|
Case Report
Rectus sheath hematoma in pregnancy following low molecular weight heparin administration
1 The Obstetrics and Gynecology Division, Soroka University Medical Center, Ben-Gurion University of the Negev, POB 151, Beer-Sheva, IL, Israel
Address correspondence to:
Sivan Chocron
The Obstetrics and Gynecology Division, Soroka University Medical Center, Ben-Gurion University of the Negev, POB 151, Beer-Sheva, IL,
Israel
Message to Corresponding Author
Article ID: 100207Z08SC2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Chocron S, Zamstein O, Segal D, Bashiri A. Rectus sheath hematoma in pregnancy following low molecular weight heparin administration. J Case Rep Images Obstet Gynecol 2025;11(1):92–96.ABSTRACT
Introduction: This case report examines a rare complication of low molecular weight (LMW) heparin use during pregnancy. It details the presentation, diagnosis, treatment, and follow-up of rectus sheath hematoma (RSH) associated with enoxaparin prophylaxis.
Case Report: A 35-year-old woman with a history of recurrent pregnancy loss and methylenetetrahydrofolate reductase deficiency (MTHFR) mutation was on enoxaparin for venous thromboembolism (VTE) prevention. At 37 weeks gestation, she presented with right abdominal pain and ecchymosis near the injection site, which was diagnosed as RSH. Initially, no further diagnostic tests were conducted. Two days later, she returned with worsening pain, significant hemoglobin drop, and required a blood transfusion. Non-stress testing showed inconclusive fetal heart rate monitoring, prompting an induction of labor. Due to a non-vertex lie, an emergency cesarean section was performed. In the cesarean section extensive blood clots were seen with a dark-colored rectus muscle. The decision was made to leave the clot in place to minimize further bleeding. Postoperatively, she received additional blood and antibiotics, and LMW heparin was stopped and pneumatic stockings for VTE prophylaxis were used. The patient recovered well and was discharged on postoperative day 3. A follow-up ultrasound showed a resolving hematoma at three days, one month, and three months postpartum.
Conclusion: This case highlights rectus sheath hematoma as a rare but serious complication of LMW heparin use during pregnancy. It underscores the need for careful monitoring and risk-benefit assessment of anticoagulant therapy in pregnant patients. Further research is needed to better understand the indications and risk factors for LMW heparin use during pregnancy.
Keywords: Anticoagulant therapy, Low molecular weight heparin, Pregnancy complications, Rectus sheath hematoma
Introduction
Pregnant patients have a fourfold to fivefold increased risk of thromboembolism compared with non-pregnant patients [1]. Subcutaneous administration of LMW heparin is first-line option for VTE prophylaxis among high-risk patients during pregnancy [2].
While the administration of LMW heparin is effective, it can lead to complications such as hematoma, bruising, and pain at the injection site [3]. Additionally, a rare but significant complication is rectus sheath hematoma (RSH). This paper discusses a case where a patient developed RSH after receiving LMW heparin during pregnancy [4].
Case Report
