|
Case Report
Rectus sheath hematoma in pregnancy following low molecular weight heparin administration
1 The Obstetrics and Gynecology Division, Soroka University Medical Center, Ben-Gurion University of the Negev, POB 151, Beer-Sheva, IL, Israel
Address correspondence to:
Sivan Chocron
The Obstetrics and Gynecology Division, Soroka University Medical Center, Ben-Gurion University of the Negev, POB 151, Beer-Sheva, IL,
Israel
Message to Corresponding Author
Article ID: 100207Z08SC2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Chocron S, Zamstein O, Segal D, Bashiri A. Rectus sheath hematoma in pregnancy following low molecular weight heparin administration. J Case Rep Images Obstet Gynecol 2025;11(1):92–96.ABSTRACT
Introduction: This case report examines a rare complication of low molecular weight (LMW) heparin use during pregnancy. It details the presentation, diagnosis, treatment, and follow-up of rectus sheath hematoma (RSH) associated with enoxaparin prophylaxis.
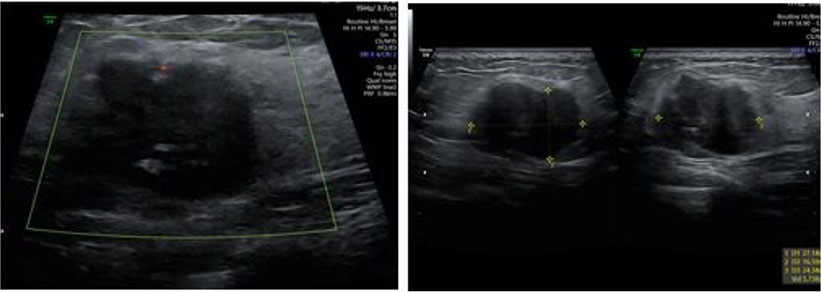
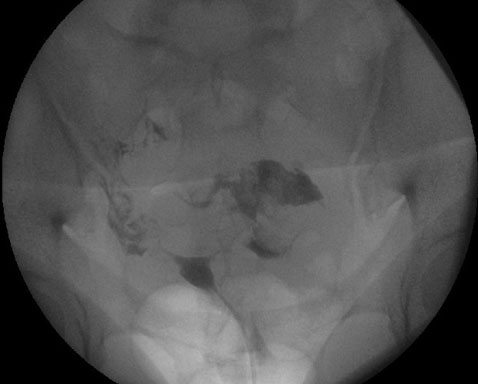
Case Report: A 35-year-old woman with a history of recurrent pregnancy loss and methylenetetrahydrofolate reductase deficiency (MTHFR) mutation was on enoxaparin for venous thromboembolism (VTE) prevention. At 37 weeks gestation, she presented with right abdominal pain and ecchymosis near the injection site, which was diagnosed as RSH. Initially, no further diagnostic tests were conducted. Two days later, she returned with worsening pain, significant hemoglobin drop, and required a blood transfusion. Non-stress testing showed inconclusive fetal heart rate monitoring, prompting an induction of labor. Due to a non-vertex lie, an emergency cesarean section was performed. In the cesarean section extensive blood clots were seen with a dark-colored rectus muscle. The decision was made to leave the clot in place to minimize further bleeding. Postoperatively, she received additional blood and antibiotics, and LMW heparin was stopped and pneumatic stockings for VTE prophylaxis were used. The patient recovered well and was discharged on postoperative day 3. A follow-up ultrasound showed a resolving hematoma at three days, one month, and three months postpartum.
Conclusion: This case highlights rectus sheath hematoma as a rare but serious complication of LMW heparin use during pregnancy. It underscores the need for careful monitoring and risk-benefit assessment of anticoagulant therapy in pregnant patients. Further research is needed to better understand the indications and risk factors for LMW heparin use during pregnancy.
Keywords: Anticoagulant therapy, Low molecular weight heparin, Pregnancy complications, Rectus sheath hematoma
SUPPORTING INFORMATION
Acknowledgments
We would like to extend our sincere gratitude to the Ultrasound Unit at the Obstetrics and Gynecology Division, Soroka University Medical Center for their invaluable support in the management and follow-up of our patient. Their expertise and dedication were crucial to the success of this study. Special thanks to Dr. Joel Baron and Dr. Dan Tirosh for their assistance and commitment throughout the process. All clinical and imaging data, including the ultrasound images presented in this case report, were obtained at Soroka University Medical Center, Obstetrics and Gynecology Division.
Author ContributionsSivan Chocron - Substantial contributions to conception and design, Acquisition of data, Analysis of data, Interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published
Omri Zamstein - Substantial contributions to conception and design, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published
David Segal - Substantial contributions to conception and design, Revising it critically for important intellectual content, Final approval of the version to be published
Asher Bashiri - Acquisition of data, Analysis of data, Interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Sivan Chocron et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.