|
Case Report
Moyamoya disease diagnosed during pregnancy
1 Resident, SLUCare/SSM Health, St. Louis, MO, USA
2 Professor, Division of Maternal Fetal Medicine, SLUCare/SSM Health, St. Louis, MO, USA
3 Assistant Professor, Division of Maternal Fetal Medicine, SLUCare/SSM Health, St. Louis, MO, USA
Address correspondence to:
Olivia Wilson
MD, 6420 Clayton Rd, 2nd Floor, Richmond Heights, MO 63117,
USA
Message to Corresponding Author
Article ID: 100213Z08OW2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Wilson O, Gross G, Buchanan C. Moyamoya disease diagnosed during pregnancy. J Case Rep Images Obstet Gynecol 2025;11(2):23–27.ABSTRACT
Introduction: Moyamoya is a rare cerebrovascular disease in which the terminal, intracranial portions of the carotid arteries are progressively narrowed, resulting in the development of a tenuous network of small, collateral vessels. The exact etiology is unknown; however, these patients are at greater risk for both ischemic and hemorrhagic strokes. The risk of stroke in patients with Moyamoya disease increases during pregnancy, likely due to changes in vascular dynamics and other pregnancy-related physiological changes. Most cases of Moyamoya in pregnancy describe patient’s diagnosed preconception. The handful of case reports of moyamoya diagnosed in pregnancy have guarded outcomes.
Case Report: This case report describes the pregnancy of a patient in her 30s who was diagnosed with Moyamoya at 13 weeks of gestation. She suffered three ischemic strokes prior to iatrogenic preterm delivery at 27 weeks and 5 days of pregnancy. Postpartum, she had a massive left hemispheric infarct leading to her demise.
Conclusion: The clinical course and outcome for this patient suggests that Moyamoya disease without evidence of medical or surgical control may be a contraindication to pregnancy.
Keywords: Moyamoya, Postpartum, Pregnancy, Stroke
Introduction
Moyamoya is Japanese for “puff of smoke” [1]. It is a rare cerebrovascular disease in which the terminal, intracranial portion of the internal carotid arteries is progressively narrowed with concomitant development of a tenuous network of small and fragile collateral vessels. The appearance of these collateral vessels on radiographic imaging is what gives the appearance of the disease’s Japanese namesake. As the intracranial blood flow grows dependent on more brittle, small caliber vessels, patients are at greater risk for both ischemic and hemorrhagic strokes. This is an idiopathic disease primarily affecting individuals of East Asian descent. It is believed to have a genetic component, with some families showing a higher incidence of the disease. In Japan, the incidence was reported to be 0.35–0.54 per 100,000 compared to the United States where the incidence was about 0.086 per 100,000 [1]. The age of onset has a bimodal distribution at approximately 10 and 40 years old. Moyamoya affects female patients over males in an 18:1 ratio. Presentation is typically with neurologic symptoms and on occasion fatal stroke. Other disease processes such as severe sickle cell disease may mimic Moyamoya, resulting in Moyamoya syndrome [1]. Understanding of the pathophysiology of true Moyamoya disease is still evolving. However, there is evidence that it is a mixed disease process including both aberrant angiogenesis and vascular wall thickening.
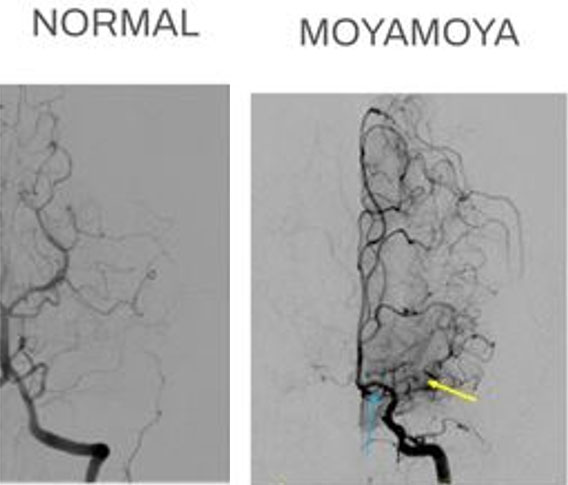
Moyamoya is typically discovered following new-onset stroke or neurologic symptoms. Diagnosis is typically made radiographically [2]. The classic appearance is best visualized on cerebral angiography with the stenosed internal carotid artery turning into the middle cerebral artery amongst a cloudy network of collaterals (Figure 1). Moyamoya is staged radiographically, with the primary focus on the degree of stenosis of the internal carotid arteries at the juncture of the middle cerebral arteries. Disease stage does not always correspond to degree of collateral vessel development [2].
