8.
Tang R, Millett CR, Green JJ. Amyopathic dermatomyositis complicated by pneumomediastinum. J Clin Aesthet Dermatol 2013;6(3):40–3.
[Pubmed]

9.
Vinicki JP, Pellet SC, Raimondi A, Dubinsky D, Nasswetter G. Antisynthetase syndrome with subcutaneous emphysema and pneumomediastinum. J Clin Rheumatol 2014;20(7):401–2. [CrossRef]
[Pubmed]

10.
11.
Park SH, Kum YS, Kim KC, Choe JY, Park SH, Kim SK. Pneumomediastinum and subcutaneous emphysema secondary to amyopathic dermatomyositis with cryptogenic organizing pneumonia in invasive breast cancer: A case report and review of literature. Rheumatol Int 2009;29(10):1231–5. [CrossRef]
[Pubmed]

12.
Ma X, Chen Z, Hu W, et al. Clinical and serological features of patients with dermatomyositis complicated by spontaneous pneumomediastinum. Clin Rheumatol 2016;35(2):489–93. [CrossRef]
[Pubmed]

13.
Kim HJ, Hong YK, Yoo WH. Dermatomyositis, complicated with pneumomediastinum, successfully treated with cyclosporine A: A case report and review of literature. Rheumatol Int 2009;29(9):1101–4. [CrossRef]
[Pubmed]

14.
Volochayev R, Csako G, Wesley R, Rider LG, Miller FW. Laboratory test abnormalities are common in polymyositis and dermatomyositis and differ among clinical and demographic groups. Open Rheumatol J 2012;6:54–63. [CrossRef]
[Pubmed]

15.
16.
Yoshimi R, Ueda A, Ozato K, Ishigatsubo Y. Clinical and pathological roles of Ro/SSA autoantibody system. Clin Dev Immunol 2012;2012:606195. [CrossRef]
[Pubmed]

17.
18.
Douglas WW, Tazelaar HD, Hartman TE, et al. Polymyositis–dermatomyositis-associated interstitial lung disease. Am J Respir Crit Care Med 2001;164(7):1182–5. [CrossRef]
[Pubmed]

19.
Rodrigues AJ, Jacomelli M, Scordamaglio PR, Figueiredo VR. Spontaneous pneumomediastinum associated with laryngeal lesions and tracheal ulcer in dermatomyositis. Rev Bras Reumatol 2012;52(5):796–9.
[Pubmed]

20.
Cozzani E, Cinotti E, Felletti R, Pelucco D, Rebora A, Parodi A. Amyopathic dermatomyositis with lung involvement responsive to mycophenolate mofetil. Immunopharmacol Immunotoxicol 2013;35(6):687–92. [CrossRef]
[Pubmed]

21.
Koler RA, Montemarano A. Dermatomyositis. Am Fam Physician 2001;64(9):1565–72.
[Pubmed]

22.
Terao M, Ozawa K, Inui S, Murota H, Yokomi A, Itami S. A case of dermatomyositis complicated with pneumomediastinum. Mod Rheumatol 2007;17(2):156–9. [CrossRef]
[Pubmed]

23.
Agarwal R, Lahiri D, Biswas A, Mukhopadhyay J, Roy MK. Fatal recurrence of pneumothorax in an adult dermatomyositis. J Assoc Chest Physicians 2015;3(1):17–9. [CrossRef]

24.
Masrouha KZ, Kanj N, Uthman I. Late-onset pneumomediastinum in dermatomyositis. Rheumatol Int 2009;30(2):291–2. [CrossRef]
[Pubmed]

25.
Saraya T, Tanaka Y, Ohkuma K, et al. Massive tension pneumomediastinum. Intern Med 2012;51(6):677. [CrossRef]
[Pubmed]

26.
Barvaux VA, Van Mullem X, Pieters TH, Houssiau FA. Persistent pneumomediastinum and dermatomyositis: A case report and review of the literature. Clin Rheumatol 2001;20(5):359–61. [CrossRef]
[Pubmed]

27.
Onishi S, Ono F, Hasegawa H, Yasukawa M. Pneumomediastinum and massive subcutaneous emphysema associated with dermatomyositis. Intern Med 2012;51(24):3449–50. [CrossRef]
[Pubmed]

28.
Korkmaz C, Ozkan R, Akay M, Hakan T. Pneumomediastinum and subcutaneous emphysema associated with dermatomyositis. Rheumatology (Oxford) 2001;40(4):476–8. [CrossRef]
[Pubmed]

29.
Yoshida K, Kurosaka D, Kingetsu I, Hirai K, Yamada A. Pneumomediastinum in dermatomyositis itself is not a poor prognostic factor: Report of a case and review of the literature. Rheumatol Int 2008;28(9):913–7. [CrossRef]
[Pubmed]

30.
Kono H, Inokuma S, Nakayama H, Suzuki M. Pneumomediastinum in dermatomyositis: association with cutaneous vasculopathy. Ann Rheum Dis 2000;59(5):372–6. [CrossRef]
[Pubmed]

31.
Sandhya P, Keshava SN, Danda D, Padhan P, Mathew J, Gibikote S. Pneumorrhachis and pneumomediastinum in connective tissue disease-related interstitial lung disease: Case series from a tertiary care teaching hospital in South India. Rheumatol Int 2012;32(5):1415–9. [CrossRef]
[Pubmed]

32.
Marie I, Dominique S, Janvresse A, Levesque H, Menard JF. Rituximab therapy for refractory interstitial lung disease related to antisynthetase syndrome. Respir Med 2012;106(4):581–7. [CrossRef]
[Pubmed]

33.
Sifuentes-Giraldo WA, García-Villanueva MJ, Gorospe L. Spontaneous pneumomediastinum complicating interstitial lung disease, associated with clinically amyopathic dermatomyositis and positive anti-MDA5 antibodies. Rev Colomb Reumatol 2017;24(4):259–64. [CrossRef]

34.
Mohan V, Cherian SV. Spontaneous pneumomediastinum in dermatomyositis. Am J Med Sci 2017;354(6):e11. [CrossRef]
[Pubmed]

35.
36.
Bakhshaee M, Jokar MH, Mirfeizi Z, Atabati E, Tarighat S. Subcutaneous emphysema, pneumomediastinum and pneumothorax in a patient with dermatomyositis. Iran J Otorhinolaryngol 2017;29(91):113–6.
[Pubmed]

37.
Rekik S, Moalla M, Boussaid S, et al. A rare case of spontaneous pneumomediastinum in dermatomyositis successfully treated With a low dose of rituximab. J Clin Rheumatol 2020;26(5):e144–5.
[Pubmed]

38.
Jain A, Misra DP, Jain VK, Negi VS. Spontaneous pneumomediastinum in dermatomyositis. Indian J Rheumatol 2017;12(1):54–5. [CrossRef]

SUPPORTING INFORMATION
Author Contributions
Shawn Zhenhui Lee - Conception of the work, Design of the work, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Mohammed Tousif Syed - Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Pranav Kumar - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2021 Shawn Zhenhui Lee et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.