|
Case Report
Vulvar extramedullar relapse (genitomegaly) of acute lymphoblastic leukemia
1 Professor, Dr, Department of Hematology-Oncology, Faculty of Medicine, Firat University, Elazig, Turkey
2 Professor, Dr, Department of Radiology, Faculty of Medicine, Firat University, Elazig, Turkey
Address correspondence to:
Saadet Akarsu
Professor, Dr, Department of Hematology Oncology, Faculty of Medicine, Firat University, Elazig,
Turkey
Message to Corresponding Author
Article ID: 100192Z08SA2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Akarsu S, Poyraz AK. Vulvar extramedullar relapse (genitomegaly) of acute lymphoblastic leukemia. J Case Rep Images Obstet Gynecol 2025;11(1):11–16.ABSTRACT
Introduction: Acute lymphoblastic leukemia (ALL) is the most common childhood malignancy. Relapse of ALL occurs in 15–20% of patients, with 2–6% occurring exclusively in extramedullary sites. Extramedullary relapse of leukemia is encountered more often than in the past. The reason is that leukemia survival rates increase with improved treatment schemes. The relapse of ALL usually involves the bone marrow, with the central nervous system being the most frequent extramedullary site. Relapses affect various organs but rarely the female genital tract. The frequency of gynecological involvement, from high to low, is ovary, cervix, uterus, and vulva.
Case Report: We wanted to present a case of vulvar extramedullary recurrence one year after the application of an inadequate ALL chemotherapy protocol.
Conclusion: The survival time of leukemia patients is increasing. The genital area is an area that chemotherapy drugs rarely penetrate. The physicians should be more careful about the genital area after the treatment of girls with leukemia.
Keywords: Acute lymphoblastic leukemia, Aleukemic cutis, Extramedullary relapse, Genital tract
Introduction
Relapse of acute lymphoblastic leukemia (ALL) occurs in 15–20% of patients, with 2–6% occurring exclusively in extramedullary sites. The relapse of ALL usually involves the bone marrow. The central nervous system (CNS) and the testis represent the primary site of extramedullary disease in patients with ALL. Relapses affect various organs but rarely the female genital tract. Relapse of ALL in gynecologic organs is extremely rare [1],[2]. We report a case of recurrence of ALL in the skin and subcutaneous areas of the genital area during complete hematological remission.
Female genital tract and particularly uterus involvement in acute ALL is extremely rare. To our knowledge, only three cases described in the literature involve the uterine cervix. It has been reported that uterine involvement was detected in a 64-year-old woman [3],[4],[5],[6]. Pelvic extramedullary leukemic relapse in females may be encountered more frequently in the future. Currently attention is focused on the testicle as a site of leukemic relapse [7]. The frequency of gynecological involvement, from high to low, is ovary, cervix, uterus, and vulva [8]. Leukemia cutis is regarded as a systemic manifestation of underlying leukemia and generally carries an unfavorable prognosis [9]. Here we present a case of vulvar (skin and subcutaneous areas of the genital area) extramedullary (EM) relapse (EMR), a rare site, in a patient in remission.
Case Report
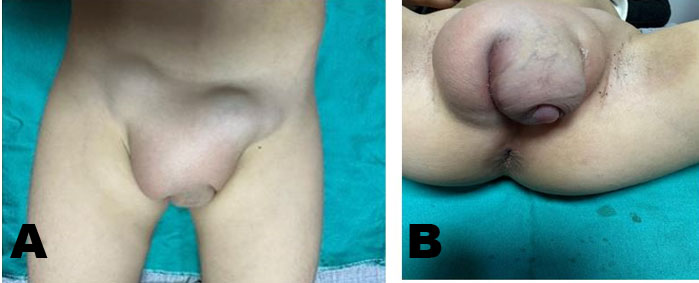
A 3.5-year-old girl was brought with the complaint of swelling in the vulvar area (The size of the vulvar swelling was 63×63×72 mm). Two years ago, she was diagnosed with infant ALL (CD10 60%, CD19 80%, B ALL, cerebrospinal fluid normal, genetic 15% cells in 11q23) and AALL0631 chemotherapy protocol was applied. While the patient had bone marrow remission in the 6th month of chemotherapy, the parents rejected chemotherapy. Two years after stopping chemotherapy for insufficient duration, the patient was brought with findings of infiltration in the vulvar area (genitomegaly, EMR, and bone marrow in remission). On physical examination, her general condition was good and she was active. The gums, eyes, and other areas of skin were completely normal. Organomegaly was not detected. Starting from the symphysis pubis (Figure 1A), there was soft tissue infiltration with a hard consistency extending to the perineum (Figure 1B). Lymph nodes (25×25 mm) were detected in the bilateral inguinal region (Figure 2A). There were no skin findings other than the genital area. Bilaterally, the labia majora were infiltrated with a hard consistency (Figure 2B). Routine biochemical and preliminary hematological investigations including a peripheral blood smear examination were performed. White blood cell count (WBC) 9.620/mm3, hemoglobin (Hb) 12.8 g/dL, and platelet count (Plt) 366×109/L were detected. In the peripheral smear, the eosinophil rate has increased by 11% and no blasts were observed. Lactate dehydrogenase (LDH) 310 U/L and uric acid were determined to be normal. Abdominal and pelvic ultrasonography revealed multiple lymphadenopathies in the right inguinal region, the largest of which was 55×37 mm in size, had thinned fatty hilus, was obliterated, had a prominent thick cortex, and contained cortical heterogeneity. Uterine, vaginal, and ovarian imaging was evaluated as normal. No blasts were observed in bone marrow aspiration and biopsy. 22% eosinophils were observed in the bone marrow.
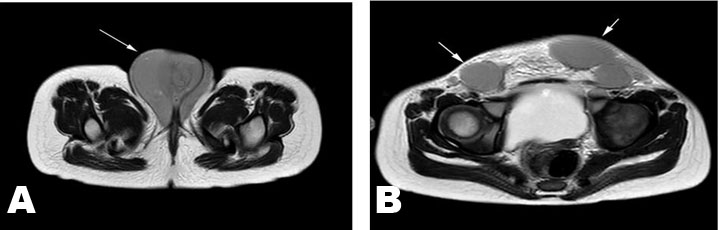
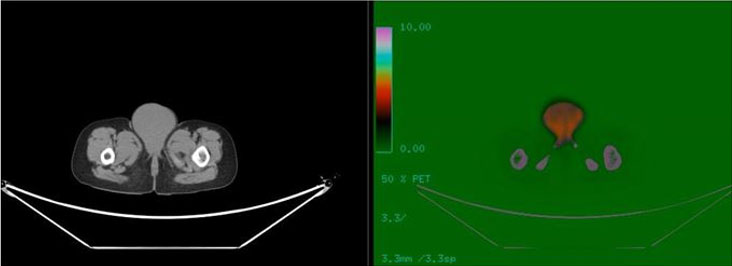
Magnetic resonance imaging (MRI) revealed an intact uterus, vagina, and ovary (Figure 2A and Figure 2B). In positron emission tomography/computed tomography (PET/CT), starting from the anterior of the symphysis pubis; increased fluorodeoxyglucose (FDG) uptake at the level of malignancy (SUVmax: 4.2) was detected in the soft tissue density mass (55×56×70 mm at its widest point) extending to the perineum (skin and subcutaneous), involving the bilateral labium majus, and having no boundaries with the vagina. The bladder and ureters were dilated and hydropic. It was evaluated as a PET/CT study compatible with malignant (SUVmax: 4.5) lymph nodes in the bilateral inguinal region, the largest of which was measured as 30×27 mm. Spleen and liver SUVmax (respectively 1 and 1.2) values were normal for her age (Figure 3A and Figure 3B). Biopsy from the genital area showed ALL infiltration into the skin and subcutaneous tissue with the same phenotype as the primary disease (proliferation of ALL cells). Reinduction chemotherapy was immediately started. Our patient was included in the bone marrow transplant program. During this period, the patient’s treatment was completed successfully.
Discussion
Acute lymphoblastic leukemia is the most common childhood malignancy. Relapse of ALL occurs in 15–20% of patients, with 2–6% occurring exclusively in extramedullary sites (leukemic relapse isolated to sites outside the bone marrow). The relapse of ALL usually involves the bone marrow, with the central nervous system (CNS) being the most frequent extramedullary site [1]. Hematolymphoid malignancies may involve the genital area in girls and boys [10]. Relapses affect various organs but rarely the female genital tract [1],[2].
The generally accepted definition of an isolated EMR is the presence of a new, isolated focus of EM leukemia, which occurs during a bone marrow relapse within 30 days [11]. Extramedullary localizations of acute myeloid leukemia (AML) are called “granulocytic sarcoma,” “chloroma,” or “myeloid sarcoma (MS).” Myeloid sarcoma is mainly reported in soft tissues, bone, peritoneum, and lymph nodes [12]. Leukemia cutis characteristically demonstrates the infiltration of the skin by neoplastic leukocytes. While EM collection of leukemic cells is considered MS, leukemia cutis describes specific cutaneous involvement. Between 25% and 30% of children with congenital leukemia are prone to developing leukemia cutis. In 2–3% of cases, skin lesions occur in the peripheral blood and/or bone marrow even before the development of leukemia. This condition termed as aleukemic cutis eventually develops leukemia [9]. The common sites of EM after hematopoietic stem cell transplantation (HSCT) coincide with those of primary MS, which include those of the breast, gastrointestinal tract, skin, and spine, in addition to the well-known sanctuary sites of the CNS and testis. Other sites of EM relapse are the pancreas, nasopharynx, paranasal sinus, bladder, peritoneal cavity, and pleura. Although it has been reported that this development occurred in a single patient in ALL, no information has been published about uterine MS after HSCT for AML [11].