Prognosis for Moyamoya disease is poor. Conservative management includes blood pressure control and antiplatelet therapy [1],[3]. There is some suggestion that calcium channel blockers may be beneficial [2]. However, nearly all cases demonstrate radiographic progression within five years [2]. This process seems to be worse in the pediatric population. The gold-standard treatment for the disease is surgical revascularization which can be completed by direct bypass or with indirect grafts. However, it is not curative. This procedure itself is associated with significant morbid, including a high risk of perioperative stroke and risk of hyperperfusion syndrome. This confers a reduced life expectancy for most patients with Moyamoya as sequela of major stroke events.
Case Report
Patient X is a 35 year old G3P0111 who was initially presented at 13w3d for dizziness, weakness, and aphasia. Her past medical history was significant for chronic hypertension, type 2 diabetes mellitus (T2DM), severe preeclampsia in a prior pregnancy, advanced maternal age, anxiety, and obesity. Her past surgical history included one prior cesarean section and an endometrial ablation. Her family and social history were non-contributory. On presentation, her vital signs were significant for hypertension to 163/109. Her exam was notable for confusion and word-finding difficulties including inability to name common. She had no motor or sensory deficits.
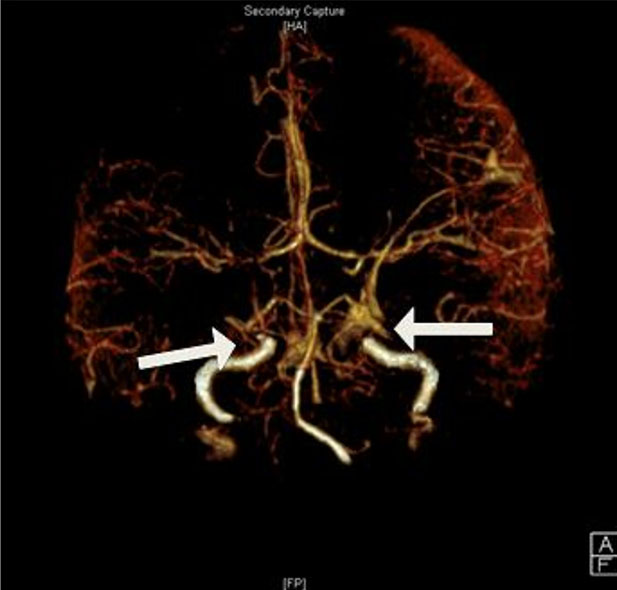
This was her first admission. Computed tomography (CT) angiogram and magnetic resonance imaging (MRI) of the brain demonstrated multifocal vessel stenosis and watershed infarct of the right middle cerebral artery territory. Differential diagnosis was at right cerebral vasospasm versus thrombosis versus posterior reversible encephalopathy syndrome (PRES). The patient was admitted for observation and further workup. Her blood pressure goal was set at <160 systolic and <100 diastolic, and she was started on nifedipine 30 mg daily. On hospital day 2, she underwent MR angiography (MRA) which showed worsening carotid artery stenosis (Figure 2) and was thus transferred to the neurological intensive care unit. On day 3, moyamoya disease was first considered as a possible etiology and hence and interval MRA was performed. The patient subsequently left the hospital against medical advice.
Three weeks later she presented for her first prenatal care visit. During this interim period, she had appointments with neurology, neurosurgery, and a maternal-fetal medicine (MFM) practice unaffiliated with our academic center. Due to the extent of her disease, these specialists recommended pregnancy termination and surgical revascularization which she declined. A multidisciplinary team approach to her care ensued that included antiplatelet therapy and permissive hypertension along with early and careful delivery planning. She was followed closely outpatient.
Her next admission at 18w6d was for abdominal pain and glycemic control in the setting of poorly controlled T2DM. Labs were negative for diabetic ketoacidosis and insulin was initiated and adjusted to optimize control. Her previous outpatient logs were too sparse to safely initiate insulin. Her antihypertensive was changed to amlodipine and she received an anesthesia consult to assess her future delivery. The abdominal pain was attributed to a resolving ruptured ovarian cyst and the patient was discharged.
She presented a week later to an outside hospital for altered mental status and an MRI showing bilateral multifocal infarcts with some present at the watershed line. Her amlodipine was discontinued and she was sent home. She saw a second Neurosurgeon outpatient who again counseled her on the risk of continuing pregnancy. Plan was for repeat angiography and surgical planning postpartum. No changes were made to her care at this time.
