|
Case Report
Targeted therapy for breast cancer during pregnancy: A case report and literature review
1 Université Cathologique de Louvain (UCL) – Cliniques Universitaires Saint Luc, Brussels, Belgium
Address correspondence to:
Marie Dieudonné
Université Cathologique de Louvain (UCL) – Cliniques Universitaires Saint Luc, Brussels, Belgium,
Belgium
Message to Corresponding Author
Article ID: 100215Z08MD2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Dieudonné M, Steenhaut P, Cornelis F, Duhoux F, Van Nieuwenhove S, Berlière M. Targeted therapy for breast cancer during pregnancy: A case report and literature review. J Case Rep Images Obstet Gynecol 2025;11(2):32–36.ABSTRACT
Introduction: Breast cancer is the most common malignancy diagnosed during pregnancy, with HER2-positive subtypes posing particular therapeutic challenges due to the fetal risks associated with targeted therapies. Despite growing data, management decisions remain complex and often individualized.
Case Report: We report the case of a 38-year-old woman diagnosed with HER2-positive, hormone receptor-positive invasive ductal carcinoma of the breast. We discovered a pregnancy at 25 weeks of gestation, while neoadjuvant chemotherapy, including trastuzumab and pertuzumab, had already been initiated. The first ultrasound revealed anhydramnios with unremarkable fetal renal morphology. From the diagnosis, anti-HER2 therapy was discontinued, but the weekly paclitaxel was continued. At 33 weeks, severe renal and pulmonary hypoplasia were detected. However, partial reversal of anhydramnios was observed by 36 weeks. The patient delivered at 38 weeks a male infant with early respiratory distress and suspected pulmonary hypertension, but with preserved renal function. The infant was discharged in good condition. The mother achieved a complete pathological response and remains in remission one year after pregnancy diagnosis.
Conclusion: The use of anti-HER2 therapy during pregnancy, particularly in the second and third trimesters, is associated with significant fetal risks. These risks may exhibit partial reversibility after drug cessation. Early pregnancy detection and individualized treatment planning are essential to optimize the maternal and neonatal outcomes.
Keywords: Anhydramnios, Breast cancer, Pertuzumab, Pregnancy, Target therapy, Trastuzumab
Introduction
Breast cancer is the most common cancer among women, with 2.3 million new cases reported in 2020 worldwide [1]. In Europe, it accounted for 2% of all deaths in 2021, and in 2022, breast cancer represented 30% of all newly diagnosed cancers in women. It is the leading cause of cancer-related mortality in women, responsible for one in six female cancer deaths [2].
Cancer during pregnancy is rare, occurring in approximately 1 in 1000 pregnancies. Among these, the most frequent is breast cancer, comprising 40% of cases according to the International Network on Cancer, Infertility and Pregnancy (INCIP) [3],[4]. The management of cancer in pregnancy requires a balance between optimizing maternal treatment and minimizing fetal risk. In this case report, we will discuss about the management of breast cancer during pregnancy, with a focus on anti-HER2 targeted therapies.
Case Report
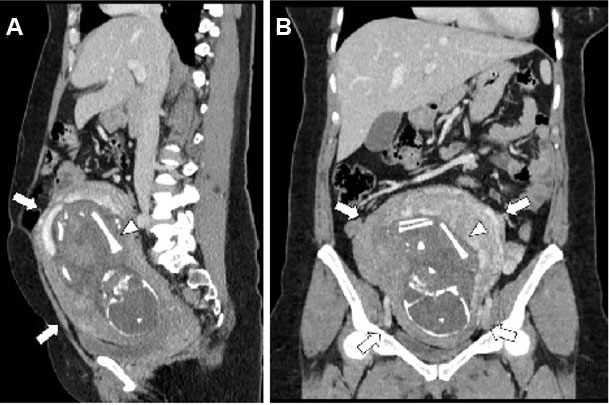
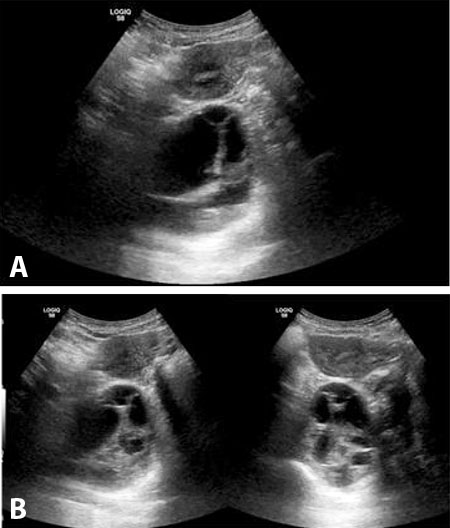
Our patient is a 38-year-old woman from Burundi with no significant past medical history. She had two full-term pregnancies. Her contraception (injection) was efficient until June 2023. During the evaluation of a breast mass, she was diagnosed with grade 3 invasive ductal carcinoma of the right breast (T1c, N1, M0, according to the AJCC 8th edition classification) which was both hormone receptor-positive (HR+) and Human Epidermal Growth Factor Receptor 2 positive (HER2+) (Figure 1).
The initial treatment initiated in October 2023 in Burundi was two cycles of epirubicin and cyclophosphamide. This was followed by eight cycles of paclitaxel, pertuzumab, and trastuzumab started in February 2024 in Belgium. No pregnancy tests were performed during the chemotherapy.
