|
Case Report
Struma ovarii coexisting with recurrent uterine fibroids in a postmenopausal patient
1 Department of Obstetrics/Gynaecology, University of Uyo, Uyo, Nigeria
2 Department of Obstetrics/Gynaecology, University of Uyo Teaching Hospital, Uyo, Nigeria
3 Department of Anaesthesia, University of Uyo Teaching Hospital, Uyo, Nigeria
4 Department of Surgery, University of Uyo Teaching Hospital, Uyo, Nigeria
5 Department of Histopathology, University of Uyo, Uyo, Nigeria
Address correspondence to:
Aniekan Monday Abasiattai
Professor, Department of Obstetrics/Gynaecology, University of Uyo, Uyo,
Nigeria
Message to Corresponding Author
Article ID: 100223Z08AA2026
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Abasiattai AM, Olisaeeke FI, Antai MMS, Ugochukwu RO, Abudu EK. Struma ovarii coexisting with recurrent uterine fibroids in a postmenopausal patient. J Case Rep Images Obstet Gynecol 2026;12(1):9–13.ABSTRACT
Introduction: Ovarian teratomas are ovarian tumors that originate from primordial germ cells. Struma ovarii (SO) is a very rare specialized monodermal variant of the mature cystic teratoma, which consists exclusively or predominantly of mature thyroid tissue and occurs most commonly in premenopausal women.
Case Report: We hereby report the rare case of a 70-year-old postmenopausal woman who presented with vaginal bleeding and abdominal distention 20 years after the menopause. She was found to have a huge uterine mass, hemorrhagic ascites, and a left adnexal cystic mass with solid areas mimicking a malignancy. Histological analysis of the surgical specimens confirmed a SO coexisting with recurrent uterine fibroids. She had a total abdominal hysterectomy and bilateral salpingo-oophorectomy with a good outcome.
Conclusion: Benign SO, a very rare variety of specialized monodermal teratomas, can coexist with ascites and large recurrent uterine fibroids several years after menopause, mimicking malignancy. Timely diagnosis and excision of both tumors via an abdominal hysterectomy and bilateral salpingo-oophorectomy resulted in a good outcome in our patient.
Keywords: Monodermal teratoma, Postmenopausal fibroids, Struma ovarii, Teratoma variant
Introduction
Ovarian germ cell tumors represent a group of histologically diverse neoplasms that arise from primitive pluripotent gonadal germ cells and undergo differentiation into either embryonic or extraembryonic tissue [1]. Teratomas are the most common germ cell tumors, and they may be polydermal (originating from more than one germ cell layer), such as the mature cystic teratoma, or monodermal (originating from a single germ cell layer) [2].
Struma ovarii (SO) is a very rare monodermal variant of the dermoid cyst comprising predominantly or exclusively of thyroid tissue [2]. It is defined as a teratoma in which thyroid tissue constitutes more than 50% of the neoplastic tissue or forms a macroscopically recognisable component of mature teratoma [3],[4]. They account for 0.5–1% of all ovarian tumors, 2% of all germ cell tumors, and 3% of all ovarian teratomas [4]. Struma ovarii are usually benign, but undergo malignant transformation in about 5% of cases [4].
Uterine fibroids, also known as leiomyomas, are benign tumors that arise from myometrial smooth muscle cells [5]. Though their etiology is unknown, they tend to develop and grow under the influence of the ovarian steroid hormones. Fibroids are rare before menarche and typically atrophy after the menopause [6]. Thus, they most commonly occur during the reproductive period and are the most common tumors found in premenopausal women [6].
This is a very rare case of a 70-year-old multiparous lady who presented with vaginal bleeding 20 years after the menopause and was found to have a SO, which coexisted with ascites and large recurrent uterine fibroids. This case report aims to inform clinicians about this rare presentation of these gynecological disorders in the same patient two decades after the menopause, mimicking a malignancy, and review existing literature. To the best of the knowledge of the authors, this is the first case report in which SO coexists with ascites and large recurrent uterine fibroids in a patient several years after menopause.
