|
Case Report
STK11 mutation and fallopian tube histology indicating hysterectomy and bilateral salpingo-oophorectomy in a 40-year-old female
1 Department of Pathology at Texas Tech Health Sciences Center, El Paso, TX, USA
Address correspondence to:
Jude Abadie
Department of Pathology at Texas Tech Health Sciences Center, El Paso, TX,
USA
Message to Corresponding Author
Article ID: 100195Z08RG2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Gamez R, Partida A, Abadie J. STK11 mutation and fallopian tube histology indicating hysterectomy and bilateral salpingo-oophorectomy in a 40-year-old female. J Case Rep Images Obstet Gynecol 2025;11(1):29–34.ABSTRACT
Introduction: Metaplastic fallopian tube changes are usually related to inflammation, infection, and/or deleterious STK11 mutations. While these fallopian tube changes have low incidence, they are clinically important to accurately identify premalignant, somatic pathogenesis in patients with Peutz–Jeghers syndrome (PJS).
Case Report: Here we describe a case of a 40-year-old female, with a chronic history of menometrorrhagia, who underwent prophylactic total hysterectomy with bilateral salpingo-oophorectomy (TAH-BSO) due to risk of non-epithelial ovarian cancer associated with a de novo, germline heterozygous STK11 exon 6 deletion. The patient’s father had colorectal carcinoma, her mother had endometrial cancer, and her older sister had breast cancer. The patient’s colonoscopy, screening mammograms, endometrial biopsy, and PAP smears were all negative. Sample morphology was negative for high-grade dysplasia and malignancy; however, prophylactic TAH-BSO was performed due to the patient’s positive carrier status, increased risk for gynecological cancers, and mucinous tumors.
Conclusion: Currently, there are no studies demonstrating how exclusion of secondary findings in PJS supports prophylactic, life-saving management in a subset of patients with strong genetic predisposition and a family history of breast cancer. Hysterectomy is generally considered a last resort after other treatments have failed in cases of heavy menstrual bleeding. As evident in this case of combined PJS and menorrhagia, a prophylactic surgical procedure was clinically indicated.
Keywords: Fallopian tube, Hysterectomy, Peutz–Jeghers syndrome, STK11 mutation, Salpingo-oophorectomy
Introduction
Fallopian tube mucinous metaplasia is an uncommon and underreported finding. However, metaplastic changes are usually secondary, resulting from epithelial trauma. Metaplastic changes in mucinous metaplasia often resemble an endocervical or gastrointestinal (GI) mucinous profile. These profiles involve the distal, fimbriated end of the fallopian tube [1]. Pathologic processes in these anatomic locations are often associated with mutations in STK11 variants on chromosome 19 (19p13.3) and are associated with mucinous fallopian tube pathology [2]. Transitional cell metaplasia is less common and characterized by metaplastic cells that resemble benign transitional (urothelial) bladder cells. Characteristically, this metaplasia involves only distal fimbriae and is often associated with mechanical irritation, inflammation, or infection.
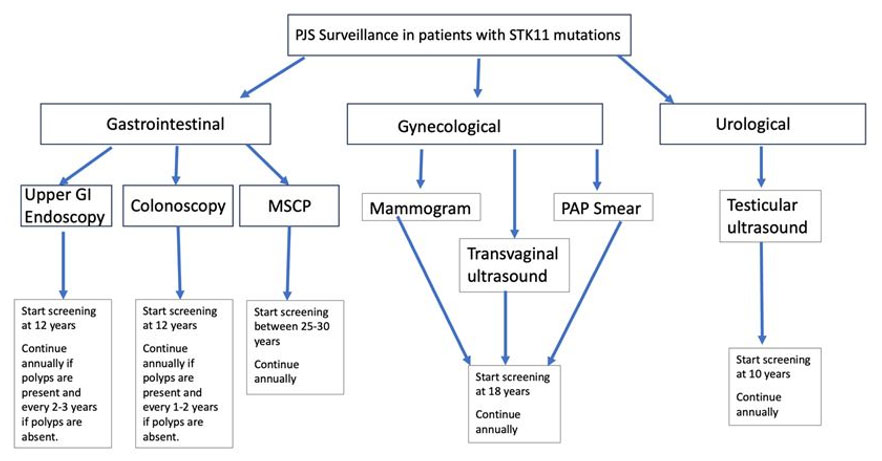
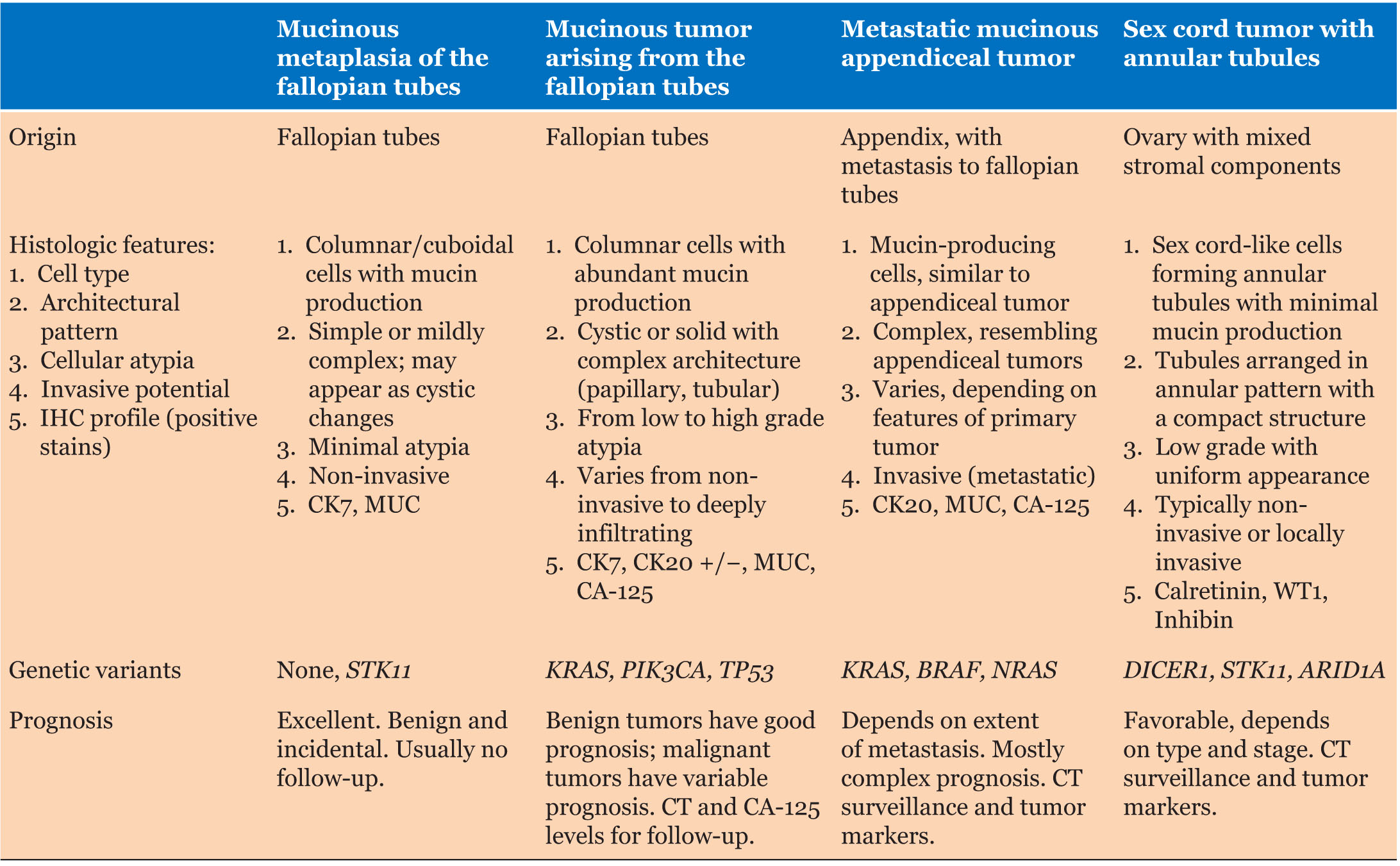
Mucinous changes in fallopian tubes often present diagnostic dilemmas because they can arise from secondary metastatic neoplastic processes, as evident in Peutz–Jeghers syndrome (PJS). These changes can be identified in ovaries, endometrium, appendix, and GI tract mucinous tumors; however, mucinous changes in fallopian tubes can occur without mucinous tumor associations [3]. Figure 1 outlines a surveillance algorithm and recommended clinical management of patients with PJS associated with STK11 pathogenic variants, and Table 1 lists comparisons of histologic features, related genetic variants, and corresponding prognoses in the context of mucinous and metastatic characteristics often associated with fallopian tube, appendiceal, and sex-cord tumors.