After one month, we made a staging computed tomography (CT) scan for a palpable pelvic mass, which incidentally revealed a pregnancy on April 3, 2024. The patient was referred to a prenatal diagnostic unit, where gestational age was estimated at 25 weeks and 4 days, with an estimated due date of July 13, 2024.
The first ultrasound examination revealed anhydramnios. The kidneys appeared normal and well-differentiated, the bladder was not visualized, and the stomach appeared small. On the same day, a cordocentesis was performed, showing elevated β2-microglobulin (6.91 mg/L), normal cystatin C (0.62 mg/L), and a normal molecular karyotype.
We adjusted the cancer treatment: paclitaxel was continued but the targeted therapy was discontinued. A right mastectomy with axillary lymph node dissection was performed on May 27, 2024.
At 33 weeks and 3 days of gestation, severe pulmonary and renal hypoplasia were noted. At 36 weeks and 2 days, a small amniotic fluid pocket became visible for the first time, along with clearly identifiable bladder and stomach structures. The kidneys remained well-differentiated and non-hypoplastic.
At 38 weeks and 3 days, the patient delivered a male infant weighing 2700 grams. The newborn required intubation 24 minutes after birth. By day 12 of life, he no longer required invasive ventilation. Cardiac ultrasound raised suspicion of pulmonary hypertension, but renal function was normal, and both kidneys were well differentiated. The newborn was discharged from the neonatal intensive care unit with a favorable prognosis.
Postpartum, the patient underwent radiotherapy and received adjuvant endocrine therapy.
One year after the diagnosis of her pregnancy, the mother remains in remission, and the infant exhibits optimal growth and neurodevelopmental outcomes.
Discussion
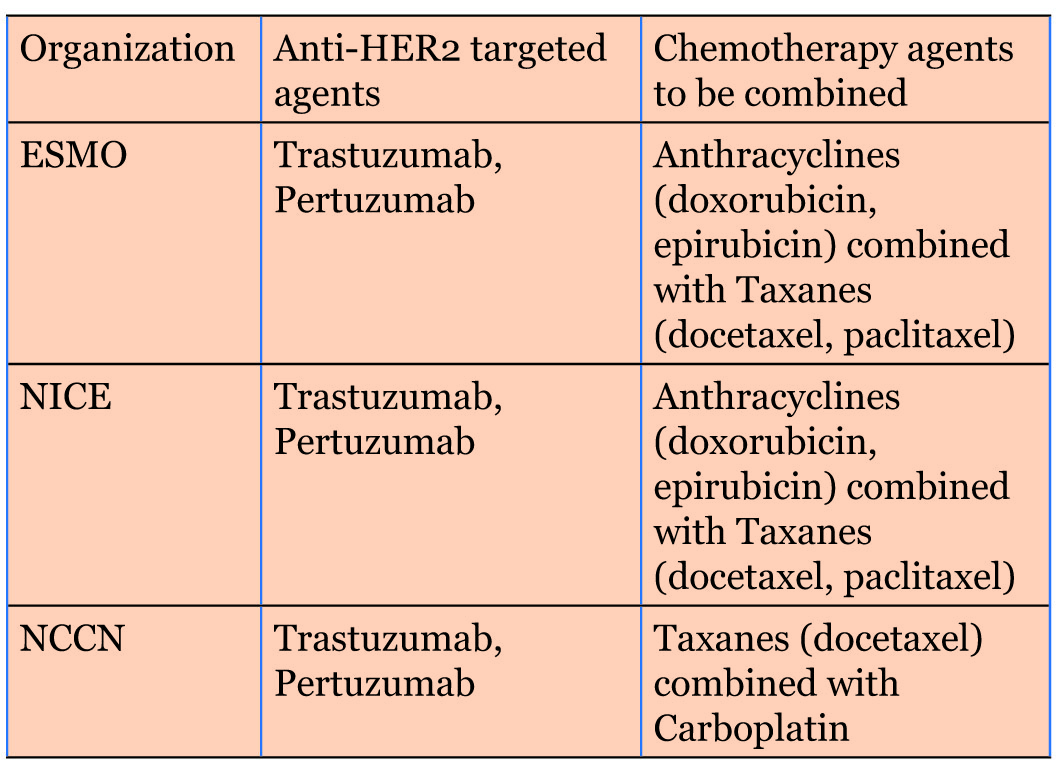
The treatment of hormone receptor-positive (HR+) and HER2-positive (HER2+) invasive breast cancer is well defined by international guidelines. Organizations such as European Society For Medical Oncology (ESMO), National Institute for Health and Care Excellence (NICE), American Society of Clinical Oncology (ASCO), and National Comprehensive Cancer Network (NCCN) concur that neoadjuvant therapy combining anti-HER2 targeted agents (trastuzumab and pertuzumab) with chemotherapy is recommended based on individual tumor characteristics [5],[6],[7],[8]. Commonly used chemotherapeutic agents include anthracyclines (e.g., doxorubicin, epirubicin) and taxanes (e.g., docetaxel and paclitaxel). NICE and ESMO guidelines support adding anti-HER2 therapy to chemotherapy, while the NCCN recommends a regimen combining trastuzumab, pertuzumab, a taxane, and carboplatin [7] (Figure 2).
Depending on tumor characteristics, surgery is usually performed and may be followed by radiotherapy. After surgery, adjuvant systemic therapy generally includes the same anti-HER2 agents used in the neoadjuvant setting if a pathological complete response was achieved. If not, the regimen is generally switched to trastuzumab emtansine. For hormone receptor-positive tumors, antihormonal therapy (e.g., tamoxifen) is added [5],[6],[7],[8].
However, the management of these therapies during pregnancy is challenging. A study by the International Network on Cancer, Infertility and Pregnancy (INCIP) found that 67% of 1170 pregnant patients diagnosed with cancer received treatment during pregnancy. 88% delivered live-born infants, although 48% of deliveries were preterm. The study concluded that increasing numbers of pregnant patients are receiving cancer therapy, which has improved live births rates and reduced rates of iatrogenic prematurity. Nonetheless, chemotherapy during pregnancy remains associated with an increased incidence of fetal complications, including small-for-gestational-age (SGA) newborns and neonatal intensive care unit (NICU) admissions [4].
