|
Case Report
Hyperreactio luteinalis with enlargement in the postpartum period: Case report and review of the literature
1 Atrium Health Wake Forest Baptist, Department of Gynecologic Oncology, Medical Center Boulevard, Watlington 4th Floor, Winston Salem, NC 27157, USA
Address correspondence to:
Christine McGough
Medical Center Boulevard, Watlington 4th floor, Winston Salem, NC, 27157,
USA
Message to Corresponding Author
Article ID: 100193Z08CM2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
McGough C, Berry L. Hyperreactio luteinalis with enlargement in the postpartum period: Case report and review of the literature. J Case Rep Images Obstet Gynecol 2024;11(1):17–21.ABSTRACT
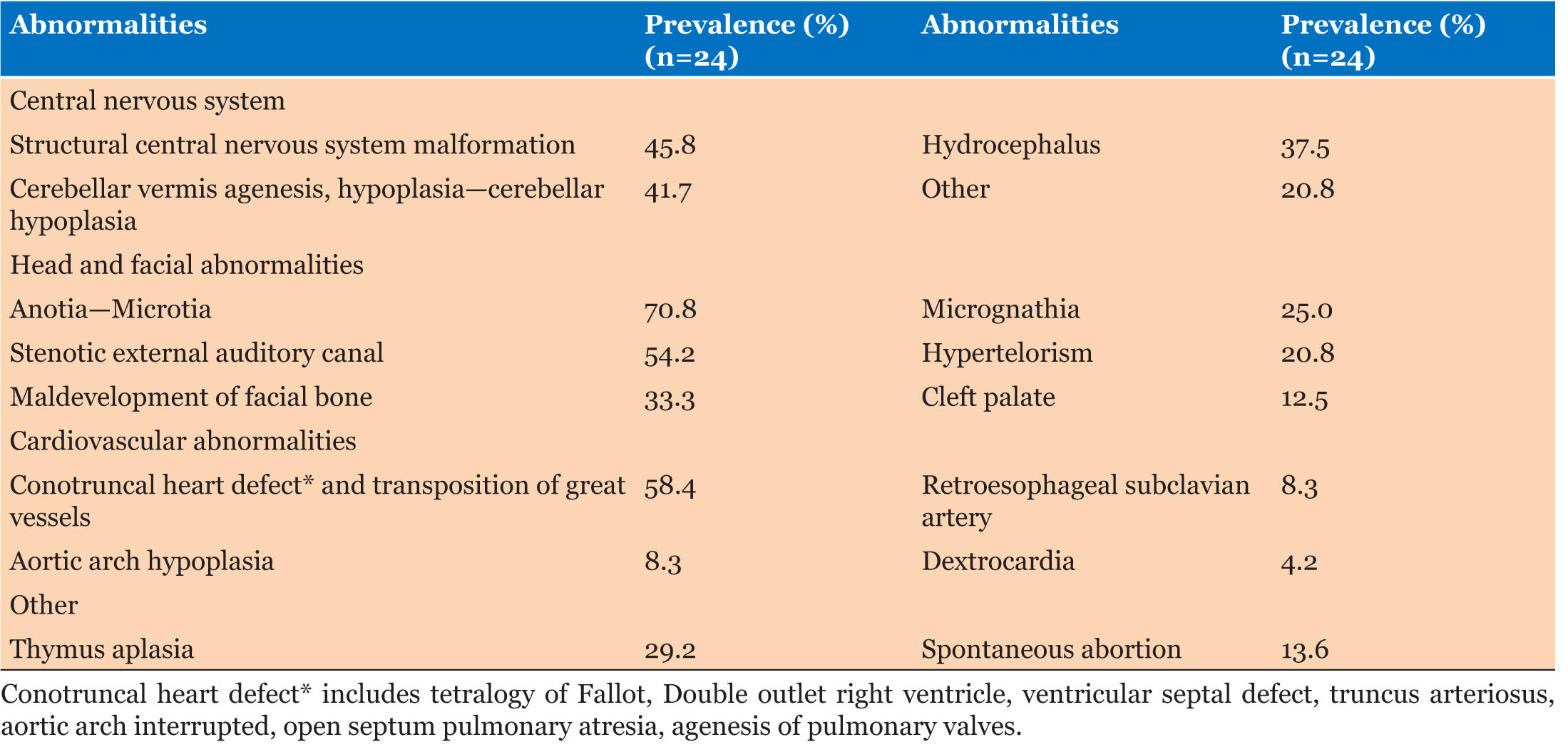
Introduction: Hyperreactio luteinalis (HL) is a rare but recognizable cause of bilateral adnexal masses in pregnancy, with approximately 130 cases reported to date in the literature. It is often associated with virilization, elevated tumor marker levels, extreme elevation in beta hCG, molar gestation, and has been linked to preeclampsia and HELLP. While there are case reports of HL first being diagnosed in the postpartum period, it is most commonly diagnosed during the third trimester or incidentally at time of cesarean section. We present a case of HL diagnosed in pregnancy with enlargement in the postpartum period that ultimately required surgical intervention.
Case Report: A 21-year-old primigravida initially presented with bilateral adnexal masses at 18 weeks gestation, which were followed throughout her pregnancy and thought to reflect HL. She was admitted at 22 weeks with worsening abdominal pain and distention, and ultimately delivered vaginally at 23 weeks due to pre-eclampsia with severe features. She then re-presented six weeks postpartum with worsening abdominal distention and pain with masses filling the abdomen and pelvis. Ultimately due to symptoms she underwent exploratory laparotomy, drainage of adnexal masses, bilateral ovarian cystectomy, and reconstruction. Pathology was consistent with HL. She had an uncomplicated post-operative course and is recovering well.
Conclusion: Hyperreactio luteinalis is a rare diagnosis but should be on the differential for adnexal masses in pregnancy. Further study should be given to normal course of cysts without intervention and surgical or interventional radiology intervention should be considered for significant symptoms.
Keywords: Adnexal mass in pregnancy, Hyperreactio luteinalis, Postpartum
Introduction
Hyperreactio luteinalis (HL) is a rare but recognizable cause of bilateral adnexal masses in pregnancy, with 96 cases reported in the literature between 1995 and 2013 and an additional 35 cases reported between 2014 and 2024 [1],[2],[3]. It can often be associated with virilization (elevated testosterone levels), mildly elevated tumor marker levels, extreme elevation in beta hCG, molar gestation, and has been linked to pre-eclampsia and HELLP in several studies [4]. In one systematic review of 30 studies, they noted a link between high beta hCG levels (>2.5 MoM) to development of pre-eclampsia [5]. Other studies have noted a link between elevated soluble FMS-like tyrosine kinase-1 (sFLT-1)/placental growth factor (PlGF) ratio with development of pre-eclampsia [6].