Fallopian tube serosal surfaces and fimbriated mucosa, similar to ovaries, are exposed to the peritoneal cavity. This anatomic proximity is prone to metastases via transperitoneal spread. In contrast, non-fimbriated fallopian tube mucosa is less likely to harbor metastases, except in settings of disseminated disease. The colonization of the non-fimbriated tubal mucosa can occur by extension of the tumor from the serosa/fimbrial regions, from sub-mucosal lymphovascular spaces, or from direct deposition of intraperitoneal tumor onto tubal mucosa [2].
Negative findings on clinical examination of fallopian tubes, ovaries, and endometrium, in conjunction with both negative radiology and histology, are critical in excluding primary and secondary neoplastic processes. Gastrointestinal neoplastic origins can be excluded through the use of imaging studies, upper endoscopy, and colonoscopy. However, cytological atypia should raise concern for metastases, and a primary tumor must be excluded. Neoplastic changes can be ruled out by absence of pseudostratification and absence of metaplastic changes in the lamina propria or myosalpinx in conjunction with benign, isolated metaplastic changes at fimbriated ends of fallopian tubes [4].
The average incidence of mucinous metaplasia of fallopian tubes is approximately 0.8% in the general population and is usually identified as an incidental finding [4]. While histologic and cytologic features are usually informative, immunohistochemistry (IHC) may not be helpful in distinguishing metaplasia from metastasis. Therefore, radiological and clinical findings are needed to exclude secondary metastatic processes, such as tubal mucosa infiltration.
Patients with PJS have a significant (approximately 93%) cumulative cancer risk [5],[6]. In patients with PJS undergoing total hysterectomy with bilateral salpingo-oophorectomy (TAH-BSO), 40% are associated with benign isolated mucinous metaplastic changes, 30% with a primary mucinous ovarian carcinoma, 10% with a primary seromucinous ovarian carcinoma, 10% with adenocarcinoma of the appendix, 5% with primary mucinous neo-plasms of fallopian tubes, and 5% associated with endometrial and breast carcinoma [7],[8].
Case Report
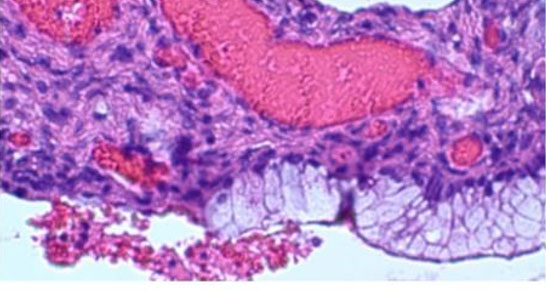
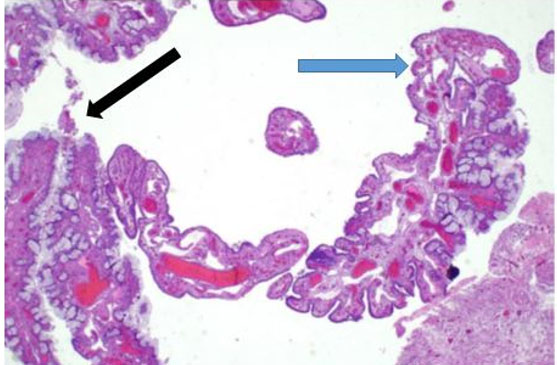
A 40-year-old pre-menopausal female presented with a history of menometrorrhagia characterized by increased uterine bleeding between menstrual cycles. Her medical history was significant for hypertension, type 2 diabetes mellitus, ulcerative colitis, rheumatoid arthritis, and PJS. Past medical procedures included two cesarean sections, urethral stents, and left ovarian cystectomy. Family history revealed paternal colorectal carcinoma, maternal endometrial cancer, and a sister with breast cancer. Histologic evaluation demonstrated abrupt transition between simple columnar ciliated epithelium into GI-epithelium (Figure 2), bland cytologic features without surgical complexity (Figure 3), and ubiquitous transitional areas between ciliated tubal epithelium and abnormal intestinal-like mucinous epithelium (Figure 4).
Exome testing was performed due to the patient’s constellation of disorders and significant family history. This revealed a de novo, heterozygous pathogenic exon 6 deletion in the STK11 tumor suppressor gene, which was diagnostic for PJS. Germline mutations in STK11 typically require a second somatic mutation for the development of PJS. Notably, no pathogenic STK11 variants were identified in patient’s immediate family, and genetic testing for variants linked to breast or colorectal cancer in the proband’s sister and father was negative. The pathogenic STK11 variant in the patient, in conjunction with the family history, raised concerns for underlying genetic predispositions and associated risk for malignancy.