While continuing cancer treatment during pregnancy is often preferable to terminating the pregnancy, certain drugs are contraindicated [4],[9],[10]. Agents such as anthracyclines, fluoropyrimidines, cyclophosphamide, taxanes, and platinum compounds may be administered safely after the first trimester, once organogenesis is complete. To allow adequate maternal and fetal bone marrow recovery before delivery, a chemotherapy-free interval of three weeks is recommended prior to birth. Surgical procedures are considered safe throughout pregnancy. Low doses of 99m-technetium and methylene blue may be used for tumor microinjection, although blue dye is generally avoided for sentinel lymph node biopsy due to the risk of maternal anaphylaxis [3],[4],[10]. Hormone therapies, by contrast, are contraindicated during pregnancy and must be postponed until the postpartum period.
With respect to targeted therapy, evidence remains limited. However, current recommendations from INCIP, ESMO, and NCCN are consistent: trastuzumab is contraindicated during the second and third trimesters, due to a significantly increased risk of oligohydramnios and/or anhydramnios, both of which can result in preterm delivery, fetal hypoxia, and perinatal death. Trastuzumab has also been associated with fetal renal and pulmonary dysfunction. As a result, anthracyclines and platinum agents are preferred during pregnancy, given their better-documented safety profile compared to monoclonal antibodies [3],[4],[7],[10].
Two meta-analyses in 2021 assessed the effects of trastuzumab exposure during pregnancy. Xia et al. included 22 studies involving 22 pregnant women and 23 fetuses, while Andrikopoulou et al. analyzed 28 studies with 30 women and 32 fetuses [11],[12] (Figure 3).
In Xia et al.’s analysis, 77.27% of trastuzumab-exposed pregnancies were complicated, with oligohydramnios being the most common adverse event (68.18%). A decrease in left ventricular ejection fraction (LVEF) was observed in 9.09%. At birth, 39.13% of neonates were healthy (mean Apgar score: 7.9; mean birth weight: 2310 g), while 60.87% experienced pulmonary complications (transient tachypnea and respiratory distress syndrome). In 17.39% of cases, the neonates had severe postnatal complications leading to death. An interessant fact is that the patients exposed only during the first trimester had significantly fewer complications, and maternal age over 30 was associated with increased neonatal risk [12].
Andrikopoulou et al. similarly identified oligo-/anhydramnios as the most frequent complication (58.1%). Only one case occurred during the first trimester; the remainder followed second- or third-trimester exposure. Among neonates, 43.3% were born healthy; the others had postnatal complications including renal agenesis, hypoplasia, pulmonary hypertension, respiratory distress, or multi-organ failure. Among fetuses exposed to trastuzumab beyond the first trimester, 41.7% were healthy, versus 75% among those exposed only during the first trimester. Maternal prognosis was favorable in all cases (median follow-up of 47 months), although 10% experienced trastuzumab-related cardiotoxicity [11].
Despite 19 overlapping studies, the findings from both meta-analyses strongly support the association between trastuzumab exposure and fetal complications, including reduced amniotic fluid, renal/pulmonary dysfunction, and reversible maternal cardiotoxicity [11],[12].
These complications are thought to be trimester-dependent. The minimal risk associated with first-trimester exposure is likely due to limited placental transfer of trastuzumab (an IgG1 monoclonal antibody, ~150 kDa), which relies on active FcRn-mediated transport that becomes effective only after 14th weeks of gestation [4],[10],[11],[12].
