|
Case Report
Unexplained third-trimester intrauterine fetal demise after vanishing twin syndrome in a large for gestational age fetus: Possible association of subclinical and multifactorial hypercoagulability factors
1 Medical Student, DeBusk College of Osteopathic Medicine, 9737 Cogdill Rd, Knoxville, TN, USA
2 Obstetrician and Gynecologist, Tennova Women’s Health Specialists & Midwifery Services, Tennova North Knoxville Medical Center, Powell, TN, USA
Address correspondence to:
Benjamin Perdomo
2521 Irvin Chapel Way, Knoxville, TN 37931,
USA
Message to Corresponding Author
Article ID: 100225Z08BP2026
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Perdomo B, Soria J, Toney J. Unexplained third-trimester intrauterine fetal demise after vanishing twin syndrome in a large for gestational age fetus: Possible association of subclinical and multifactorial hypercoagulability factors. J Case Rep Images Obstet Gynecol 2026;12(1):18–21.ABSTRACT
Introduction: Diagnosing late intrauterine fetal demise (IUFD) can be challenging, especially with an otherwise uncomplicated pregnancy course. Identifying maternal risk factors and multifactorial thrombophilia patterns can be helpful to further understand its etiology and provide medical guidance for future pregnancies.
Case Report: A 31-year-old primigravida with a history of polycystic ovary syndrome (PCOS), obesity, and anemia conceived dichorionic-diamniotic twins with clomiphene. The patient had spontaneous early loss of Twin A at about eight weeks’ gestation, which was confirmed by ultrasound as vanishing twin syndrome. The surviving fetus (Twin B) demonstrated appropriate developmental growth and was large-for-gestational-age (LGA) through 33 weeks. At 34+4 weeks, the patient presented with decreased fetal movement, and IUFD was confirmed. Placental pathology and fetal karyotype were unremarkable, despite grossly abnormal placental architecture upon delivery. Two months postpartum, thrombophilia testing revealed a methylenetetrahydrofolate reductase (MTHFR) mutation, low protein S activity, and low PT/INR, highlighting the possibility of a subclinical thrombotic or microvascular process leading up to the fatal event.
Conclusion: This case demonstrates the potential significance of subclinical hypercoagulable or microvascular factors in an otherwise unexplained late IUFD, especially in patients with comorbid conditions such as PCOS, obesity, and vanishing twin syndrome. Consideration of earlier detection and individualized thrombophilia screening may be helpful in developing preventative approaches in similar high-risk pregnancies.
Keywords: Case report, Intrauterine fetal demise, Thrombophilia, Vanishing twin syndrome
Introduction
Approximately 1 in 160 pregnancies in the United States end in intrauterine fetal demise (IUFD) after 20 weeks’ gestation. The most common causes of IUFD include infection, placental insufficiency, hypertensive disorders, and chromosomal abnormalities; however, up to 10–25% of cases are from unexplained causes [1].
The combination of thrombophilic and metabolic disorders, such as polycystic ovary syndrome (PCOS) and obesity, in addition to the physiologic hypercoagulable state of pregnancy, has the potential to contribute to placental vascular pathology even in the absence of histopathologic changes. The objective of this report is to describe a case of late IUFD in a large for gestational age (LGA) fetus following vanishing twin syndrome (VTS), in a mother with multiple prothrombotic and metabolic risk factors.
Case Report
A 31-year-old G1P0 woman with a past medical history of PCOS and obesity [body mass index (BMI) 37.7] conceived a dichorionic–diamniotic twin gestation on 100 mg clomiphene. Her first trimester ultrasound revealed the loss of Twin A at eight weeks’ gestation, consistent with VTS. The surviving twin demonstrated appropriate growth throughout the pregnancy, with estimated-fetal-weight (EFW) increasing from the 63rd percentile at 28 weeks to the 92nd percentile at 33 weeks, consistent with antenatally suspected LGA or an accelerated growth pattern.
Due to her moderate risk factors for preeclampsia, including nulliparity and obesity, the decision was made to start low-dose aspirin at 20 weeks’ gestation. She developed anemia early in the third trimester, in which oral iron supplementation was started at 30 weeks. At 34+4 weeks, she presented with decreased fetal movement and contractions with vaginal bleeding. A bedside sonogram demonstrated a 34-week fetus in vertex position, with no color-flow over the heart, brain, or through the umbilical cord. An anterior placenta was noted with no apparent placental abruption and clear appearing fluid. There was no ultrasound explanation for demise. At this time, plans were discussed for labor induction using high dose misoprostol and spontaneous vaginal delivery ensued without complications.
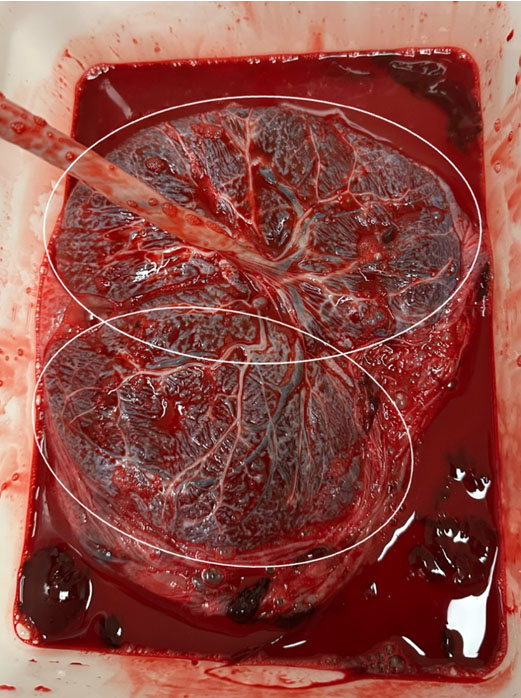