A 35-year-old gravida 9 para 6 Bedouin patient, at 37+2 gestation age, was admitted to the obstetric emergency department of our tertiary care center. She presented with right abdominal pain near the site of LMW heparin injection. Reviewing her obstetrical history, the patient had experienced six uncomplicated spontaneous vaginal deliveries at term and two first-trimester pregnancy losses. Following these losses, a comprehensive evaluation revealed that she was a carrier of the MTHFR mutation, a gene promoter hypermethylation controversially associated with recurrent pregnancy loss (RPL). Although not a standard indication, she was prescribed daily subcutaneous LMW heparin 40 mg daily, starting from her first obstetrical check-up at week 9, based on a previous successful pregnancy with this regimen. Apart from this, her pregnancy follow-up and medical history were unremarkable. The patient was under good checkup including biochemical screen and fetal anatomy screen.
Upon admission, shallow ecchymoses were noted on the middle right abdomen near the injection site. Laboratory tests were not conducted. The patient received analgesic medication and was subsequently discharged with instructions to rest at home. The patient returned to the emergency department two days after the initial presentation at 37+4 weeks, complaining of persistent pain near the injection site. Although she was hemodynamically stable, blood tests revealed a significant decrease in hemoglobin (7.9 g/dL) and hematocrit (24.6%) levels compared to her previous results from two months earlier, which were 12.8 g/dL and 36.9%, respectively. Fetal evaluation with non-stress testing (NST) showed an intermittent non-reassuring fetal heart rate. Due to the transverse lie of the fetus, an urgent cesarean delivery (CD) was planned. In preparation for the CD, and given her relatively low hemoglobin levels, a blood transfusion was started prior to the procedure.
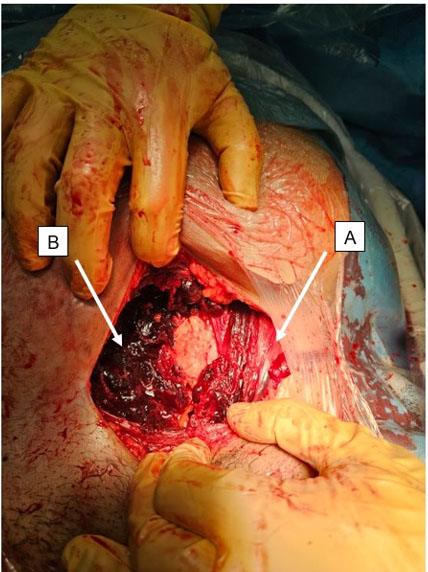
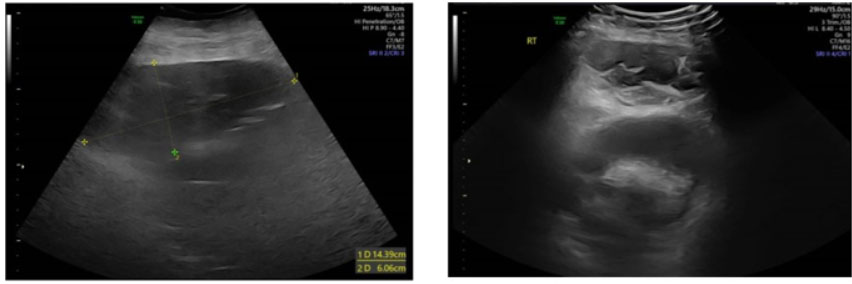
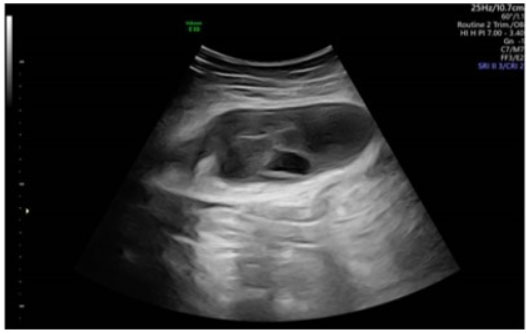
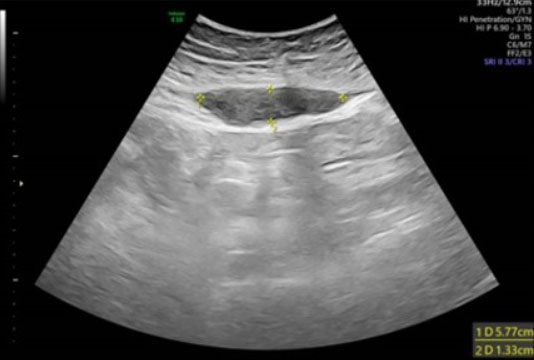
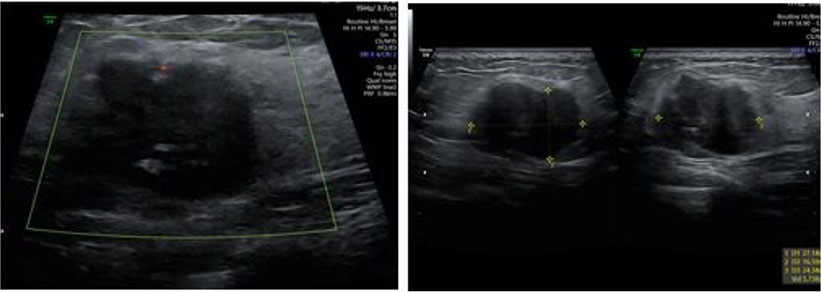
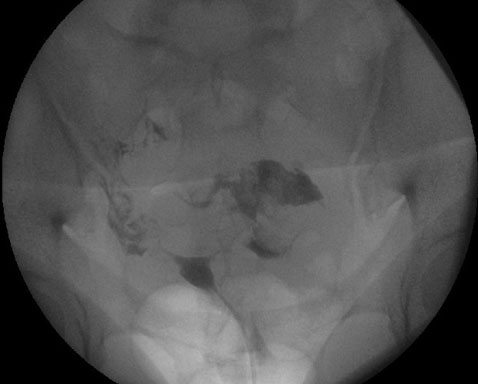
During the cesarean delivery, after dissecting the subcutaneous tissues, the abdominal fascia was found to be unusually weak and could be easily dissected with blunt fingers. Beneath the fascia, a significant amount of blood clots was discovered, while the right rectus muscle appeared dark purple-black with clot-adherent areas (Figure 1). No major source of bleeding was identified. After delivering the fetus and closing the uterus, a review of the abdominal cavity revealed no other hematomas. Due to the absence of active bleeding and the hemodynamically stable condition of the patient, a decision was made to leave the blood-infiltrated right rectus muscle and adherent clots in place and proceed with the standard closure of the abdominal wall layers. To minimize the risk of hematoma infection, the patient received prophylactic antibiotics (amoxicillin) for 48 hours. Given the increased postpartum risk for VTE (due to parity, maternal age, and surgery) and the presence of increased bleeding risk, pneumatic stockings were used during the postpartum hospitalization instead of LMW heparin. The patient’s postoperative course was unremarkable, with stable vital signs, no fever, minimal pain requiring minimal medication, free ambulation, and stable blood test and imaging results. She was discharged home on postoperative day 3 in good condition, per standard protocol. A follow-up ultrasound at the day of discharge, one month and three months postoperatively revealed a smaller hematoma (Figure 2, Figure 3, Figure 4). Throughout the puerperium period, she experienced no fever or hospital readmission.