Case Report
A 70-year-old Para 2+0 retiree presented in our facility with vaginal bleeding of three months duration. The bleeding was intermittent, initially slight, but later increased to moderate to heavy. This was associated with abdominal distension, which had also increased, but there was no history of post-coital bleeding, hormone replacement therapy, or symptoms of thyroid dysfunction. She was 20 years postmenopausal and had undergone myomectomy 30 years earlier with an uncomplicated postoperative period. She was a known asthmatic and hypertensive and was compliant with her antihypertensives. Her last childbirth, a full-term normal vaginal delivery, occurred 37 years before presentation.
On examination, her general condition was satisfactory, she weighed 59 kilograms, and had no goiter. Abdominal examination revealed a healthy subumbilical midline scar. There was also an 18-week-sized abdominopelvic mass, which was firm, non-tender, and smooth-surfaced, with limited mobility.
Pelvic examination revealed an atrophic vulva and vagina. The cervix was also atrophied and deviated superiorly by a firm mass which occupied the entire posterior vaginal fornix, extending to both adnexa and appeared to be continuous with the abdominopelvic mass. Packed cell volume, urea/creatinine, and electrolytes, urinalysis, fasting blood sugar, chest X-ray, and an electrocardiogram did not reveal any abnormality. Urine culture revealed heavy growth of Staphylococcus aureus, for which she received a 10-day course of oral cefuroxime based on the sensitivity result.
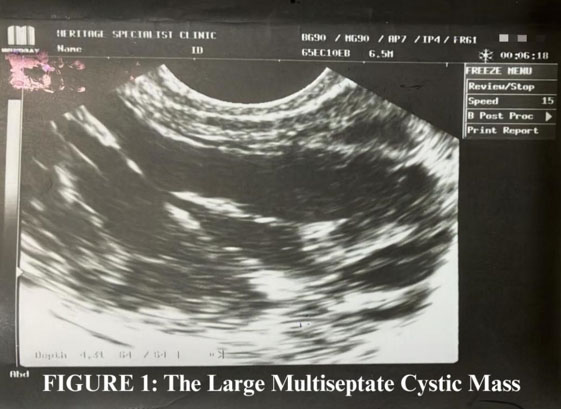
Ultrasonography revealed a fairly large, multiloculated, multiseptated, mixed echogenic mass extending from the left adnexium through the pouch of Douglas to the right, measuring 8.9 × 11.7 cm with a volume of 414 cm³. Irregularly shaped, nodular soft tissue masses were seen within the cystic mass (Figure 1). The uterus was stretched upward by the low-lying echogenic mass with an enlarged upper half having an expanded fundal end with generalised interstitial leiomyoma changes. The endometrial stripe was well appreciated, 2 mm thick, lying more posteriorly.
A tentative diagnosis of postmenopausal bleeding, probably secondary to a malignant uterine disease, to exclude a coexisting malignant ovarian tumor, was entertained.
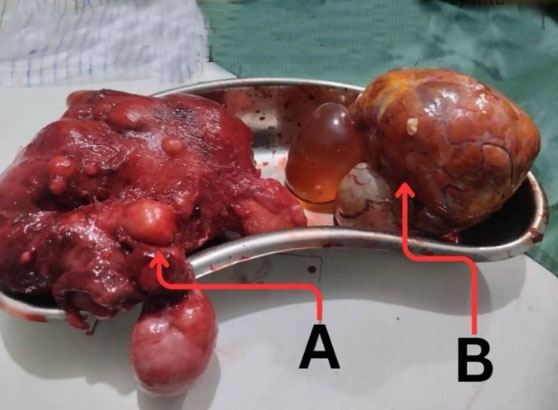
At laparotomy, the abdomen was entered through the old scar, and the incision was extended above the umbilicus. There was approximately 800 mL of hemorrhagic ascitic fluid from which 5 mL was sent for cytology. There was also a large left ovarian cyst that contained some solid areas. The uterus, which was nodular, was enlarged to approximately 18 weeks’ gestational size (Figure 2), and the right ovary was atrophic. A total abdominal hysterectomy and bilateral salpingo-oophorectomy was then performed. Her postoperative period was uneventful, and she was discharged on the 7th postoperative day. At the follow-up clinic, her general condition was satisfactory, all the symptoms had subsided, the abdominal wound had healed well, and the thyroid function test was normal.
