|
Case Report
A late presentation of uterine rupture following a vaginal birth after cesarean section (VBAC): A case report
1 MBBS, Senior House Officer, Accident and Emergency Department, Queen Elizabeth Hospital, Barbados
2 MBBS, DM, Emergency Medicine, Senior Registrar, Accident and Emergency Department, Queen Elizabeth Hospital, Barbados
3 MBBS PgCUTL DM (UWI), MSc (Aberd), MRCOG, FACOG, Consultant Obstetrician and Gynaecologist, Queen Elizabeth Hospital, Barbados, Lecturer in Obstetrics and Gynaecology at Faculty of Medical Sciences, University of the West Indies, Cave Hill Campus, Barbados
Address correspondence to:
Keturah Murray
Queen Elizabeth Hospital, Martindales Road, St. Michael,
Barbados
Message to Corresponding Author
Article ID: 100177Z08KM2024
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Murray K, Drakes K, Best D. A late presentation of uterine rupture following a vaginal birth after cesarean section (VBAC): A case report. J Case Rep Images Obstet Gynecol 2024;10(2):6–12.ABSTRACT
Over the past 30 years, rates of cesarean section (CS) delivery have increased significantly. According to the World Health Organization (WHO), cesarean section rates have risen from around 7% worldwide in 1990, to 21% in 2021. As a result, physicians are more likely to encounter patients who have opted to undergo vaginal birth after cesarean section (VBAC). Uterine rupture is a rare but potentially life-threatening complication of VBAC. It typically has an acute intrapartum presentation, with abdominal pain, vaginal bleeding, and abnormal cardiotocography; however, there have been a few reported cases occurring postpartum.
Here, we present a case of a 31-year-old woman seen at the emergency department of the Queen Elizabeth Hospital, Barbados, who was found to have a uterine rupture following a VBAC. She had no acute intrapartum signs of rupture, but rather, presented with symptoms consistent with generalized peritonitis and bowel obstruction three days following delivery. She ultimately underwent a laparotomy with repair of the rupture and had an uncomplicated recovery.
Keywords: Labor, Postpartum, Uterine rupture, VBAC
Introduction
Vaginal birth after cesarean delivery (VBAC) can provide a safe means of birth for women who have had a previous lower segment caesarean delivery. As of 2020, the reported rate of trial of labor after cesarean delivery (TOLAC) in the United States of America (USA) was 21.7% [1],[2], and according to the Royal College of Obstetricians and Gynaecologists, the success rate of a planned VBAC is 72–75% [3] based on pooled meta-analyses[4],[5].
Uterine rupture most often occurs in persons with a uterine scar; more than 90% of cases occur in women with a previous cesarean delivery [6]. The risk of rupture varies by prior incision type. Lower transverse incision has rupture rates of 0.2–1.5% [7], while patients with a previous classical uterine incision have risk as high as 4–9% [7]. The published CS rate in Barbados as of 2016 is 21% [8]; however the rate of uterine rupture among the Barbados population is currently unknown.
In an unscarred uterus, the cause of rupture is mainly traumatic or iatrogenic [9]. These occurrences are rare; traumatic ruptures may be seen in pregnant patients involved in motor vehicle accidents or falls from a significant height [10]. Iatrogenic rupture has been reported in association with use of uterine manipulators [11].
Given the very low likelihood of presentation with uterine rupture postpartum, clinicians must maintain a high index of suspicion in any woman presenting with abdominal pain in this period. Imaging modalities such as ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI) may assist with the diagnosis. Once the diagnosis has been made, prompt surgical intervention must be made to reduce maternal morbidity and mortality. A mortality rate of 0.1% in women with a scarred uterus has been reported, compared to 10% following rupture of an unscarred uterus [12].
Case Report
A 31-year-old para 4+1 woman attended the emergency department of the Queen Elizabeth Hospital Barbados on Saturday, October 7, 2023, with a two-day history of frequent episodes of vomiting, numbering more than ten per day. In addition, she complained of reduced appetite, lethargy and severe, cramping lower abdominal pain, which was non-radiating, and gradually worsening. She did not have any diarrhea, fever, or ill contacts. Bleeding per vaginam was minimal, and there were no clots.
She had undergone a spontaneous vertex vaginal delivery of a term female neonate weighing 3475 g (7 lb 10 oz) two days prior. Her labor and delivery were uncomplicated, and the total estimated blood loss was 100 mL. She was discharged two days following delivery, with vital signs including a blood pressure of 124/84 mmHg, pulse of 120 beats per minute (bpm), and oxygen saturation (SpO2) of 97% on room air. Her hemoglobin at the time was 12.3 g/dL. Her medical record suggested that her postpartum stay was routine, with her eating well, ambulating, and breastfeeding. However, upon presentation, the patient now reported that her abdominal pain had in fact begun while on the post-natal ward after her delivery.