Because differential leukemic development may occur, a skin biopsy and immunophenotyping must be performed in all suspected patients regardless of previous leukemia history. The identification of leukemia can be performed based on cluster of differentiation (CD) markers [9]. The initial diagnosis of our patient was infant B-cell ALL. At the time of discovery in the genital area, peripheral blood smear and bone marrow samples did not contain leukemic cells.
The pathogenesis of EMR in patients with ALL is not fully understood. The CNS, testes, and uterus are sanctuary areas, perhaps due to the poor drug penetration of systemic chemotherapy into these areas [3]. The factors predisposing primary MS apply equally to isolated EMR, and include cytogenetic abnormalities such as t(8;21) and inv [13], the mixed-lineage leukemia (MLL) rearrangement, and French-American-British (FAB) classification M2, M4, and M5 subtype. A specific cell surface molecule expression may be on possible explanation [11].
The incidence of EMR after allogeneic hematopoietic stem cell transplantation (allo-HSCT) varies between 6% and 20%. The incidence of EMR in ALL patients (12.9%) is higher than in AML patients (4.6%) [14]. Extramedullary relapse of ALL following allo-HSCT has a poor prognosis [15]. Microscopic evaluation and immunohistochemistry studies make the definitive diagnosis. The prognosis is poor in case of recurrence of EM disease in the female genital system [1]. Two patients (23-year-old AML and 49-year-old ALL) presenting with EM tumors in the ovary following allo-HSCT have been previously reported [15].
Acute myeloid leukemia may extend to EM sites at diagnosis or at relapse, either isolated or associated with bone marrow disease. Myeloid sarcoma of uterine cervix is rare [12]. The occurrence of relapse in the uterus may indicate a case of leukemia that is particularly difficult to eradicate, although it may initially respond to chemotherapy [3]. Therefore, we started chemotherapy for our patient and included her in the bone marrow transplant program.
French-American-British subtype M4 and M5 are mostly associated with EM tissue involvement. Myeloid sarcoma, which represents a rare malignancy that comprises of myeloid blasts occurring at extramedullary sites, closely correlates with the onset and relapse of AML and other hemopoietic neoplasm. Female genital system is an uncommon location of MS. The vulvar MS is more rarely seen. The incidence of MS is between 1.1% and 9.1% in patients with AML, myelodysplastic syndromes (MDS), or myeloproliferative neoplasms (MPN). Myeloid sarcoma occurs in nearly any sites, and the most common sites include lymphoid tissues, CNS, lung, kidney, and gastrointestinal tract. Female genital system is a much rarer location that less than a hundred cases have been reported. Gynecological MS is listed as cervix (40%), ovary (23.6%), vulva (10.9%), uterine body (5.5%), and vagina (3.6%). Multifocal lesions are present in one-sixth of the cases [8]. Leukemic involvement of the ovary has been reported to be 11–50% in patients with bone marrow recurrence. Ovarian involvement has been rarely reported during the clinical course of leukemia [7]. Penile infiltration is rare [8]. Myeloid sarcoma of the uterine cervix requires systemic treatment such as chemotherapy [12]. The uterus could be a site of relapse in patients suffering from ALL. Even though an MRI examination could better demonstrate the disease in cases of suspected female genital tract involvement by ALL, the comparison of differences between a present and a previous CT examination is sufficient to suspect the diagnosis [6].
In the differential diagnosis of leukemia cutis, neoplastic, inflammatory, and infectious skin lesions (lymphoma, solid tumors, pyoderma gangrenosum, vasculitis, drug reactions, urticaria, viral exanthema, hidradenitis, erythema multiforme, erythema nodosum, Graft vs. host disease) should be considered [9].
Cutaneous lesions may appear at the presentation of leukemia (23–44%). The clinical and morphological findings of leukemia cutis vary widely. These lesions may be localized or disseminated. It may occur alone or in combination on any site of the skin. It is one of the causes of blueberry muffin syndrome in infants [9].