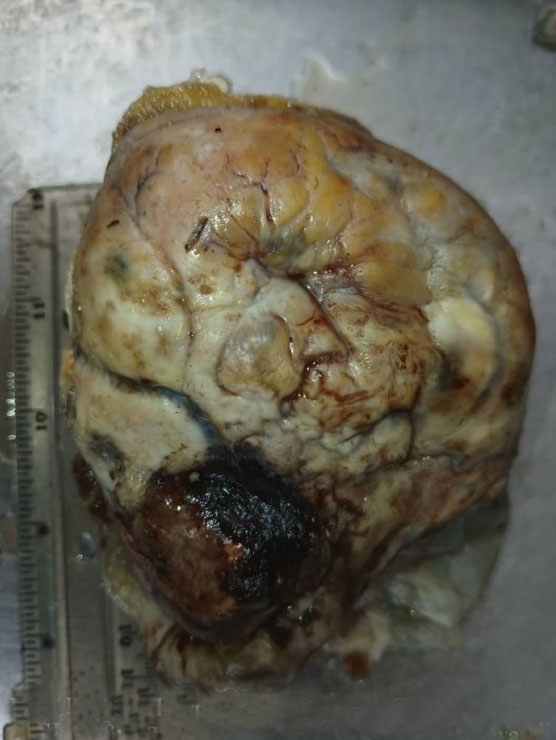
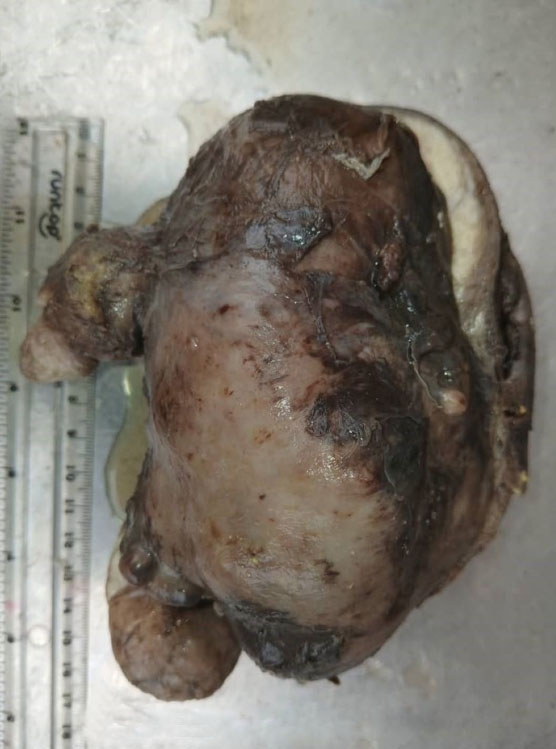
On macroscopic examination, the left ovary contained a dark brown cystic mass with a cystic area of 5.8 cm × 5.0 cm × 3.5 cm occupying a quarter of the entire mass; the mass as a whole measured 12.1 × 8.0 × 7.0 cm and weighed 380 g (Figure 3). Its cut section showed 3 multi-lobular cystic cavities containing light green to grayish-brown mucoid semi-solid materials and measured 1.5, 4.0, and 5.0 cm in widest diameters, respectively. The uterus was huge, irregularly shaped, firm, and multinodular, measuring 14 cm × 9.0 cm × 8.5 cm, weighing 380.0 g, and contained 10 intramural fibrous nodules ranging from 0.5 to 5.5 cm in the largest diameter (Figure 4). There were also 6 nodules in the parametrium.
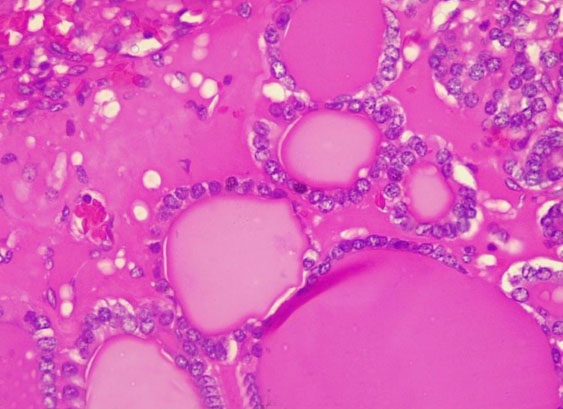
Histology of the left ovary revealed thyroid follicles, cholesterol clefts, and focal areas of multifollicular proliferation with follicular cells exhibiting vesicular nuclei and no features of malignancy, confirming a benign germ cell tumor, specifically SO (Figure 5).