Her obstetric history, outlined in Table 1, included one prior lower segment transverse cesarean delivery, one termination of pregnancy, and three subsequent vaginal deliveries, including this most recent one. She had no personal or family history of medical illness. She reported an allergy to non-steroidal anti-inflammatory drugs, and she did not smoke or drink alcoholic beverages.
When examined by an emergency physician, she was visibly ill, and exhibited hemodynamic instability, with a blood pressure of 75/45 mmHg and a pulse of 150 bpm. These were addressed with aggressive intravenous crystalloid fluid replacement; vasopressors were not required. Her abdomen was globally distended, and when palpated, she exhibited generalized abdominal tenderness, with guarding and rebound tenderness. Abdominal percussion revealed tympany. On speculum examination, her cervix appeared normal, with a closed external os, and there were no blood clots, or fleshy material visualized. Upon digital vaginal and bimanual examination, there were no cervical motion tenderness, adnexal tenderness, or uterine tenderness.
A bedside point of care ultrasound (POCUS) revealed distended bowel loops, and free fluid in her Morrison’s Pouch. Her baseline laboratory investigations were as outlined in Table 2 and Table 3.
There was a high suspicion of a postpartum complication based on the immediate post-natal timing and the clinical findings, therefore a decision was taken to proceed with imaging studies looking for any evidence of uterine pathology.
An emergency contrast-enhanced CT scan of the abdomen and pelvis was performed. This showed distended loops of small bowel with no clear transition point, as illustrated in Figure 1 and Figure 2 and reported in Figure 3. A naso-gastric tube was inserted, and this drained 1400 mL of bilious liquid. A urinary catheter was also inserted, which yielded 100 mL of concentrated urine.
On the basis of her symptoms, examination findings, POCUS findings, and nasogastric tube drainage, she was advised by the on-call general surgery team of the need for an urgent laparotomy, for which she gave informed consent. A group-and-crossmatch sample was taken preoperatively. In the hospital operating theatre, a laparotomy was performed using a midline incision.
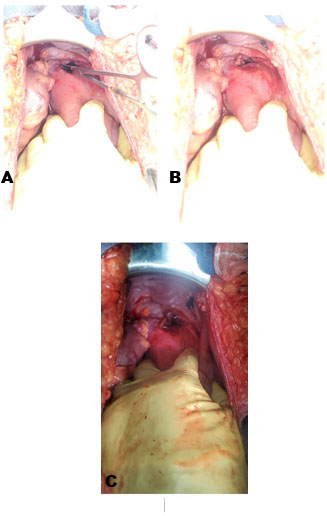
Intraoperative findings included a hemoperitoneum of 600 mL with clots, and an 8 cm longitudinal defect in the lower segment of the anterior uterine wall (Figure 4A, Figure 4B, Figure 4C). Upon discovery of this defect, the on-call obstetrics team was consulted, and they led in its repair. The bladder was found to be adherent to the notably paperthin lower uterine segment. Pieces of fetal membranes were seen in the uterine cavity. The membranes and clots were removed from the cavity, the bladder was dissected off the lower uterine segment and the uterine defect was repaired using a 2-0 polyglactin 910 (Vicryl®) continuous suture. A single layer only was able to be used, owing to how thin the uterus was. Closure of the rectus sheath, subcutaneous fat, and skin were performed with 1 polydioxanone, 1 polyglactin 910 (Vicryl®), and 3-0 polypropylene (Prolene®) respectively.
The patient had an uneventful postoperative period and was discharged after completing a one-week course of intravenous cefazolin and metronidazole. Prior to discharge, she was counseled comprehensively regarding her risk of recurrent rupture in future pregnancies and the recommendation for a cesarean delivery. She was seen six weeks later at the hospital’s obstetrics postnatal clinic where she was noted to be in good health with no issues.
Discussion
Over the past 30 years, rates of cesarean section (CS) delivery have increased significantly. According to the World Health Organization (WHO), cesarean section rates have risen from around 7% worldwide in 1990, to 21% in 2021 [1]. As a result, physicians are more likely to encounter patients who opt to undergo vaginal birth after cesarean section (VBAC). Uterine rupture (UR) is a rare complication seen in patients with a history of previous cesarean delivery. It is defined by a complete tearing of all three layers of the uterus, including the endometrium (inner epithelial layer), myometrium (smooth muscle layer), and perimetrium (serosal outer surface) [12]. While the overall risk after one lower segment CS is low, it is notably higher in patients who undergo VBAC (0.5%), than those who have an elective repeat cesarean section (ERCS) (0.02%) [3]. Uterine rupture most commonly occurs intrapartum, but in some rare instances, it may occur postpartum [13]. This necessitates that physicians maintain a high index of suspicion, even after a successful delivery.