The most common types of leukemic cutis are chronic lymphocytic leukemia (CLL) and AML with monocytic or myelomonocytic morphology. Involvement by chronic myeloid leukemia (CML) is extremely rare. Patients usually have a prior diagnosis of leukemia or myelodysplasia. In rare cases, skin lesions may occur as the primary symptom of systemic disease. In AML patients, a survival rate of 6% in those with skin involvement and 30% in those without skin infiltration has been demonstrated. Cutaneous involvement is generally indicative of advanced disease and should stipulate the investigation of other body sites for EM involvement [9]. Our patient was infant leukemia and was diagnosed at the age of one year.
Leukemia cutis is not uncommon in patients with leukemia, especially AML [8]. Its pathophysiology is due to the migration of leukemic cells into the skin. The exact mechanism is unknown. Recent molecular analyses provide more information about on cell–cell interaction and the role of adhesion molecules. Mechanisms involving various chemokines and molecular expression on leukemic cells mediating the migration of leukemic cells to the skin through skin-selective homing processes are the proposed etiology. While neoplastic cells carry the genetic abnormality of the underlying leukemia, aneuploidy of chromosome 8, chromosome 3 translocations, and t(6;9) are particularly striking [9]. Leukemia cutis is often associated with an aggressive disease. The survival rate is different in patients with leukemia cutis. Biopsy shows diffuse dermal infiltration of monomorphic, immature blast cells with hyperchromatic, oval to irregularly shaped nuclei, occasional indentations, abundant pale staining to granular cytoplasm, scattered mitoses, and immature eosinophilic cells. Tumor cells infiltrate around vascular channels [16]. No EMR was observed in the CNS and other organs of our patient. Since there was aleukemic cutis in the genital area, the ovary and uterus that were examined were found to be intact. 11q23 was detected in our patient’s genetics.
Malignancy (0.8%) is among the infrequent causes of eosinophilia. Lymphoid neoplasms are well-known diseases that presenting with eosinophilia long before the detection of underlying malignancies [13]. Eosinophilic dermatosis of hematological malignancy (EDHM) occurs in patients with a variety of hematological neoplasms. It is a chronic, recurrent itchy skin disease. Its pathogenesis is not fully understood. It has been linked to hematoproliferative disorders and cytokine imbalance (excess of IL-4, which plays a major role in type 2 immune responses, and of IL-5, which facilitates the production, differentiation, and migration of eosinophils). Its prognostic significance is unclear. Eosinophilic dermatosis of hematological malignancy may be associated with aggressive hematological diseases. Chemotherapy may play a role in the development of EDHM. Monomorphic urticarial plaques or polymorphic purpuric papules and nodules occur on the extremities, face, and trunk. A wide variety of clinical features have been described, ranging from erythematous papules or nodules to blisters and vesicles, urticarial or cellulite-like plaques, or cutaneous ulcerations. It is most commonly seen together with CLL. In our patient, infiltration covering only the genital area was detected without rash. The rate of eosinophilia in our patient was 11% in the peripheral blood smear and 22% in the bone marrow. This situation can be explained by tissue/organ damage. Eosinophilic dermatosis of hematological malignancy is part of the reaction type associated with Wells’ syndrome (WS). Wells’ syndrome is a rare dermatosis characterized by a variety of pruritic cutaneous symptoms that are usually recurrent over several years. It can present in morphologically varying forms, such as a plaque-type lesion, annular granuloma-like, urticaria-like, papulovesicular, bullous, papulonodular, or fixed drug eruption-like. Different lesions may occur simultaneously in a patient. It is mostly located on the extremities. It has been associated with diseases such as hematological diseases and arthropod bites. Peripheral eosinophilia, leukocytosis, or elevated inflammatory markers may be observed. Histopathological findings of WS are nonspecific. It includes subepidermal edema, granulomas, and frequent flame figures [17].