The patient returned for follow-up approximately one year after delivery, presenting a new pregnancy at six weeks’ gestation. A viable intrauterine pregnancy with fetal cardiac activity was confirmed on ultrasound. She was referred to further evaluation, including measurement of homocysteine levels, to assess the need for prophylactic LMW heparin therapy.
Discussion
Pregnancy is associated with a four- to fivefold increased risk of VTE, which may be further exacerbated by factors such as a personal history of VTE, inherited thrombophilia, multiparity, sepsis, and surgery. To mitigate this risk, prophylactic administration of LMW heparin is often employed [5]. However, this case underscores the potential complications associated with LMW heparin use during pregnancy. The decision to initiate prophylactic anticoagulation must therefore weigh the benefits of VTE prevention against the risks of adverse effects [6].
Complication risk increases in patients receiving higher doses of LMW heparin, those with renal impairment (as approximately 40% of LMW heparin is renally excreted), and in cases involving technical issues during administration [7]. Pregnant women are particularly susceptible due to abdominal wall stretching, with contributing factors including multiparity, chronic coughing, minor abdominal trauma, and advanced maternal age [8]. Anticoagulation, including LMW heparin even at prophylactic doses [9], is a well-documented precipitating factor.
One such complication is RSH, a rare but serious cause of acute abdominal pain in pregnancy [10]. Rectus sheath hematoma results from bleeding into the rectus abdominis muscle sheath, typically due to injury of the superior or inferior epigastric vessels or direct muscle tears
Rectus sheath hematoma often mimics other obstetric emergencies—such as uterine rupture, placental abruption, or appendicitis—which can lead to diagnostic confusion [10]. Such misdiagnosis may delay appropriate treatment and has been associated with unnecessary laparotomies, emergency cesarean deliveries, preterm birth, and increased perinatal morbidity and mortality [11].
Similar cases in the literature reinforce the clinical relevance of RSH as a complication of LMW heparin use in pregnancy. Anwari described a 32-year-old woman at 33 weeks’ gestation who developed a large RSH following therapeutic enoxaparin for pulmonary embolism. The hematoma was initially misdiagnosed as placental abruption, leading to an emergency cesarean delivery. Intraoperatively, a 2-liter rectus sheath hematoma was discovered, underscoring the diagnostic challenges and potential for unnecessary delivery [12]. Similarly, Azharuddin et al. reported a series of five patients who developed RSH after enoxaparin injections, one of which was fatal [9].
Clinically, RSH commonly presents with abdominal pain (84–97%), a palpable abdominal wall mass (63–92%), and localized tenderness (71%). Diagnosis is typically confirmed via imaging, with ultrasonography as the first-line modality. However, computed tomography (CT) offers superior sensitivity and specificity [7]. Laboratory tests may reveal a drop in hematocrit, which can compromise fetal circulation and result in bradycardia [13].
Management and prevention