Our patient had two prior uncomplicated VBACs following her first delivery by lower segment cesarean section (LSCS) in 2014. Planned VBAC is appropriate for, and may be offered to, the majority of women with a singleton pregnancy of cephalic presentation at 37+0 weeks or beyond, who have had a single previous LSCS delivery, with or without a history of previous vaginal birth [3]. Successful vaginal delivery rates vary between 72 and 75% [3]. The chance of success increases with the increasing number of prior vaginal deliveries [14]. Previous vaginal delivery, particularly previous VBAC, is the single best predictor of successful VBAC and is associated with a planned VBAC success rate of 85–90% [3]. Mercer and colleagues found that frequency of UR declined with increasing number of successful VBACs, (0.87% with no prior VBACs to 0.43% for those with two or more prior VBACs) [15]; thus our index patient was at perceptibly low risk.
During the antenatal period, all women considering VBAC should be counseled on the benefits and risks of VBAC compared to ERCS. Advantages for women who achieve VBAC include avoidance of major abdominal surgery and lower rates of hemorrhage, thromboembolism, and infection, and a shorter recovery period than women who have an ERCS delivery. In addition, it may decrease the risk of maternal consequences related to multiple cesarean deliveries (for example, hysterectomy, bowel or bladder injury, transfusion, infection, and abnormal placentation such as placenta previa and placenta accreta) [16]. Our patient underwent intrapartum care of the expected standard, including continuous electronic fetal monitoring for the duration of planned trial of labor, commencing at the onset of regular uterine contractions [3]. Unexpectedly, no evidence of uterine rupture arose until she presented two days postpartum!
Uterine scarring from a previous CS is the greatest risk factor for uterine rupture [17], and our patient had a prior lower transverse scar, albeit nine years old, and evidently remaining intact through two prior vaginal deliveries. Women with a previous vertical (inverted T or J shaped or classical CS) uterine incision are at two to three times higher risk for uterine rupture than those with a previous low segment transverse incision [12], and are thus not usually allowed attempts at VBAC. The likelihood of UR is also increased by a short period (<12 or <24 months) after previous CS, augmentation of labor using oxytocin, abnormal fetal position, polyhydramnios, abnormally invasive placenta (especially placenta increta and placenta percreta), placental abruption, connective tissue diseases, adenomyosis, trauma, and uterine abnormalities [18]. None of these risk factors were present in our patient.
Her clinical presentation with abdominal pain was expected, as pain is the most common symptom in postpartum uterine rupture patients [19]. Antepartum patients or those in active labor can also have vaginal bleeding [12]; though most commonly fetal compromise is detected using cardiotocography [19]. She had the expected abdominal tenderness on examination; this is often accompanied by rebound and guarding [19]. Digital examination of uterine cavity may reveal the rupture. However, failure to detect the tear manually does not rule out the diagnosis [20]. In this instance, the patient’s cervix was closed, which would have precluded this finding.
The diagnosis is usually made after imaging studies. The abdominal ultrasound and contrast CT scans of abdomen and pelvis performed on our patient unfortunately failed to detect a uterine rupture and, thus, her diagnosis was subsequently made intraoperatively. In a stable patient with a suspected minor rupture, transabdominal ultrasound (TAS) can be a helpful first line investigation [12]. It is cost-effective, can be performed at the bedside, and does not require ionizing radiation [20]. An abnormality in the uterine wall, a hematoma next to a hysterotomy scar, free fluid in the peritoneum, or in a still-pregnant patient, anhydramnios, or fetal parts outside the uterus, are all findings which support the diagnosis [12]. In the emergency department, CT scan with contrast is probably the most useful test in helping to confirm the diagnosis, and exclude other possible etiologies for abdominal pain, including appendicitis, ovarian vein thrombosis, and mesenteric thrombosis [20]. Compared to ultrasound, CT is more sensitive in confirming a diagnosis of uterine rupture [19], though in the end neither was superior in this case. Magnetic resonance imaging (MRI) was not available at our hospital at the time of writing, but this offers a number of advantages over these other modalities, including allowing for better evaluation of soft tissues than both ultrasound and CT [21]. Magnetic resonance imaging can visualize the uterine wall defect, and provides good anatomical detail of the entire pelvis [22].