Treatment of leukemia cutis points toward addressing underlying leukemia using aggressive systemic chemotherapy. Systemic chemotherapy for leukemia depends on cancer subtype. Usually, remission of hematological abnormalities occurs concurrently with complete or partial resolution of cutaneous lesions. While chemotherapy is the mainstay of treatment, radiation therapy, in the form of electron beam therapy may be used for refractory or palliative cases [9].
Patients with hematological malignancies involving the genital tract usually present with symptoms of vaginal bleeding, abdominal pain due to a pelvic mass along with other constitutional symptoms such as B symptoms (weight loss, night sweats, and fever). Our patient presented with nonspecific diffuse vulvar mass [18]. Hematolymphoid malignancies rarely present as penile lesions (penile mass, balanoposthitis, and priapism) in men [10]. B-cell lymphoblastic lymphoma (LBL) is a highly aggressive malignant proliferation of lymphoblasts of B-origin grouped with ALL. Multiple studies have demonstrated the distribution of extramedullary sites involved among patients with LBL. These sites were mainly the pleura, pericardium, CNS, liver and spleen, and to a lesser extent the skin and kidneys. However, the involvement of the female genital tract is rare and accounts for <1% of extranodal lymphoma and/or leukemia [18]. Isolated EMR of ALL in the breast is extremely rare [15]. In the context of known hematological malignancy, namely LBL, any soft tissue mass can represent infection, secondary neoplasms, or relapse of primary disease [18]. Chronic lymphocytic leukemia in the vagina may present as an asymptomatic lesion. Hematologic malignancies should be considered in the differential diagnosis during workup of abnormal vaginal cytology [19].
Positron emission tomography scan is sensitive to detect tumors with a high rate of proliferation. The genital system is rarely affected by hematological malignancies [18]. Our case has been reported to involve the external vulvar structure without affecting the internal genitalia in PET/CT evaluation.
The majority (60–90%) of patients with ALL achieve complete remission after initial treatment. However, relapses also occur. Recurrence will occur at a rate of 1/3 in standard risk ALL and 2/3 in high risk ALL. Regardless of the type of initial treatment (bone marrow transplant or chemotherapy), one-year survival is 22% and five-year survival is only 7% [1],[20],[21],[22],[23]. The prognosis for EMR of acute leukemia (AL) is poor. Treatment options are very limited. The estimated 3-year overall survival (OS) is lower in patients with AML patients [14]. Prognosis in the situation of EMR in the female genital tract is poor [2]. Chemotherapy has been found to be more effective than radiation or surgical radiation therapy. Additionally, allogeneic or autologous BM transplantation has been appeared to increase the odds of prolonged survival [8]. Our patient was taken to the HCST program after remission. Since these tumors (EMR) are extremely radioresponsive, radiation therapy (RT) is a safe and effective treatment option for isolated EMR of ALL [15]. Due to our patient’s low age, RT was not performed due to fear of secondary malignancy.
Some patients experience improvement in skin lesions after systemic chemotherapy. The diagnosis of leukemia cutis is strongly associated with additional sites of EM involvement. The hematoencephalic barrier may not be crossed by intravenous chemotherapy [9].
Conclusion
The survival time of leukemia patients is increasing. The genital area is an area that chemotherapy drugs rarely penetrate. The physicians should be more careful about the genital area after the treatment of girls with leukemia
REFERENCES
1.
Robillard DT, Kutny MA, Chewning JH, Arbuckle JL. Extramedullary relapse of acute lymphoblastic leukemia presenting as abnormal uterine bleeding: A case report. J Pediatr Adolesc Gynecol 2017;30(3):431–4. [CrossRef]
[Pubmed]