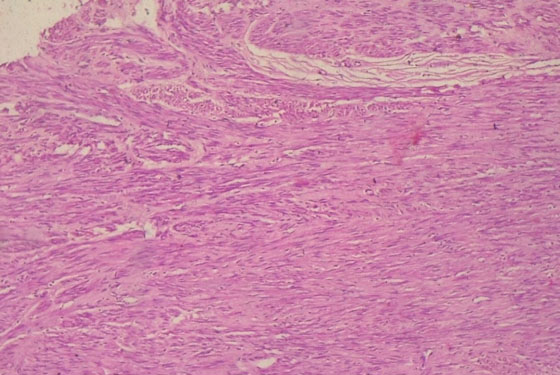
Histology sections of the myometrium and the intramural nodules revealed benign mesenchymal neoplasms composed of proliferating mature smooth muscle cells disposed in a criss-cross pattern within a fibrocollagenous stroma. The tumor cells possessed plump, spindle-shaped, bland nuclei and abundant cytoplasm with no evidence of malignancy (Leiomyomata) (Figure 6). That of the endometrium showed numerous cystically dilated glands lined by hyperplastic columnar epithelia, admixed with fibrocellular stroma with no evidence of atypia or malignancy (simple endometrial hyperplasia without atypia). There was also no evidence of malignancy in the histologic sections of the cervix, the cytology of the ascitic fluid was negative for malignant cells, and nodal status was also negative.
Discussion
Monodermal teratomas are rare, mostly benign neoplasms composed of usually one tissue type. Struma ovarii, in which thyroid tissue predominates, is a very rare variety of monodermal teratomas [1],[4]. It was first described in 1889 by Robert Boettlin, who observed thyroid follicular tissue in ovarian teratomas [7]. While approximately 5–15% of all teratomas contain a small, insignificant focus of thyroid tissue, to qualify as a stroma, more than 50% of the lesion should consist of thyroid tissue [7]. Struma ovarii is usually unilateral and typically occurs between the ages of 30 and 50 years [8]. It is reported to account for about 0.5% to 1.0% of all ovarian tumors and also to occur more frequently in countries where goiter is endemic [9].
Most cases (80%) of SO are asymptomatic; however, when symptoms occur, they are usually non-specific and could include abdominal pain or distension, an asymptomatic pelvic mass, signs and symptoms of urinary and intestinal obstruction, and dyspnoea [9]. As seen in our patient, despite the abundance of mature thyroid tissue in the ovarian mass, hyperthyroidism is rare in patients with SO, which is reported to occur in only about 3–8% of cases [8]. Since benign SOs contain normal thyroid tissue, they have a high uptake of iodine and may secrete thyroid hormones [8]. As in the case presented, ascites occurs in 15–20% of cases either in isolation or with pleural effusion as part of pseudo-Meigs syndrome, and this usually regresses following surgical removal of the tumor [9].
Several hypotheses have been proposed for the genesis of the ascites in SO. These include mechanical irritation of the peritoneum by the tumor leading to fluid accumulation, obstruction of blood and/or lymph vessels by the tumor, and production of vasoactive substances resulting in increased capillary permeability, and fluid leakage into the abdominal cavity [10].
Our case is unique and interesting because our patient, who was 70 years old, presented with a large, multiloculated, multiseptate cystic ovarian mass, which had soft tissue masses and hemorrhagic ascitic fluid two decades after the menopause, which are all the features associated with ovarian malignancy [9]. Distinguishing between SO and ovarian carcinoma before surgery can be difficult, as the symptoms and clinical presentation are identical [10]. Most cases of SO are diagnosed during histopathologic examination following surgery [9], as exemplified by our patient. Macroscopically, the tumor is usually solid or, as seen in our patient, cystic, with solid areas or protrusions, while on microscopy, it is composed of mature thyroid tissue consisting of colloid follicles of various sizes [10].