The acute management of RSH during pregnancy, particularly when associated with LMW heparin, depends primarily on maternal hemodynamic stability and fetal status. In most stable cases, conservative treatment—including discontinuation of LMW heparin, bed rest, and analgesia—is sufficient and safe. Permuy et al. (2020) described a 32-week pregnant patient successfully managed expectantly, allowing pregnancy continuation to term [14]. In cases of ongoing hemorrhage or hemodynamic compromise, interventional radiology with selective arterial embolization has proven effective, as highlighted by Clark et al. (2007), who reported high success rates in anticoagulated patients [15]. Surgical intervention, such as hematoma evacuation and vessel ligation, should be reserved for cases where embolization is unavailable or unsuccessful, as demonstrated in a case by Eckhoff et al. (2016) [16]. In addition to definitive management, protamine sulfate may be administered in severe cases, such as in hemodynamically unstable patients or those with expanding rectus sheath hematomas, to partially reverse the anticoagulant effect of low-molecular-weight heparin (LMWH), neutralizing approximately 60–80% of its activity. However, its use remains uncommon, as LMWH is more difficult to reverse promptly due to its subcutaneous administration and longer biological half-life [17].
Preventive strategies focus on minimizing injection-related trauma and optimizing anticoagulation management. Proper subcutaneous LMW heparin administration technique—ensuring subcutaneous injection and rotating sites—plays a crucial role in risk reduction. The Swedish National Guidelines for Obstetric Thromboprophylaxis recommend using the thigh as an injection site in the immediate postpartum period to prevent abdominal wall hematomas [18]. Post-RSH, close clinical and ultrasound monitoring is warranted, particularly if anticoagulation must be resumed. Reassessment of anticoagulant dosing given the potential for subtherapeutic or excessive anticoagulation, the question arises whether anti-Xa-guided dose adjustment could help optimize LMW heparin prophylaxis and reduce complications. However, major guidelines do not recommend routine anti-Xa monitoring during pregnancy or postpartum. American College of Obstetricians and Gynecologists (ACOG), American Society of Hematology (ASH), and Royal College of Obstetricians and Gynecologists (RCOG) all favor weight-based dosing without regular anti-Xa testing, citing insufficient evidence of clinical benefit [1],[4],[19]. Other factors such as renal function and patient-specific risk factors are essential. Patient education regarding early warning signs is also critical to facilitate timely recognition and intervention in recurrent cases.
When appropriately managed, RSH in pregnancy is associated with favorable long-term outcomes, with most cases, including ours, showing complete recovery without infection, recurrence, or thrombotic events.
Conclusion
This case underscores the critical need to carefully evaluate the risks associated with LMW heparin use during pregnancy and to implement effective management strategies for complications such as RSH. In this instance, opting to leave the hematoma in place during surgery proved beneficial in preventing further bleeding, given the patient’s stable condition. Continuous reassessment of LMW heparin therapy and its indications are essential to minimize potential adverse effects and to ensure optimal outcomes for both the mother and fetus.
REFERENCES
1.
American College of Obstetricians and Gynecologists’ committee on practice bulletins—obstetrics. ACOG practice bulletin No. 196: Thromboembolism in pregnancy. Obstet Gynecol 2018;132(1):e1–17. [CrossRef]
[Pubmed]