One proposed mechanism for the amniotic fluid reduction is trastuzumab-mediated blockade of epidermal growth factor receptors (EFGRs) in the fetal renal epithelium, which impairs nephrogenesis. Additional hypotheses include inhibition of VEGF expression (which regulates amniotic fluid balance) and interference with aquaporin function [11],[12] [13],[14],[15].
Notably, studies suggest that oligo-/anhydramnios is reversible upon treatment discontinuation, with normalization of fluid levels often occurring within days to weeks. There is a correlation between the severity of oligohydramnios and the duration of trastuzumab exposure, and limited exposure may have minimal impact on fetal growth [3],[11],[12][14],[15]. Trastuzumab-induced cardiotoxicity is typically reversible, though its precise mechanisms remain unclear. Hypotheses include HER2 inhibition-induced cardiomyocyte apoptosis, reduced reactive oxygen species (ROS) neutralization, and impaired nitric oxide synthesis via HER4 signaling [12].
The second anti-HER2 agent, pertuzumab lacks sufficient human data regarding its use during pregnancy [4],[10],[14]. Food and Drug Administration (FDA) labeling and preclinical studies have raised concerns about oligohydramnios, delayed renal development, and fetal demise [16]. A single case report documented the discontinuation of trastuzumab and pertuzumab at 10 weeks of gestation after early exposure; the patient experienced reversible cardiac dysfunction, and both maternal and neonatal outcomes were favorable [17].
In our case, the patient received trastuzumab and pertuzumab during the second trimester, and subsequently developed anhydramnios, pulmonary and renal hypoplasia, and neonatal respiratory complications—findings consistent with the literature. As previously reported, some effects were partially reversible after treatment cessation, notably renal changes and amniotic fluid volume. However, due to the limited data on pertuzumab during pregnancy, its specific role in fetal toxicity remains unclear. Like trastuzumab, pertuzumab is a large monoclonal antibody targeting HER2, but binds a different epitope-subdomain II, inhibiting HER2 dimerization with other HER family receptors. Combined administration of trastuzumab and pertuzumab enhances blockade of HER2-driven signaling pathways [18].
Given the pharmacological similarities, it is plausible that pertuzumab contributes to the fetal adverse effects observed when used in combination with trastuzumab during pregnancy.
Conclusion
This clinical case reveals the complexity of managing hormone receptor-positive, HER2-positive breast cancer during pregnancy. It demonstrates the importance of individualized, multidisciplinary care to balance maternal treatment efficacy with fetal safety. Current studies confirms that exposure to trastuzumab—and potentially pertuzumab—during the second and third trimesters is associated with a significantly increased risk of fetal complications, including oligo-/anhydramnios, renal and pulmonary hypoplasia, and neonatal respiratory disorders. However, many of these effects appear to be partially reversible if the therapy is discontinued. This case illustrates this affirmation by the positive neonatal outcome in this case.
The close clinical follow-up and multidisciplinary collaboration enabled appropriate therapeutic adjustments in real time, guided by clinical progression and evolving fetal imaging findings. This case underscores the value of a personalized approach in managing cancer during pregnancy.
Further research is urgently needed to evaluate the safety profile of anti-HER2 agents, particularly pertuzumab, during pregnancy and to establish evidence-based guidelines. In the meantime, the clinical decision must be guided by the best available data, careful maternal-fetal risk assessment, and a shared decision making process centered on the patient’s values and priorities.
REFERENCES
1.
Arnold M, Morgan E, Rumgay H, Mafra A, Singh D, Laversanne M, et al. Current and future burden of breast cancer: Global statistics for 2020 and 2040. Breast 2022;66:15–23. [CrossRef]
[Pubmed]