She was admitted for fourth time at 25w1d with slurred speech and bilateral upper extremity weakness. Magnetic resonance imaging demonstrated new, multifocal ischemic strokes. The patient again declined inpatient management and requested ongoing outpatient care despite her presenting symptoms persisting. At her next prenatal visit the following week, she continued to demonstrate right upper extremity weakness and slurred speech, which was attributed to her most recent stroke.
In the outpatient setting at 26w3d, neurology ordered another brain MRI which demonstrated new infarcts compared to her study two weeks prior. Given the progression of her disease a multidisciplinary care coordination meeting was held to determine optimal delivery timing for maternal benefit. Consensus between MFM and neurology was for delivery at 28 week given a perceived rising risk of maternal mortality with the ongoing pregnancy.
She was admitted at 27w3d following extensive counseling to the antepartum service for delivery optimization, still planning for 28 week. On admission, her blood pressure was 137/67 and neurological exam was significant for slurred speech, aphasia, and right sided weakness, worse in the face and upper extremity. Blood glucose on admission was 122. Neurology, neonatology, anesthesia, and ethics were consulted and the surgical intensive care unit (ICU) was notified of delivery planning. She received antenatal corticosteroids, and her insulin regimen was adjusted to counteract steroid effect. Her goal blood pressure range was 140–150s systolic over 90–100s diastolic.
Overnight from hospital day 2, she was transitioned to an insulin drip to optimize glucose control. In the early morning of hospital day 3, she developed sustained hypertension in the 180–190s systolic and 80–90s diastolic. She received a single dose of short-acting Labetalol with plans to transition to an intra-arterial Nicardipine drip if she did not respond. She was transferred to the ICU and 10 minutes later her blood pressure returned to 143/92.
Later that same day she experienced new onset dysphagia and repeat MRI demonstrated a novel ischemic stroke in the middle cerebral artery (MCA) territories. Hence, the decision was made to proceed with delivery. Major components of her care plan included an arterial line, slow bolus epidural, magnesium sulfate for fetal neuroprotection, maintenance of intrapartum blood pressure, and surgical ICU with transfer to the University Hospital neurological ICU if she required a higher level of care.
She was delivered on hospital day 3 at 27w5d via repeat cesarean section via classical hysterotomy. Procedure was complicated by postpartum hemorrhage of 2.3 liters due to uterine atony that was managed with intra-myometrial Oxytocin, Carboprost, rectal Misoprostol and oxidized cellulose powder. Surgical intervention included figures of eight sutures, B-lynch stitching, and bilateral suture ligation of utero-ovarian ligaments. Goal blood pressure range was maintained intraoperatively and planned bilateral salpingectomy was completed.
Post-operatively, the patient was transferred back to the ICU as planned and within one hour post-op, she became obtunded with a flaccid right upper extremity and became apneic. She was intubated and bagged. Continuous pulse oximetry showed sats at or above 95% throughout and repeat imaging did not demonstrate new infarct or hemorrhage. She was transferred to the University Hospital Neurological ICU.
On postpartum day 1, she remained intubated but was responding to painful stimuli. She developed a sustained fever with maximum temperature of 102.8 Fahrenheit. Workup was negative and she was empirically treated with Piperacillin-Tazobactam and Clindamycin to cover endometritis per local institution protocol. On postpartum day 2, she was unresponsive to painful stimuli and underwent diagnostic cerebral angiogram which demonstrated severe stenosis of the left MCA and occlusion of the right MCA. It was explained to the patient’s family that this was suggestive of expansive deficits with limited to no projected recovery.
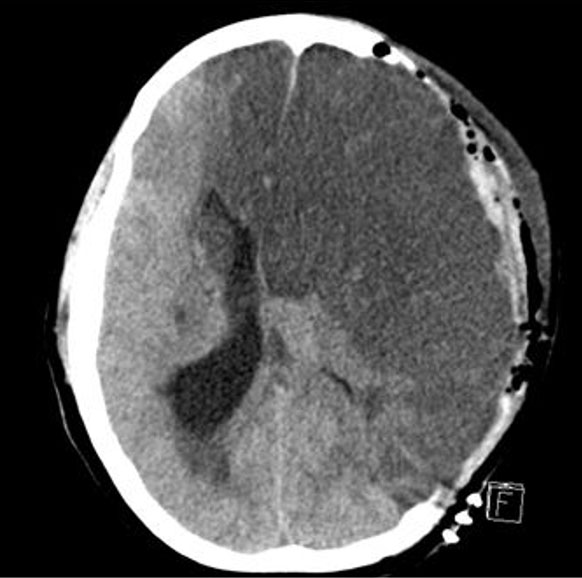
Her exam worsened and was significant for bilateral fixed and dilated pupils. Stat head CT showed a 9 mm midline shift of the left hemisphere (Figure 3). She was taken for emergency, decompressive left hemicraniectomy and intracerebral pressure monitor placement. Imaging from postpartum day 4, postoperative day 1 was significant for a devastating left hemispheric infarct.