The exact etiology of HL is unknown, but it is thought to be related to increased sensitivity to beta hCG [4]. Since there are similar structural subunits for beta hCG, follicle stimulating hormone (FSH), and thyroid stimulating hormone (TSH), excessive FSH can mimic the effect of high beta hCG and cause luteal hyperreactivity [7],[8]. Case reports have noted an association of HL with abnormal follicle-stimulating hormone receptor (FSHR)-related genetic testing and recommend FSHR genetic testing in patients with normal thyroid and testosterone levels and recurrent HL [7].
In one study of 96 cases of HL, pre-eclampsia was noted in 24% of cases and fetal growth restriction (FGR) in 12% [2]. Mean gestation at delivery was 35 weeks, with a range of 25–42 weeks [2]. In this case series, surgical treatment was performed in 48% of cases, with oophorectomy in 40% of patients who underwent surgery [2].
We performed a Clavirate literature search from 2008 to 2024 using the key words hyperreactio luteinalis and hypertension or hyperreactio luteinalis and HELLP and found two cases describing HL associated with HELLP, and an additional five cases describing HL associated with hypertensive disorders of pregnancy [3],[4],[9],[10],[11],[12]. In one case of HL associated with HELLP, the patient was delivered at 34 weeks and the cysts resolved spontaneously within three months postpartum [9]. In the other case of HL associated with HELLP, the patient presented with ovarian torsion at 13 weeks and thus underwent laparoscopic right salpingo-oophorectomy [4]. She subsequently developed HELLP syndrome and was delivered at 33 weeks with resolution of her remaining cyst postpartum [4].
While the differential of a pelvic mass in pregnancy should remain broad and should include ruling out neoplasm, HL can be a cause of adnexal masses in pregnancy. The characteristic “spoke-on-a-wheel” appearance on ultrasound is one way to differentiate this diagnosis from other causes of adnexal mass [13]. While there are some case reports of HL first being diagnosed in the postpartum period, it is most commonly found during the third trimester or incidentally at time of caesarean section [14],[15],[16]. There are also several case reports of first and second trimester diagnoses of HL [15]. In our literature review, our case presents the first case of enlarging adnexal masses related to HL after delivery. Yang et al. report a case of HL diagnosed in the postpartum period; however, they do not have any pre-delivery imaging to compare to [17]. In their case, the patient had smaller bilateral cysts (11 and 14 cm) which were managed conservatively with resolution of the cysts three months postpartum [17].
Case Report
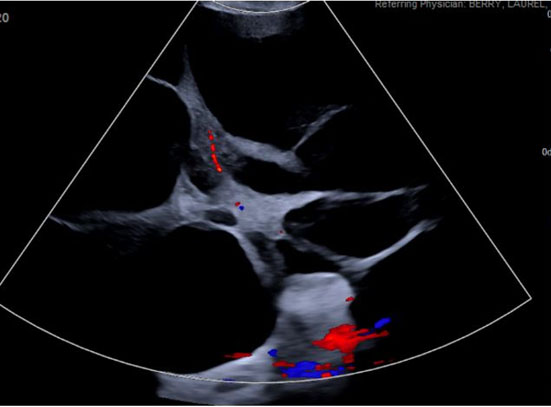
We present the case of a 21-year-old primigravida who was diagnosed with bilateral adnexal masses at 18 weeks gestation on routine ultrasound. She had imaging at 10 weeks with normal appearing bilateral ovaries and no mention of adnexal masses. Her beta hCG at 10 weeks was 93,057 mIU/mL. These masses were followed closely in pregnancy with imaging [ultrasound and magnetic resonance imaging (MRI)] and referral to Gynecologic Oncology at that time. Her diagnosis was thought to be most consistent with hyperreactio luteinalis (HL) and she was managed conservatively. The MRI performed at 20 weeks demonstrated cystic mass measuring 26.6 × 15.5 × 29 cm (Figure 1).