Postpartum evaluation revealed unremarkable infectious or chromosomal anomalies. Though not routinely done, coagulation studies were ordered due to the unexplained etiology of the late IUFD and early VTS. Studies do show that third-trimester IUFD is significantly associated with thrombophilia [2]. The coagulation studies for this patient revealed low PT at 9.4 seconds (ref: 10–13 seconds), INR at 0.8 seconds (ref: 0.9–1.1 seconds), as well as low protein S activity at 43% (ref: 60–140%). Genetic thrombophilia testing identified heterozygosity for the MTHFR C677T mutation. Physical examination of the placenta noted grossly abnormal architecture with two separate identifiable lobes (Figure 1), but there was no histopathologic evidence of infarction, thrombi, or inflammatory infiltrates.
Discussion
A 31-year-old primigravida presented to the Emergency Department (ED) at 34 weeks’ gestation with complaints of one day decreased fetal movement and contractions. Upon bedside sonogram, IUFD was confirmed and the patient had a spontaneous vaginal delivery. Many of the cases of IUFD are associated with fetal growth restriction and chronic placental insufficiency. In contrast, this fetus demonstrated an accelerated growth pattern, suggesting the terminal event was likely consistent with an acute incident, rather than a chronic process. Given the maternal metabolic dysfunctions of PCOS and obesity that contributed to a hypercoagulable state, this raises consideration of an acute vascular compromise, such as a microthrombotic occlusion or abrupt placental perfusion failure. Microthrombi may form and later dissolve via fibrinolysis, and terminal thrombotic events occurring near the time of demise may leave minimal to no histopathologic evidence [3]. Another possibility may be perfusion failure without accompanying substantial tissue infarction.
Although MTHFR C677T heterozygosity alone is not considered to be an independent risk factor for pregnancy loss, this patient also exhibited a lower protein S activity and reduced prothrombin time/international normalized ratio (PT/INR) values. These abnormal lab values in addition to her underlying metabolic comorbidities, PCOS and obesity, both of which are associated with a chronic inflammatory state and increased thrombotic risk [4],[5], may indicate a clinical profile consistent with a subclinical prothrombotic state. The complex relationship of these factors may have potentially contributed to a transient placental microvascular compromise.
An additional consideration is the rapid increase in EFW following the initiation of maternal iron therapy. The fetus increased from the 63rd percentile at 28 weeks to the 92nd percentile at 33 weeks’ gestation, shortly after maternal iron supplementation was started at 30 weeks. While improved oxygen delivery and nutritional support to the fetus is generally beneficial, this observed accelerated growth may indicate an increase in metabolic and perfusion demand on the placenta. If the placenta were to already be functioning under suboptimal perfusion conditions, this may have exacerbated the issue, and the placenta may not have been able to withstand the demand. In a hypercoagulable and prothrombotic state, such as this case, even a transient microvascular thrombosis may have exacerbated the slight mismatch between fetal demand and placental capacity, ultimately leading to IUFD.
Lastly, the pregnancy was further complicated by early VTS, which may disrupt early placental development [6]. The grossly abnormal bilobed placental architecture (Figure 1) noted postpartum supports the consideration of disproportionate placental vascularization and development. Taken together, the maternal prothrombotic state, higher placental metabolic demand associated with the late accelerated fetal growth, and the structural abnormality of the placenta associated with early twin loss, may have contributed to an acute terminal perfusion event, resulting in IUFD. The maternal use of low-dose aspirin that was started secondary to preeclampsia risk factors may not have been adequate to prevent the underlying physiologic concerns in this case.
Conclusion
This case highlights the potential additive effect of metabolic dysfunctions and subclinical thrombophilia, as well as how they may contribute to placental perfusion compromise, even in the absence of histopathological abnormalities. In this patient, the physiologic pregnancy changes and low Protein S activity created a hypercoagulable state, which may have been further amplified by the prothrombotic and proinflammatory effects of obesity and PCOS. The MTHFR mutation may have served as a minor contributing factor to her total thrombotic risk profile, although it is not thought to be an independent cause of thrombosis.
This case may help serve as a guide for the management of similar pregnancies, underlining the significance of early detection of identified overlapping risk factors and early individualized thrombophilia screening, based on the patient’s presentation. For subsequent pregnancies, improved surveillance and risk-stratified management, including prophylactic anticoagulation in addition to antiplatelet therapy, may help reduce the risk of recurrent adverse pregnancy outcomes.
REFERENCES
1.
Stillbirth Collaborative Research Network Writing Group. Causes of death among stillbirths. JAMA 2011;306(22):2459–68. [CrossRef]
[Pubmed]