2.
Tsagkas N, Troussa A, Vorgias G, Tzaida O, Zagorianakou N, Gonidi M. Extramedullary leukemia, presenting at the cervix of the uterus. Case Rep Obstet Gynecol 2020;2020:8492036. [CrossRef]
[Pubmed]

3.
Imbriaco M, De Iuri AB, Camera L. Isolated uterine relapse in a child with acute lymphoblastic leukemia. AJR Am J Roentgenol 1998;171(4):1166–7. [CrossRef]
[Pubmed]

4.
Zutter MM, Gersell DJ. Acute lymphoblastic leukemia. An unusual case of primary relapse in the uterine cervix. Cancer 1990;66(5):1002–4. [CrossRef]
[Pubmed]

5.
Tsuruchi N, Okamura J. Childhood acute lymphoblastic leukemia relapse in the uterine cervix. J Pediatr Hematol Oncol 1996;18(3):311–3. [CrossRef]
[Pubmed]

6.
Mazzei MA, Bettini G, Pozzessere C, et al. A solitary uterine relapse in T-cell acute lymphoblastic leukaemia: CT features and pathologic correlation. J Biol Regul Homeost Agents 2016;30(3):871–5.
[Pubmed]

7.
Cecalupo AJ, Frankel LS, Sullivan MP. Pelvic and ovarian extramedullary leukemic relapse in young girls: A report of four cases and review of the literature. Cancer 1982;50(3):587–93. [CrossRef]
[Pubmed]

8.
Zhang X, Huang P, Chen Z, Bi X, Wang Y, Wu J. Vulvar myeloid sarcoma as the presenting symptom of acute myeloid leukemia: A case report and literature review of Chinese patients, 1999–2018. Diagn Pathol 2019;14(1):126. [CrossRef]
[Pubmed]