Our patient also had postmenopausal bleeding and a huge uterine mass, which seemed to have recently increased in size; these are suggestive of a malignant uterine disease, typically, a leiomyosarcoma (LMS) [11]. Fibroids do not characteristically develop or grow after menopause without hormone therapy, particularly several years after, and in fact, tend to regress during this period [11]. Continued growth of any uterine mass with or without vaginal bleeding after the menopause is a distinct feature of the more sinister LMS, which is associated with a poor prognosis and high mortality rate [11],[12].
There are no specific ultrasonographic features for SO, but an ovarian cyst with a large solid component may heighten suspicion [9]. Struma ovarii has a good prognosis, and definitive treatment is surgical removal of the tumor [3],[4]. Our patient had a total abdominal hysterectomy and a bilateral salpingo-oophorectomy because she was postmenopausal, had completed her family, and also had recurrent uterine fibroids. This led to a good outcome.
Conclusion
Benign struma ovarii, a very rare variety of specialized monodermal teratomas, can coexist with ascites and large recurrent uterine fibroids several years after menopause, mimicking malignancy. Timely diagnosis and excision of both tumors via an abdominal hysterectomy and bilateral salpingo-oophorectomy resulted in a good outcome in our patient. Large multiseptate multicystic ovarian tumors coexisting with large recurrent uterine fibroids and ascites several years after the menopause can indeed be benign lesions and should be managed as such.
REFERENCES
1.
2.
3.
Wee JYS, Li X, Chern BSM, Chua ISY. Struma ovarii: Management and follow-up of a rare ovarian tumour. Singapore Med J 2015;56(1):35–9. [CrossRef]
[Pubmed]

4.
Singh P, Lath N, Shekhar S, Goyal M, Gothwal M, Yadav G, et al. Struma ovarii: A report of three cases and literature review. J Midlife Health 2018;9(4):225–9. [CrossRef]
[Pubmed]

5.
Abasiattai AM, James R, Atat B, Edemekong V, Etuknwa U. Nonpuerperal uterine inversion complicating a huge prolapsed submucous myoma: A case report. Int J Gynaecol Obstet 2025;169(3):1240–2. [CrossRef]
[Pubmed]

6.
Senbango OC, Ottan TA, Akinlinsi FM, Oshodi YA, Rabiu KA, Adewunmi AA. Clinical presentation of uterine Leiomyoma and the outcome of surgical management options in a Nigerian tertiary health facility. Annals of Health Research 2023;9(2):136–45. [CrossRef]

7.
Yoo SC, Chang KH, Lyu MO, Chang SJ, Ryu HS, Kim HS. Clinical characteristics of struma ovarii. J Gynecol Oncol 2008;19(2):135–8. [CrossRef]
[Pubmed]

8.
9.
Costa MA, Póvoa AM, Pires MC, Paiva VL, Pinto C, Martínez-de-Oliveira J. Struma ovarii: A rare form of presentation and clinical review. Acta Obstet Gynecol Scand 2005;84(8):819–20. [CrossRef]
[Pubmed]

10.
Rim SY, Kim SM, Choi HS. Struma ovarii showing clinical characteristics of ovarian malignancy. Int J Gynecol Cancer 2005;15(6):1156–9. [CrossRef]
[Pubmed]

11.
Ulin M, Ali M, Chaudhry ZT, Al-Hendy A, Yang Q. Uterine fibroids in menopause and perimenopause. Menopause 2020;27(2):238–42. [CrossRef]
[Pubmed]

12.
Yang Q, Madueke-Laveaux OS, Cun H, Wlodarczyk M, Garcia N, Carvalho KC, et al. Comprehensive review of uterine leiomyosarcoma: Pathogenies, diagnosis, prognosis and targeted therapy. Cells 2024;13(13):1106. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Aniekan Monday Abasiattai - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Felix Ikenna Olisaeeke - Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Mary Magdalene Sylvester Antai - Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Richard Odionyeme Ugochukwu - Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Emmanuel Kunle Abudu - Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2026 Aniekan Monday Abasiattai et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.