The patient’s CT scan showed distended loops of small bowel and fluid-filled (but normal caliber) large bowel, and therefore an initial diagnosis of small bowel obstruction or paralytic ileus was entertained. However, it can be noted that paralytic ileus is the most common cause of intestinal pseudo-obstruction and can be seen in patients such as this with peritonitis, as the entire bowel becomes paralyzed and distended [23].
The most important initial laboratory test is the hemoglobin or hematocrit [12]. If significant bleeding has occurred, coagulation tests should be considered [12]. If significant bleeding has not yet occurred, baseline hemoglobin or hematocrit can be used to monitor for ongoing blood loss [12]. As seen in Table 2, her initial hemoglobin was normal, at 15.6 g/dL.
Our patient had ongoing losses of gastric contents and was volume depleted on examination. The mainstay of care in such cases is volume resuscitation as soon as possible, to restore effective circulatory blood volume, and this was appropriately instituted. It is prudent to start with a warm isotonic crystalloid solution of 30 mL/kg body weight, infused rapidly to restore tissue perfusion quickly. Effective resuscitation can be monitored by heart rate, blood pressure, urine output, mental status, and peripheral edema [24].
Definitive management of peritonitis is an exploratory laparotomy, which she had. This is operation of choice in any patient in whom peritonitis is suspected [25]. Most women survive a uterine rupture, with the mortality rate being lower in women with rupture of a scarred uterus (0.1%) than with an unscarred uterus (10%) [12]. This is likely due to higher vigilance in the former population [12]. The treatment of uterine rupture mainly depends on the location of the rupture, the degree of involvement of the parauterine tissue, the desire for a subsequent pregnancy, and the willingness of patients and their families to make decisions [26]. About 10% of uterine ruptures are managed through hysterectomy, and the remaining cases undergo repair without removal of the uterus [27]. Fortunately, the patient was able to undergo the latter, more conservative approach.
The rate of uterine rupture in subsequent pregnancies depends on the site of the initial scar and type of previous surgery. The overall incidence of uterine rupture in the subsequent pregnancy after classical cesarean delivery was estimated to range from 0.88% to 1.10%. One study showed that up to 10.6% of classical cesarean deliveries resulted in uterine rupture when accompanied by labor in the next delivery. The incidence of uterine rupture after myomectomy has been reported to be 0.25–0.42% after laparotomy and about 1.0% after laparoscopy [27]. If the site of the ruptured scar is confined to the lower segment of the uterus, the rate of repeat rupture or dehiscence in labor is 6%. If the scar includes the upper segment of the uterus, the repeat rupture rate is reported to be as high as 32% [16].
The incidence of recurrent uterine rupture in patients such as this, with a history of uterine rupture, is 10%. Residence in a developed country is associated with a significantly lower incidence of recurrent uterine rupture compared with residence in developing countries (6% in developed countries and 15% in developing countries) [27]. According to the United Nations, Barbados is considered one of the 39 small island developing states [28].
Frank et al. used a decision analysis model with a theoretical cohort of 1000 women with prior uterine rupture and proposed that delivery by CS should be planned between 34 and 36 weeks of gestation [29]. Delivery timing will ultimately be based on an assessment of the balance between complications of prematurity and any evidence of an increased risk of recurrent uterine rupture [27]. Patients with a history of uterine rupture should be instructed to visit the hospital as soon as signs of labor appear, and they need to be evaluated promptly [27]. In addition to recurrent rupture, other known complications of uterine rupture include abnormally invasive placenta (placenta accreta spectrum [PAS]) and hysterectomy [30]. Al-Zirqi reported a rate of 2.8% PAS and 2.8% hysterectomy rate in their study [30].
Conclusion
A successful delivery of the neonate after VBAC may significantly decrease a physician’s suspicion for uterine rupture. This could result in a delay in diagnosis and the institution of appropriate management should a patient present with acute abdominal pain. It is necessary to consider the risk of rupture when attempting VBAC and to maintain a high index of suspicion even after delivery.
REFERENCES
1.
Caesarean section rates continue to rise, amid growing inequalities in access. 2023. [Available at: https://www.who.int/news/item/16-06-2021-caesarean-section-rates-continue-to-rise-amid-growing-inequalities-in-access]