9.
Parsi M, Go MS, Ahmed A. Leukemia Cutis. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2023.
[Pubmed]

10.
Gatto-Weis C, Topolsky D, Sloane B, Hou JS, Qu H, Fyfe BS. Ulcerative balanoposthitis of the foreskin as a manifestation of chronic lymphocytic leukemia: Case report and review of the literature. Urology 2000;56(4):669. [CrossRef]
[Pubmed]

11.
Maeng H, Cheong JW, Lee ST, et al. Isolated extramedullary relapse of acute myelogenous leukemia as a uterine granulocytic sarcoma in an allogeneic hematopoietic stem cell transplantation recipient. Yonsei Med J 2004;45(2):330–3. [CrossRef]
[Pubmed]

12.
Beziat G, Tavitian S, Arnaud FX, et al. Brachytherapy for uterine cervix-limited acute myeloid leukemia relapse. J Contemp Brachytherapy 2022;14(6):601–4. [CrossRef]
[Pubmed]

13.
Cetinkaya PG, Aytekin ES, Esenboga S, et al. Eosinophilia in children: Characteristics, etiology and diagnostic algorithm. Eur J Pediatr 2023;182(6):2833–42. [CrossRef]
[Pubmed]

14.
Ge L, Ye F, Mao X, et al. Extramedullary relapse of acute leukemia after allogeneic hematopoietic stem cell transplantation: Different characteristics between acute myelogenous leukemia and acute lymphoblastic leukemia. Biol Blood Marrow Transplant 2014;20(7):1040–7. [CrossRef]
[Pubmed]

15.
Ghosh V, Roy S, Dhamija E, Rathore R, Kumar L, Biswas A. Extramedullary relapse of B cell acute lymphoblastic leukemia in bilateral breasts treated with whole breast radiotherapy. J Adolesc Young Adult Oncol 2023;12(6):942–7. [CrossRef]
[Pubmed]

16.
Ritchie SA, Lee MP, Kao GF, Gaspari AA. An ulceration of the glans penis. Am J Dermatopathol 2015;37(7):544–5, 585. [CrossRef]
[Pubmed]

17.
Michelerio A, Tomasini C, Fiandrino G, et al. Eosinophilic dermatosis of hematologic malignancy in patients with chronic lymphocytic leukemia/nonHodgkin’s B lymphoma: A single center prospective clinico-pathological study. Front Med (Lausanne) 2023;10:1231003. [CrossRef]
[Pubmed]

18.
Abou Chakra R, Najib B, Abdallah W, et al. B-cell lymphoblastic lymphoma in relapse presenting as a uterine mass: A case report and review of literature. Case Rep Oncol 2021;14(2):868–73. [CrossRef]
[Pubmed]

19.
Katsnelson M, Laughingwell R, Rose LJ, Rosenblum NG. Colposcopic and cytologic detection of chronic lymphocytic leukemia in the vagina: A case report. J Reprod Med 2011;56(7–8):344–6.
[Pubmed]

20.
Fielding AK, Richards SM, Chopra R, et al. Outcome of 609 adults after relapse of acute lymphoblastic leukemia (ALL); An MRC UKALL12/ECOG 2993 study. Blood 2007;109(3):944–50. [CrossRef]
[Pubmed]

21.
Kawano N, Maeda T, Kawano S, et al. Uterine relapse of Philadelphia chromosome-negative acute lymphoblastic leukemia. J Clin Exp Hematop 2020;60(3):103–7. [CrossRef]
[Pubmed]

22.
Imanaka R, Katayama Y, Iwato K, et al. Acute leukemia recurring as extramedullary tumors of the ovary following allogeneic hematopoietic stem cell transplantation. [Article in Japanese]. Rinsho Ketsueki 2018;59(1):58–63. [CrossRef]
[Pubmed]

23.
Jacobs JE, Hastings C. Isolated extramedullary relapse in childhood acute lymphocytic leukemia. Curr Hematol Malig Rep 2010;5(4):185–91. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Saadet Akarsu - Conception of the work, Design of the work, Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ahmet Kürşat Poyraz - Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Saadet Akarsu et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.