2.
Dean RE, Maniam G, Vo T. Rectus sheath hematoma following enoxaparin administration. Proc (Bayl Univ Med Cent) 2020;33(3):460–1. [CrossRef]
[Pubmed]

3.
Li Y, Dong S, Wang P, Sun J, Jiang H, Liu F. Influence of low-molecular-weight heparin injection sites on local bruising and pain: A systematic review and meta-analysis. J Clin Pharm Ther 2021;46(3):688–97. [CrossRef]
[Pubmed]

4.
Bates SM, Rajasekhar A, Middeldorp S, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: Venous thromboembolism in the context of pregnancy. Blood Adv 2018;2(22):3317–59. [CrossRef]
[Pubmed]

5.
Greer IA, Nelson-Piercy C. Low-molecular-weight heparins for thromboprophylaxis and treatment of venous thromboembolism in pregnancy: A systematic review of safety and efficacy. Blood 2005;106(2):401–7. [CrossRef]
[Pubmed]

6.
Lutfi A, O'Rourke E, Crowley M, et al. VTE risk assessment, prevention and diagnosis in pregnancy. Thromb Res 2024;235:164–74. [CrossRef]
[Pubmed]

7.
Takahashi K, Nihei T, Aoki Y, et al. Spontaneous rectus sheath hematoma associated with warfarin administration: A case report. J Rural Med 2019;14(2):245–8. [CrossRef]
[Pubmed]

8.
Mendes D, Penedones A, Martins M, Cavadas S, Alves C, Batel-Marques F. Rectus sheath hematoma in patients receiving subcutaneous enoxaparin: A case series of five patients. Clin Case Rep 2020;8(12):3432–9. [CrossRef]
[Pubmed]

9.
Azharuddin M, Gupta M, Maniar M. Subcutaneous heparin leads to rectus sheath hematoma: A rare complication. Cureus 2018;10(6):e2769. [CrossRef]
[Pubmed]

10.
Zaidi B, Gazzah W, Saad MB, Sindi S, Maraach W, Mensi Z. Spontaneous rectus sheath hematoma revealed by abdominal pain during pregnancy: A case report. Int J Surg Case Rep 2024;118:109695. [CrossRef]
[Pubmed]

11.
Sufficool MM, Sheikh IB, Shapiro RE, Dueñas-Garcia OF. Post-partum rectus sheath hematoma complication: Case report. AME Case Rep 2021;5:16. [CrossRef]
[Pubmed]

12.
Anwari L. Rectus sheath hematoma in pregnancy: A case report. Radiol Case Rep 2020;15(10):2022–5. [CrossRef]
[Pubmed]

13.
Thornburg KL, Louey S. Uteroplacental circulation and fetal vascular function and development. Curr Vasc Pharmacol 2013;11(5):748–57. [CrossRef]
[Pubmed]

14.
15.
16.
Eckhoff K, Wedel T, Both M, Bas K, Maass N, Alkatout I. Spontaneous rectus sheath hematoma in pregnancy and a systematic anatomical workup of rectus sheath hematoma: A case report. J Med Case Rep 2016;10(1):292. [CrossRef]
[Pubmed]

17.
Holmes SJ, Yale SH, Mazza JJ. Rectus sheath hematoma as a cause of acute abdominal pain. Am Fam Physician 2001;64(10):1681–2.
[Pubmed]

18.
SUPPORTING INFORMATION
Acknowledgments
We would like to extend our sincere gratitude to the Ultrasound Unit at the Obstetrics and Gynecology Division, Soroka University Medical Center for their invaluable support in the management and follow-up of our patient. Their expertise and dedication were crucial to the success of this study. Special thanks to Dr. Joel Baron and Dr. Dan Tirosh for their assistance and commitment throughout the process. All clinical and imaging data, including the ultrasound images presented in this case report, were obtained at Soroka University Medical Center, Obstetrics and Gynecology Division.
Author ContributionsSivan Chocron - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Omri Zamstein - Conception of the work, Design of the work, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
David Segal - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Asher Bashiri - Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Sivan Chocron et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.