Tumor markers were notable for AFP 74.2 ng/mL with elevation to 359 ng/mL at 20 weeks. hCG was 97,000 mIU/mL at 10 weeks, >250,000 mIU/mL at 20 weeks, 1962 mIU/mL six weeks postpartum, and 1419 mIU/mL seven weeks postpartum. Her CA-125 (cancer antigen 125), testosterone, and estradiol were also elevated at 289 U/mL, 965 ng/dL, and 2288 pg/mL respectively. Her lactate dehydrogenase (LDH), inhibin A and inhibin B were all within normal limits.
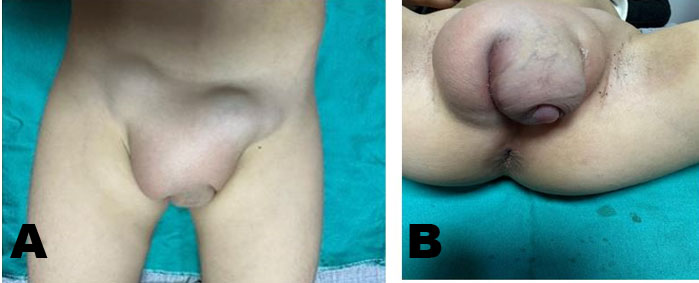
She was admitted at 22 weeks gestation due to worsening abdominal pain and distention, anasarca, and ultimately diagnosed with gestational hypertension, with mildly elevated liver function tests (LFTs) (60–70s). She was largely normotensive with two mild range blood pressures; thus, anti-hypertensive therapy was not initiated. She was counseled on termination versus expectant management and ultimately desired expectant management. She had previously had low risk cell free fetal DNA testing. She declined amniocentesis and consultation with genetic counseling at that time. She was recommended ongoing inpatient admission due to her LFT abnormalities and early gestational age at time of diagnosis of hypertensive disorder of pregnancy. Ultimately, she left against medical advice at 22w6d with plans for close outpatient follow-up.
She was then re-admitted at 23 weeks with worsening pain and distention and elevation in LFTs to the 100s. She received betamethasone during this admission and desired resuscitation of neonate if delivered. She had liver ultrasound with no evidence of cholelithiasis and otherwise no explanation for her transaminitis. Ultimately, she underwent induction of labor due to worsening transaminitis and delivered vaginally at 23w5d with neonatal demise. Karyotype of placental villi notable for 47, XY +16 or trisomy 16, which is associated with uniform fatal fetal outcome in non-mosaic trisomy 16.
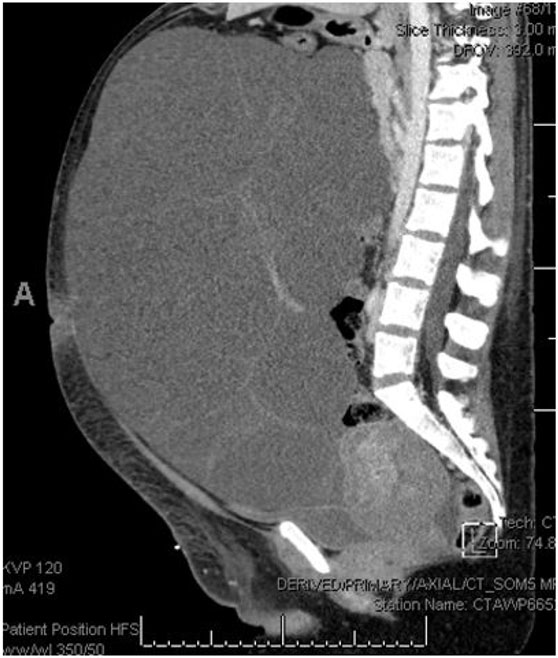
She had an uncomplicated immediate postpartum course and was discharged postpartum day 4 with plan for outpatient follow-up. Serum creatinine was normal at 0.64 mg/dL and urine protein/creatinine ratio was elevated at 0.343. She was discharged with a three day course of Lasix for lower extremity edema but was normotensive at the time of discharge with no anti-hypertensive therapy. Ultimately, she presented to another tertiary hospital emergency department (ED) roughly six weeks postpartum with worsening abdominal pain and abdominal distention. At this encounter, she was again normotensive not on anti-hypertensive therapy with urine protein 20 mg/dL on urinalysis. Transvaginal ultrasound (TVUS) demonstrated large multi-septated cystic mass filling abdomen and pelvis, measuring 35.2 × 31.2 × 16.3 cm (Figure 2). Repeat computed tomography (CT) at that time demonstrated enlargement of bilateral adnexal masses to “large multi-septated cystic mass (vs two large adjacent multi-cystic masses) nearly replacing the entire abdomen and pelvis, measuring 25.1 × 32.1 × 35.7 cm” (Figure 3). She was referred outpatient to Gynecologic Oncology and ultimately underwent exploratory laparotomy, drainage of adnexal masses, bilateral ovarian cystectomy, and reconstruction. She had large, thin walled, multi-septated cysts from both ovaries taking up the entire abdomen and pelvis. Twelve liters of serous appearing fluid was drained intraoperatively and approximately 50–100 cysts were drained individually. Pathology intraoperatively was benign and final pathology demonstrated benign ovarian cysts with features of HL.