2.
Bruno AM, Allshouse AA, Metz TD. Trends in attempted and successful trial of labor after cesarean delivery in the United States from 2010 to 2020. Obstet Gynecol 2023;141(1):173–5. [CrossRef]
[Pubmed]

3.
Birth after Previous Caesarean Birth (Green-top Guideline No. 45). RCOG. 2023. [Available at: https://www.rcog.org.uk/guidance/browse-all-guidance/green-top-guidelines/birth-after-previous-caesarean-birth-green-top-guideline-no-45/]

4.
Guise JM, Berlin M, McDonagh M, Osterweil P, Chan B, Helfand M. Safety of vaginal birth after cesarean: A systematic review. Obstet Gynecol 2004;103(3):420–9. [CrossRef]
[Pubmed]

5.
Mozurkewich EL, Hutton EK. Elective repeat cesarean delivery versus trial of labor: A meta-analysis of the literature from 1989 to 1999. Am J Obstet Gynecol 2000;183(5):1187–97. [CrossRef]
[Pubmed]

6.
Uterine Rupture - An overview | ScienceDirect Topics. 2023. [Available at: https://www.sciencedirect.com/topics/medicine-and-dentistry/uterine-rupture]

7.
Kan A. Classical cesarean section. Surg J (N Y) 2020;6(Suppl 2):S98–103. [CrossRef]
[Pubmed]

8.
OECD. Caesarean section rates in 27 Latin American and Caribbean countries, 2016 or latest year available. Paris: Organisation for Economic Co-operation and Development; 2020. [Available at: https://www.oecd-ilibrary.org/social-issues-migration-health/caesarean-section-rates-in-27-latin-american-and-caribbean-countries-2016-or-latest-year-available_065ff172-en]

9.
Ali MB, Ali MB. Late presentation of uterine rupture: A case report. Cureus 2019;11(10):e5950. [CrossRef]
[Pubmed]

10.
Suchecki G, Tilden H, Roloff K, Chandwani D, Neeki M. Management of traumatic uterine rupture in blunt abdominal trauma: A case report and literature review. Cureus 2020;12(6):e8396. [CrossRef]
[Pubmed]

11.
Wu HH, Yeh GP, Hsieh TC. Iatrogenic uterine rupture caused by overinflation of RUMI manipulator balloon. J Minim Invasive Gynecol 2005;12(2):174–6. [CrossRef]
[Pubmed]