Given the increased risk for gynecological cancers, particularly non-epithelial ovarian cancers (NEOCs), the patient underwent prophylactic TAH-BSO. Non-epithelial ovarian cancers are rare malignancies that include germ cell tumors, sex cord-stromal tumors, small cell carcinomas, and sarcomas. Their etiology remains poorly understood but may overlap with cancer syndromes, such as PJS.
Histologic evaluation following the TAH-BSO demonstrated areas of abrupt transition between normal ciliated tubal epithelium and intestinal-like mucinous epithelium (Figure 2). Although the findings included metaplasia and benign cytologic changes (Figure 3), the mild architectural complexity observed (Figure 4) necessitated further clinical investigation to exclude neoplastic secondary changes. Negative cancer screening included colonoscopy, mammograms, endometrial biopsy, and pap smears. Appendiceal pathology was excluded through imaging studies.
While family risk assessment and counseling are critical, genetic testing had not yet been performed on the patient’s offspring. This case highlights the critical need for comprehensive genetic evaluation in patients with pathogenic STK11 variants. It also underscores the importance of early prophylactic interventions and tailored management strategies to mitigate cancer risks in individuals with genetic predispositions.
Discussion
Peutz–Jeghers syndrome is an autosomal dominant condition characterized by GI hamartomatous polyposis, mucocutaneous macular pigmentation, positive family history, and predisposition to some cancers. The syndrome is caused by mutations in STK11, a tumor suppressor gene located on chromosome at 19p13.3 [9]. STK11 mutations disrupt cell growth regulation and contribute to an increased lifetime risk of developing various malignancies, including GI, pancreatic, breast, gynecologic, and testicular cancers [10].
Tumors of the genital tract may be infrequent in PJS; however, gastric-type endocervical adenocarcinoma is often present in affected females [9]. Furthermore, there is an increased risk of other gynecological cancers, including ovarian sex cord tumors, specifically sex cord tumors with annular tubules (SCTAT), and mucinous tumors involving the ovaries and fallopian tubes. For males with PJS, clinical sequlae include testicular Sertoli cell tumors that often secrete estrogen. Clinical presentations in males include gynecomastia, advanced bone age, and short stature. Continued follow-up is clinically indicated in both sexes due to increased risk for malignancies.
While most high-grade serous carcinomas (HGSCs) originate from fallopian tubes, the frequency of abnormal fallopian tube findings has been largely unknown in cases of ovarian tumors. Abnormal fallopian tube findings are identified in about 80% of HGSCs and in 83% of sero-mucinous carcinomas [3]. Mucinous metaplasia of the fallopian tubes is a rare, benign condition that is frequently underreported. This metaplasia, often resulting from epithelial insults, is the most commonly underreported subtype. It is characterized by metaplastic cells that resemble endocervical or GI mucinous epithelium. The distal, fimbriated end of the fallopian tube is typically affected, and cases are associated with STK11 mutations [4].
The second subtype is transitional cell metaplasia, where metaplastic cells resemble benign transitional (urothelial) cells of the urinary bladder. This subtype is confined to the distal fimbriae and is linked to mechanical irritation, inflammation, or infection.