2.
Many A, Elad R, Yaron Y, Eldor A, Lessing JB, Kupferminc MJ. Third-trimester unexplained intrauterine fetal death is associated with inherited thrombophilia. Obstet Gynecol 2002;99(5 Pt 1):684–7. [CrossRef]
[Pubmed]

3.
Bonasoni MP, Muciaccia B, Pelligra CB, Goldoni M, Cecchi R. Third trimester intrauterine fetal death: Proposal for the assessment of the chronology of umbilical cord and placental thrombosis. Int J Legal Med 2022;136(3):705–11. [CrossRef]
[Pubmed]

4.
Dasanu CA, Clark BA 3rd, Ichim TE, Alexandrescu DT. Polycystic ovary syndrome: Focus on platelets and prothrombotic risk. South Med J 2011;104(3):174–8. [CrossRef]
[Pubmed]

5.
Lentz SR. Thrombosis in the setting of obesity or inflammatory bowel disease. Blood 2016;128(20):2388–94. [CrossRef]
[Pubmed]

6.
Li J, Li J, Zhang Y, Hu K, Chen N, Gao J, et al. The influence of the vanishing twin on the perinatal outcome of surviving singleton in IVF pregnancy. Front Endocrinol (Lausanne) 2022;13:832665. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Acknowledgments
We thank Nairmeen Awad Haller PhD, CHRC, the Corporate Senior Director for Research Operations of Community Health Systems for assistance and guidance in writing this case report.
Artificial intelligence (AI) use in the article
No artificial intelligence was used in the creation of this manuscript.
Author ContributionsBenjamin Perdomo - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Joslynn Soria - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Jeanette Toney - Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2026 Benjamin Perdomo et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.