12.
Togioka BM, Tonismae T. Uterine Rupture. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2024.
[Pubmed]

13.
Choi LA, Chung AA, Pierce B. Late presentation of uterine rupture following vaginal birth after cesarean delivery: A case report. AJP Rep 2020;10(3):e300–3. [CrossRef]
[Pubmed]

14.
Trojano G, Damiani GR, Olivieri C, et al. VBAC: Antenatal predictors of success. Acta Biomed 2019;90(3):300–9. [CrossRef]
[Pubmed]

15.
Mercer BM, Gilbert S, Landon MB, et al. Labor outcomes with increasing number of prior vaginal births after cesarean delivery. Obstet Gynecol 2008;111(2 Pt 1):285–91. [CrossRef]
[Pubmed]

16.
Practice Bulletin No. 184: Vaginal birth after cesarean delivery. Obstet Gynecol 2017;130(5):e217–33. [CrossRef]
[Pubmed]

17.
Marwah S, Singh S, Bharti N, Gupta PK. Risk factors and outcome analysis in rupture of gravid uterus: Lessons for obstetricians. Cureus 2022;14(2):e21890. [CrossRef]
[Pubmed]

18.
Savukyne E, Bykovaite-Stankeviciene R, Machtejeviene E, Nadisauskiene R, Maciuleviciene R. Symptomatic uterine rupture: A fifteen year review. Medicina (Kaunas) 2020;56(11):574. [CrossRef]
[Pubmed]

19.
Waseem M, Cunningham-Deshong H, Gernsheimer J. Abdominal pain in a postpartum patient. J Emerg Med 2011;41(3):261–4. [CrossRef]
[Pubmed]

20.
Has R, Topuz S, Kalelioglu I, Tagrikulu D. Imaging features of postpartum uterine rupture: A case report. Abdom Imaging 2008;33(1):101–3. [CrossRef]
[Pubmed]

21.
Hruska KM, Coughlin BF, Coggins AA, Wiczyk HP. MRI diagnosis of spontaneous uterine rupture of an unscarred uterus. Emerg Radiol 2006;12(4):186–8. [CrossRef]
[Pubmed]

22.
Hamrick-Turner JE, Cranston PE, Lantrip BS. Gravid uterine dehiscence: MR findings. Abdom Imaging 1995;20(5):486–8. [CrossRef]
[Pubmed]

23.
Paralytic Ileus - An overview | ScienceDirect Topics. 2023. [Available at: https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/paralytic-ileus]

24.
Taghavi S, Nassar AK, Askari R. Hypovolemic Shock. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2024.
[Pubmed]

25.
Petersen S, Deder A, Prause A, et al. Transverse vs. median laparotomy in peritonitis and staged lavage: A single center case series. Ger Med Sci 2020;18:Doc07. [CrossRef]
[Pubmed]

26.
Cai YQ, Liu W, Zhang H, He XQ, Zhang J. Laparoscopic repair of uterine rupture following successful second vaginal birth after caesarean delivery: A case report. World J Clin Cases 2020;8(13):2855–61. [CrossRef]
[Pubmed]

27.
Sugai S, Yamawaki K, Haino K, Yoshihara K, Nishijima K. Incidence of recurrent uterine rupture: A systematic review and meta-analysis. Obstet Gynecol 2023;142(6):1365–72. [CrossRef]
[Pubmed]

28.
List of SIDS | Office of the High Representative for the Least Developed Countries, Landlocked Developing Countries and Small Island Developing States. 2024. [Available at: https://www.un.org/ohrlls/content/list-sids]

29.
Frank ZC, Lee VR, Hersh AR, Pilliod RA, Caughey AB. Timing of delivery in women with prior uterine rupture: A decision analysis. J Matern Fetal Neonatal Med 2021;34(2):238–44. [CrossRef]
[Pubmed]

30.
Al-Zirqi I, Vangen S. Pregnancies in women with a previous complete uterine rupture. Obstet Gynecol Int 2023;2023:9056489. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Keturah Murray - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Keisha Drakes - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Damian Best - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2024 Keturah Murray et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.