The serosal surface of fallopian tubes and the fimbriated mucosa are also exposed to the peritoneal cavity, making them susceptible to metastases via transperitoneal spread. Non-fimbriated mucosa may have metastases through direct extension from the fimbriated end, submucosal lymphovascular spaces, or intraperitoneal tumor deposition [8].
Mucinous changes in the fallopian tubes can present diagnostic challenges due to secondary metastatic neoplasms that may originate from ovary, endometrium, appendix, or GI tract. However, mucinous changes can also occur independently of mucinous tumors. Prophylactic hysterectomy with bilateral salpingo-oophorectomy is often recommended for women with PJS. Indications include cancer risks of gynecologic malignancies necessitating removal of uterus, fallopian tubes, and ovaries [3],[10]. Challenges exist with surveillance (e.g., tumor marker assessment) because routine screening for gynecologic cancers may not reliably detect malignancies in early stages of PJS, especially during asymptomatic periods [10]. Sex cord tumors with annular tubules risk can be mitigated in early PJS presenters due to increased hormone production precipitating menstrual irregularities or precocious puberty. Prophylactic surgery reduces the risk of developing these rare but concerning tumors [5],[11],[12].
Timing of prophylactic surgery should be individualized based on the patient’s age, family history, and reproductive plans. Fertility preservation options, such as egg or embryo freezing, should be discussed with women of reproductive age before surgery. Multidisciplinary care involving genetic counselors, gynecologic oncologists, and reproductive specialists is essential to optimize outcomes and address patient preferences.
Our patient’s comprehensive evaluations (microscopic, radiographic, and clinical) of tubes, ovaries, and endometrium ruled out both primary and secondary neoplastic processes, including those originating from the appendix or GI tract. In cases involving a prevalent STK11 mutation and previous malignancies or concurrent pelvic/GI masses, it is imperative to consider tubal mucosal metastasis, highlighting the need for ongoing vigilance and comprehensive care for this patient population. Because IHC cannot differentiate metaplasia from metastasis, radiological and clinical evaluations are essential for ruling out secondary processes. Incorporating these considerations into the management of patients with PJS emphasizes the importance of early genetic testing, individualized cancer risk assessments, and risk reduction strategies.
Conclusion
To support life-saving management, it is essential to exclude secondary findings (e.g., gynecological neoplastic processes) in patients with PJS with or without a family history of breast cancer. Given our patient’s PJS and increased risk for gynecologic cancers, including ovarian SCTAT and mucinous tumors, a prophylactic total hysterectomy with TAH-BSO was performed. To better manage associated risks, this approach may be recommended for similar cases after child-bearing is completed. Due to the high risk for cancer in patients with incidental findings of mucinous metaplasia, genetic counseling is also recommended to rule-out PJS, as well as subsequent adherence to screening guidelines. Conversely, in cases of heavy menstrual bleeding, hysterectomy is generally considered a last resort after other treatments have failed. As demonstrated in this case of PJS and menorrhagia, a prophylactic surgical procedure was warranted.
REFERENCES
1.
Seidman JD. Mucinous lesions of the fallopian tube. A report of seven cases. Am J Surg Pathol 1994;18(12):1205–12. [CrossRef]
[Pubmed]

