|
Case Report
Androgenetic/biparental mosaic/chimeric gestation: A case report
1 DO, OB/GYN Resident, Naval Medical Center San Diego, San Diego, CA, USA
2 MD, Maternal Fetal Medicine Staff, Naval Medical Center San Diego, San Diego, CA, USA
Address correspondence to:
Sara Peters
2740 32nd Street, San Diego, CA 92104,
USA
Message to Corresponding Author
Article ID: 100181Z08SP2024
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Peters S, Schmiedecke S. Androgenetic/biparental mosaic/chimeric gestation: A case report. J Case Rep Images Obstet Gynecol 2024;10(2):26–29.ABSTRACT
Introduction: Androgenetic/biparental mosaic/chimeric (ABMC) conceptions are a rare group of gestational trophoblastic disease (GTD) that arise as a result of mosaicism or chimerism and are characterized by hydropically enlarged and variably sized villi. Currently, there are very few reports describing ABMC conceptions; there are 13 total in the literature, and even more rare are those with a molar component.
Case Report: A 27-year-old female patient at 6+4wga presented to the clinic with a complaint of new vaginal bleeding and was diagnosed with a pregnancy of unknown location. The patient re-presented to the clinic at 8+4wga by last menstrual period (LMP) for a follow-up ultrasound. A serum human chorionic gonadotropin (hCG) was ordered and returned at 424,008 mIU/mL. A transvaginal ultrasound demonstrated a heterogeneous “snowstorm” appearance throughout uterine cavity, with a concern for complete mole in the setting of a significantly elevated beta hCG. The patient underwent a suction dilation and curettage with ultrasound guidance. Pathology returned the following report: The specimen was sent to pathology and DNA ploidy returned as diploid; however, p57 demonstrated a discordant staining pattern characterized by expression in villous cytotrophoblast but the absence in villous stromal cells was characteristic of androgenetic/biparental mosaic/chimeric gestation. No fetal parts were identified in gross examination of the specimen.
Conclusion: Androgenetic/biparental mosaic/chimeric (ABMC) conceptions are important to accurately diagnose because the molar form carries an increased risk for persistent gestational trophoblastic diseases. There are few case reports published regarding ABMC cases. We present this case to provide additional data to the field and emphasize the utility for p57 immunohistochemistry and genetic testing to be performed.
Keywords: Chimerism, Dispermy, Gestational trophoblastic disease, Hydropic, Monospermy, Mosaicism
Introduction
Androgenetic/biparental mosaic/chimeric (ABMC) conceptions are a rare group of gestational trophoblastic disease (GTD) that are difficult to diagnose and recognize. They are composed of a mix of two cell populations, one androgenic and one biparental, and are a combination of morphologic, immunohistochemical, and genotypic features: multiple villous populations, discordant p57 immunostaining, and mixed genotypes. Androgenetic/biparental mosaic/chimeric conceptions arise as a result of mosaicism, mitotic error in a zygote, or chimerism, fusion of two zygotes, and are characterized by hydropically enlarged and variably sized and shaped villi [1]. Androgenetic/biparental mosaic/chimeric conceptions are important to accurately diagnose because the molar form carries an increased risk for persistent gestational trophoblastic diseases [2]. Currently, there are very few reports describing ABMC conceptions; there are 13 total in the literature, and even more rare are those with a molar component [1],[2].
Case Report
A 27-year-old female patient at 6+4 weeks gestational age (wga) presented to the clinic complaining of vaginal bleeding. The patient reported a history of a spontaneous abortion in the previous year, at approximately eight weeks. She had regular menses and was monogamous without a history of sexually transmitted infections. The patient denied current use of contraception nor a history of abnormal pap smears. She had no other comorbidities during this pregnancy, including diabetes or hypertensive disorders.
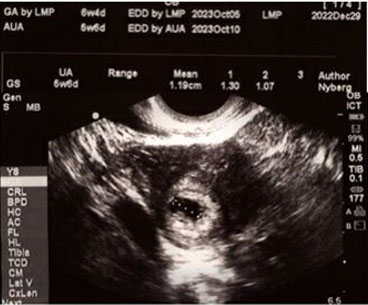
The patient was diagnosed with a pregnancy of unknown location due to visualization of an intrauterine gestational sac without a yolk sac or fetal pole (Figure 1). The patient was instructed to follow up in 14 days for a viability check, as fetal cardiac activity should be seen by this time if it is a normal pregnancy.
The patient re-presented to the clinic at 8+4wga by LMP for a follow-up ultrasound. She reported minimal brown spotting 1–2 days prior to this appointment without any cramping but did endorse nausea and breast tenderness at this time. Additionally at this appointment, she stated that she had presented to a different obstetric provider three days earlier without an intrauterine pregnancy observed. Normotensive blood pressures with a maternal heart rate of 114 bpm were measured in the clinic. A serum hCG was ordered and returned at 424,008 mIU/mL. A transvaginal ultrasound (TVUS) performed in the clinic found a heterogeneous “snowstorm” appearance throughout uterine cavity (Figure 2), with a concern for complete mole in the setting of a significantly elevated beta hCG. No obvious fetal parts were identified. Based on the laboratory and imaging results, a uterine evacuation via dilation and curettage was recommended for a suspected complete molar pregnancy, which the patient agreed to.