3.
Maggen C, Wolters VERA, Cardonick E, Fumagalli M, Halaska MJ, Lok CAR, et al. Pregnancy and cancer: The INCIP project. Curr Oncol Rep 2020;22(2):17. [CrossRef]
[Pubmed]

4.
Boere I, Lok C, Poortmans P, Koppert L, Painter R, Vd Heuvel-Eibrink MM, et al. Breast cancer during pregnancy: Epidemiology, phenotypes, presentation during pregnancy and therapeutic modalities. Best Pract Res Clin Obstet Gynaecol 2022;82:46–59. [CrossRef]
[Pubmed]

5.
6.
Giordano SH, Franzoi MAB, Temin S, Anders CK, Chandarlapaty S, Crews JR, et al. Systemic therapy for advanced human epidermal growth factor receptor 2-positive breast cancer: ASCO guideline update. J Clin Oncol 2022;40(23):2612–35. [CrossRef]
[Pubmed]

7.
Gradishar WJ, Moran MS, Abraham J, Abramson V, Aft R, Agnese D, et al. Breast cancer, Version 3.2024, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 2024;22(5):331–57. [CrossRef]
[Pubmed]

8.
Loibl S, André F, Bachelot T, Barrios CH, Bergh J, Burstein HJ, et al. Early breast cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol 2024;35(2):159–82. [CrossRef]
[Pubmed]

9.
Janssen JM, Van Calsteren K, Dorlo TPC, Halaska MJ, Fruscio R, Ottevanger P, et al. Population pharmacokinetics of docetaxel, paclitaxel, doxorubicin and epirubicin in pregnant women with cancer: A study from the international network of cancer, infertility and pregnancy (INCIP). Clin Pharmacokinet 2021;60(6):775–84. [CrossRef]
[Pubmed]

10.
Loibl S, Azim HA Jr, Bachelot T, Berveiller P, Bosch A, Cardonick E, et al. ESMO expert consensus statements on the management of breast cancer during pregnancy (PrBC). Ann Oncol 2023;34(10):849–66. [CrossRef]
[Pubmed]

11.
Andrikopoulou A, Apostolidou K, Chatzinikolaou S, Bletsa G, Zografos E, Dimopoulos MA, et al. Trastuzumab administration during pregnancy: An update. BMC Cancer 2021;21(1):463. [CrossRef]
[Pubmed]

12.
Xia LY, Hu QL, Zhou Q. Use of trastuzumab in treating breast cancer during pregnancy: A systematic review and meta-analysis. BMC Womens Health 2021;21(1):169. [CrossRef]
[Pubmed]

13.
Piccart-Gebhart M, Holmes E, Baselga J, de Azambuja E, Dueck AC, Viale G, et al. Adjuvant lapatinib and trastuzumab for early human epidermal growth factor receptor 2-positive breast cancer: Results from the randomized phase III adjuvant lapatinib and/or trastuzumab treatment optimization trial. J Clin Oncol 2016;34(10):1034–42. [CrossRef]
[Pubmed]

14.
Goller SS, Markert UR, Fröhlich K. Trastuzumab in the treatment of pregnant breast cancer patients – An overview of the literature. Geburtshilfe Frauenheilkd 2019;79(6):618–25. [CrossRef]
[Pubmed]

15.
17.
Al-Shamsi HO, Abdelwahed N, Singh M, Abyad AM, Elsabae S, Abdelgawad T, et al. First reported case of successful conception and delivery during stage iv breast cancer treatment: A case report and literature review. Cureus 2023;15(10):e47201. [CrossRef]
[Pubmed]

18.
Yildirim N, Bahceci A. Use of pertuzumab and trastuzumab during pregnancy. Anticancer Drugs 2018;29(8):810–13. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Marie Dieudonné - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Patricia Steenhaut - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Frank Cornelis - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
François Duhoux - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Sandy Van Nieuwenhove - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Martine Berlière - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Marie Dieudonné et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.