Post-operatively she was seen at two and six weeks and was symptomatically improved. She had repeat transvaginal ultrasound three months post-operatively noting resolution of previously visualized adnexal masses. The left ovary showed a 4 cm simple cyst, with otherwise normal-appearing ovaries. She was continued on combined birth control pills to further reduce risk of cyst formation. She will follow with OBGYN for annual exams and was referred to maternal-fetal medicine (MFM) to discuss risks in future pregnancies.
Discussion
We present a case report of a patient with HL in pregnancy with enlargement in the immediate postpartum period. Her pregnancy was also complicated by pre-eclampsia with severe features (transaminitis) which has been linked to HL in other studies. This case is unique in that we have imaging describing enlargement of bilateral adnexal cysts postpartum. Traditionally, since HL is thought to be hormonally driven, the teaching is to expectantly manage with expected resolution in four to six months postpartum [18]. Ovarian suppression with oral contraception can also be used to facilitate resolution [18]. In some cases, such as in our patient, these cysts can be significantly enlarged and can impact quality of life.
More study should be given to the time of natural regression of cysts caused by HL and the expected changes in the postpartum period. In most cases, HL can be managed expectantly; however, surgical intervention or drainage should be considered if patients experience significant pain or symptoms related to their adnexal masses. If considering surgery for suspected HL, ovarian preservation should be attempted if feasible. In our patient’s case, given her significant abdominal distention and pain, as well as enlarging cysts noted six weeks postpartum, the decision was made to proceed with surgery for symptomatic relief as well as for tissue diagnosis. Although interventional radiology (IR) drainage would have been feasible, due to the multi-septated nature of the cysts (approximately 100 septations drained intraoperatively for 12 L of serous fluid), the authors suggest that in this case laparoscopic or IR drainage would not have provided significant symptomatic relief.
Conclusion
Hyperreactio luteinalis is a rare diagnosis but should be on the differential of adnexal masses in pregnancy. It can most typically be managed conservatively with expected resolution of cysts four to six months postpartum. However, with significant symptoms impacting quality of life, or rapid enlargement of cysts, consideration should be given to procedural intervention for relief of symptoms. Further study should be given to risk factors for development of HL, normal course of cysts postpartum without intervention, and when to intervene based on symptoms or size of masses.
REFERENCES
1.
Wang CW, Liu WM, Chen CH. Hyperreactio luteinalis mimicking malignancy during pregnancy with elevated CA-125. Taiwan J Obstet Gynecol 2019;58(6):885–7. [CrossRef]
[Pubmed]