On postpartum day 5, Neurology counseled the family on the patient’s unlikely recovery. Her code status was changed to do not resuscitate (DNR) and into postpartum day 6, she had several episodes of ICP crisis (>22–24 mmHg). As her status worsened, her labs demonstrated marked metabolic abnormalities including hypernatremia, acidosis, and hyperglycemia. The neonatal intensive care unit (NICU) coordinated a visit with her newborn as she was transitioned to comfort care. The following day the decision was made to withdraw care. On postpartum day 7, a path of honor was held, and the patient died. She saved the lives of 3 individuals via organ donation.
Discussion
Available evidence suggests that pregnancy increases the risk of stroke by 34% in patients with Moyamoya disease [1],[2]. This has been attributed to physiologic changes of pregnancy including vasodilation, activation of the renin-angiotensin system, hypercoagulability, and normal labor along with the increased risk of hypertensive disorders [4]. The major factor influencing outcomes in pregnancy is whether the diagnosis of Moyamoya disease was made prior to conception or in the antepartum period.
When Moyamoya diagnosis is made prior to pregnancy, it allows for medical and/or surgical optimization. If advanced stage, limited literature suggests that patients should undergo cerebral revascularization prior to conception [5]. During pregnancy, these patients should be managed by a multidisciplinary team with care taken to optimize blood pressure control and maintain adequate hydration. Delivery and the immediate postpartum period are associated with most dramatic fluid shifts and blood pressure fluctuations. Thus meticulous delivery planning is essential [6]. In case reports of patients who were diagnosed with Moyamoya disease prior to pregnancy, the cesarean section rate is reported at 76.3%, and good maternal outcomes without significant stroke morbidity and mortality is reported at over 90% [6].
The prognosis is very different for patients who are diagnosed with Moyamoya in the antepartum period. According to a meta-review of 54 articles published on Moyamoya disease diagnosed in pregnancy, the mean gestational age of diagnosis is in the 28th week. Up to 70% of these diagnoses are made following hemorrhagic stroke. In this group, maternal mortality is 13.6% and risk of fetal demise is 23.5%. Most neurosurgical experts recommend delaying surgical revascularization until after delivery [6].
Conclusion
Pregnancy significantly increases the risk of stroke in patients with Moyamoya disease. Two key components in managing these patients are multidisciplinary care and maintaining hemodynamic stability. This case highlights the significant risk associated with uncontrolled Moyamoya disease during pregnancy, suggesting that timely diagnosis and intervention are critical for reducing maternal and fetal risks. The outcomes of this case may even suggest that untreated Moyamoya disease is a contraindication to pregnancy.
REFERENCES
1.
Inayama Y, Kondoh E, Chigusa Y, Io S, Funaki T, Matsumura N, et al. Moyamoya disease in pregnancy: A 20-year single-center experience and literature review. World Neurosurg 2019;122:684–91.e2. [CrossRef]
[Pubmed]

2.
Ma JC, Burlingame JM. Moyamoya disease in pregnancy: A case series and review of management options. Hawaii Med J 2011;70(8):161–3.
[Pubmed]

3.
Pankaj P, Sharma M. Moya moya disease and stroke in pregnancy (4599). Neurology 2021;96(15_suppl). [CrossRef]

4.
5.
Weston E, Mehta N. Moyamoya disease in pregnancy and delivery planning: A case series and literature review. Obstet Med 2016;9(4):177–80. [CrossRef]
[Pubmed]

6.
Maragkos GA, Ascanio LC, Chida K, Boone MD, Ogilvy CS, Thomas AJ, et al. Moyamoya disease in pregnancy: A systematic review. Acta Neurochir (Wien) 2018;160(9):1711–9. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Acknowledgments
Thank you to all the OB/GYN residents and faculty who contributed to the care of this patient throughout her prenatal care and several admission.
Author ContributionsOlivia Wilson - Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Gilad Gross - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Christopher Buchanan - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Olivia Wilson et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.