2.
Karpathiou G, Chauleur C, Venet M, Clemenson A, Peoc'h M. Pathology of the fallopian tube: Tubal involvement by ovarian tumors and incidental findings in the nontumoral setting. Pathobiology 2020;87(1):37–44. [CrossRef]
[Pubmed]

3.
Giardiello FM, Brensinger JD, Tersmette AC, et al. Very high risk of cancer in familial Peutz-Jeghers syndrome. Gastroenterology 2000;119(6):1447–53. [CrossRef]
[Pubmed]

4.
Wong AK, Seidman JD, Barbuto DA, McPhaul LW, Silva EG. Mucinous metaplasia of the fallopian tube: A diagnostic pitfall mimicking metastasis. Int J Gynecol Pathol 2011;30(1):36–40. [CrossRef]
[Pubmed]

5.
Wu M, Krishnamurthy K. Peutz-Jeghers Syndrome. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025.
[Pubmed]

6.
Mayor S. NICE says hysterectomy must be last option for heavy menstrual bleeding. BMJ 2007;334(7586):175. [CrossRef]
[Pubmed]

7.
van der Meij E, Emanuel MH. Hysterectomy for heavy menstrual bleeding. Womens Health (Lond) 2016;12(1):63–9. [CrossRef]
[Pubmed]

8.
Bronte Anaut M, Arredondo Montero J, Fernández Seara MP, Guarch Troyas R. Gastric-phenotype mucinous carcinoma of the fallopian tube with secondary ovarian involvement in a woman with Peutz-Jeghers syndrome: A case report. Int J Surg Pathol 2023;31(1):92–7. [CrossRef]
[Pubmed]

9.
Zhou Y, Wang X, Li Y, et al. When synchronous mucinous metaplasia and neoplasia of the female genital tract and Peutz-Jeghers syndrome meet: A case report and literature reviews. BMC Womens Health 2024;24(1):375. [CrossRef]
[Pubmed]

10.
Hearle N, Schumacher V, Menko FH, et al. Frequency and spectrum of cancers in the Peutz-Jeghers syndrome. Clin Cancer Res 2006;12(10):3209–15. [CrossRef]
[Pubmed]

11.
Gordhandas SB, Kahn R, Sassine D, et al. Gastric-type adenocarcinoma of the cervix in patients with Peutz-Jeghers syndrome: A systematic review of the literature with proposed screening guidelines. Int J Gynecol Cancer 2022;32(1):79–88. [CrossRef]
[Pubmed]

12.
Zhou F, Lv B, Dong L, Wan F, Qin J, Huang L. Multiple genital tract tumors and mucinous adenocarcinoma of colon in a woman with Peutz-Jeghers syndrome: A case report and review of literatures. Int J Clin Exp Pathol 2014;7(7):4448–53.
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Roberto Gamez - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Alejandro Partida - Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Jude Abadie - Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Roberto Gamez et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.