2.
Cavoretto P, Giorgione V, Sigismondi C, et al. Hyperreactio luteinalis: Timely diagnosis minimizes the risk of oophorectomy and alerts clinicians to the associated risk of placental insufficiency. Eur J Obstet Gynecol Reprod Biol 2014;176:10–6. [CrossRef]
[Pubmed]

3.
4.
Grgic O, Radakovic B, Barisic D. Hyperreactio luteinalis could be a risk factor for development of HELLP syndrome: Case report. Fertil Steril 2008;90(5):2008.e13–6. [CrossRef]
[Pubmed]

5.
Skogler J, Moberg T, Tancredi L, et al. Association between human chorionic gonadotropin (hCG) levels and adverse pregnancy outcomes: A systematic review and meta-analysis. Pregnancy Hypertens 2023;34:124–37. [CrossRef]
[Pubmed]

6.
Miyatake R, Fujii T, Kumasawa K, et al. The sFlt-1/PlGF ratio trend is useful in predicting preeclampsia severity in hyperreactio luteinalis complicated with preeclampsia. Case Rep Obstet Gynecol 2023;2023:7352947. [CrossRef]
[Pubmed]

7.
Yang WX, Qin Y, Wang TT, Zhao S. Hyperreactio luteinalis and follicle-stimulating hormone receptor gene activation mutations: A case report. Int J Surg Case Rep 2024;121:109965. [CrossRef]
[Pubmed]

8.
Duan J, Xu P, Zhang H, et al. Mechanism of hormone and allosteric agonist mediated activation of follicle stimulating hormone receptor. Nat Commun 2023;14(1):519. [CrossRef]
[Pubmed]

9.
Haq AN. Hyperreactio luteinalis associated with pregnancy induced hypertension. J Coll Physicians Surg Pak 2010;20(2):137–9.
[Pubmed]

10.
Gatongi DK, Madhvi G, Tydeman G, Hasan A. A case of hyperreactio luteinalis presenting with eclampsia. J Obstet Gynaecol 2006;26(5):465–7. [CrossRef]
[Pubmed]

11.
Gherman RB, Mestman JH, Satin AJ, Goodwin TM. Intractable hyperemesis gravidarum, transient hyperthyroidism and intrauterine growth restriction associated with hyperreactio luteinalis. A case report. J Reprod Med 2003;48(7):553–6.
[Pubmed]

12.
Saisto T, Tiitinen A, Ulander VM, Kaaja R. Clinical cure of severe, early onset preeclampsia with low molecular weight heparin therapy in primigravida with hyperreactio luteinalis and thrombophilia. Hum Reprod 2004;19(3):725–8. [CrossRef]
[Pubmed]

13.
Ghossain MA, Buy JN, Ruiz A, Jacob D, Sciot C, Hugol D, Vadrot D. Hyperreactio luteinalis in a normal pregnancy: Sonographic and MRI findings. J Magn Reson Imaging 1998;8(6):1203–6. [CrossRef]
[Pubmed]

14.
Watkins JC, Lebok P, Young RH. Hyperreactio luteinalis (multiple luteinized follicle cysts): A report of 10 cases. Int J Gynecol Pathol 2021;40(5):427–34. [CrossRef]
[Pubmed]

15.
Edell H, Shearkhani O, Rahmani MR, Kung RC. Incidentally found hyperreactio luteinalis in pregnancy. Radiol Case Rep 2018;13(6):1220–3. [CrossRef]
[Pubmed]

16.
Malinowski AK, Sen J, Sermer M. Hyperreactio luteinalis: Maternal and fetal effects. J Obstet Gynaecol Can 2015;37(8):715–23. [CrossRef]
[Pubmed]

17.
Yang C, Wang H, Zou Y, et al. Hyperreactio luteinalis after delivery: A case report and literature review. Int J Clin Exp Med 2015;8(4):6346–8.
[Pubmed]

18.
Ghassa A, Adwan D, Safadi MF. Hyperreactio luteinalis associated with fetal hyperandrogenism and cystic hygroma. Clin Case Rep 2022;10(9):e6310. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Christine McGough - Conception of the work, Design of the work, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Laurel Berry - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Christine McGough et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.