The procedure was carried out three days later, with the pre-operative beta hCG level demonstrating an uptrend to 472,526 mIU/mL and the labs also notable for acutely elevated liver transaminases (aspartate aminotransferase (AST)/alanine aminotransferase (ALT) 129/50). The chest X-ray obtained for a gestational trophoblastic disease workup was normal. The patient ultimately underwent a suction dilation and curettage, performed under ultrasound guidance in the operating room (OR). On exam, the patient had a mobile uterus on bimanual exam, approximately 10–12 weeks in size. Intraoperative findings were notable for cystic structures spontaneously passing from the cervix after the cervical dilation was initiated.
The material from the curettage was sent for pathological examination. Pathology returned the following report: The specimen was sent to Esoterix Genetic Laboratories, LLC for DNA ploidy and p57 testing. DNA ploidy returned as diploid; however, p57 demonstrated a discordant staining pattern characterized by expression in villous cytotrophoblast but the absence in villous stromal cells was characteristic of androgenetic/biparental mosaic/chimeric gestation (Figure 3). No fetal parts were identified in gross examination of the specimen. In order to definitively diagnose androgenetic/biparental mosaicism, further workup should have been performed, including short tandem repeat (STR) genotyping of the collected tissue. Additionally, a complete mole can only be confirmed when the loss of p57 in both cytotrophoblast and villous stromal cells and abnormal trophoblastic hyperplasia are visualized. Unfortunately, neither of these tests were performed In this case.
The patient’s follow-up care was managed using the International Federation of Gynecology and Obstetrics (FIGO) recommendations, as referenced in Society of Gynecologic Oncology’s (SGO) evidence-based review [3]. The patient’s serum beta hCG was monitored until undetectable (Table 1). The patient was then instructed to return for monthly labs; however, she was lost to follow-up and did not receive additional labs after the level was undetectable.
Discussion
There are only 13 cases of androgenetic/biparental mosaic/chimeric conceptions currently available in the literature, 11 of which were characterized by Lewis et al. and identified out of 473 total products of conception that underwent p57 immunostaining and short tandem repeat genotyping [1],[2].
It is important to identify this group of gestational trophoblastic disease, as the molar component increases the risk for gestational trophoblastic neoplasia (GTN) in patients. Making an accurate diagnosis is largely reliant on ordering the pathology correctly. Specifically, one should be sure to include a note regarding their concern for a molar pregnancy with any suspected case of a mole and request that DNA testing is performed. Androgenetic/biparental mosaic/chimeric conceptions are important to identify because they dictate clinical follow-up. Regardless of the molar component being identified on pathology, it is recommended that all cases be treated like a complete mole for follow-up [3].
We recognize the shortcomings of this case, including the lack of genetic testing to confirm both the androgenetic/biparental mosaicism as well as the complete mole. It is not uncommon, at least at our facility, for genetic testing to be missing in the workup simply because it was not requested and the lab does not reflexively perform genetic testing in these cases. Thus, we cannot emphasize enough the importance of ordering confirmatory genetic testing and further pathologic evaluation of the specimen when there is concern for a molar pregnancy.
Conclusion
Androgenetic/biparental mosaic/chimeric conceptions are rare, and thus we add this additional case to the literature. These conceptions require unique genetic and histologic studies in order to be identified. Pathologists should be encouraged to thoroughly examine the products of conception tissue for ABMC when atypical p57 immunostaining, diverse chorionic villi, and complex genotyping are discovered.
REFERENCES
1.
Hodgson A, Dube V, Strickland S, et al. Androgenetic/biparental mosaic/chimeric conceptions with a molar component: A diagnostic and clinical challenge. Int J Gynecol Pathol 2021;40(5):510–7. [CrossRef]
[Pubmed]

2.
Lewis GH, DeScipio C, Murphy KM, et al. Characterization of androgenetic/biparental mosaic/chimeric conceptions, including those with a molar component: Morphology, p57 immnohistochemistry, molecular genotyping, and risk of persistent gestational trophoblastic disease. Int J Gynecol Pathol 2013;32(2):199–214. [CrossRef]
[Pubmed]

3.
Horowitz NS, Eskander RN, Adelman MR, Burke W. Epidemiology, diagnosis, and treatment of gestational trophoblastic disease: A society of gynecologic oncology evidenced-based review and recommendation. Gynecol Oncol 2021;163(3):605–13. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Acknowledgments
Thank you to Sarah A. L. Caccamise, Ph.D. for the support in editing and completing this manuscript.
Author ContributionsSara Peters - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Stacey Schmiedecke - Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2024 Sara Peters et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.