|
Case Report
Postpartum deep septic pelvic thrombophlebitis after cesarean section: A case report
1 MBBS, Senior House Officer, Department of Obstetrics and Gynecology, Queen Elizabeth Hospital, St Michael, Barbados
Address correspondence to:
Keturah Murray
Elizabeth Hospital, Martindales Road, St Michael,
Barbados
Message to Corresponding Author
Article ID: 100226Z08KM2026
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Murray K. Postpartum deep septic pelvic thrombophlebitis after cesarean section: A case report. J Case Rep Images Obstet Gynecol 2026;12(1):22–26.ABSTRACT
Deep septic pelvis thrombophlebitis is a diagnosis that should be entertained in any patient who develops persistent fever postpartum which is unresponsive to antibiotic therapy. In this article, we report a case of a 42-year-old multiparous female who was developed a prolonged febrile illness postpartum following a caesarean section. Diagnosis of deep septic pelvis thrombophlebitis was suspected and confirmed using contrast enhanced computed tomography. She was managed conservatively and successfully treated with broad spectrum antibiotics and anticoagulation with low molecular weight heparin. She was discharged 17 days post-operatively, symptom free, with a course of extended anticoagulation therapy.
Keywords: Anticoagulation therapy, Postpartum, Septic pelvic thrombophlebitis
Introduction
Septic pelvic thrombophlebitis (SPT) is an uncommon infectious disease that occurs during the postpartum period and forms blood clots in the pelvic veins, with infection and inflammation [1]. Two entities have been described in the literature; deep septic pelvic thrombophlebitis (DSPT) and ovarian vein thrombosis (OVT) [2]. These may differ in clinical presentation and diagnostic findings, but they share the same pathogenic mechanisms and often occur together. Septic pelvic thrombophlebitis is an important differential diagnosis of abdominal pain and fever in the postpartum period [3]. Diagnosis can be challenging, since clinically, symptoms are non-specific; however, imaging has proven to be a useful tool in evaluation of these patients [4]. The cornerstone of management involves the dual approach of administration of anticoagulation and broad spectrum antibiotics [5].
Case Report
A 42-year-old multiparous woman, G3P1+1, underwent an emergency cesarean section at 37 weeks 4 days secondary to non-reassuring fetal status. She was admitted with prelabor rupture of membranes (PROM). A Foley catheter was inserted on arrival, 8.5 hours after membrane rupture. Prophylactic cefazolin 2 g was administered at the time of catheter placement. A cardiotocogram performed following catheter insertion was reassuring. The catheter was subsequently expelled spontaneously, after which she progressed into early labor and remained clinically stable. Approximately 8 hours after admission, she developed a fever of 38.7 °C and antibiotics (cefazolin, metronidazole, and gentamycin) were commenced to treat suspected chorioamnionitis. Upon review, she was noted to have a suspicious cardiotocogram trace. Her vaginal examination indicated that she was remote from delivery therefore birth was expedited by Cesarean section. The surgery was uneventful, with the delivery of a female infant weighing 2730 g, APGAR scores of 9 and 9 at 1 and 5 minutes. The delivery occurred 17 hours after ROM.
On postpartum day 2 the patient reported fever, chills, joint pain, and photophobia. She denied any abnormal lochia, respiratory, or urinary symptoms. Her vital signs included a temperature of 38.2 °C, she was tachycardic at 105 beats per minute, with a normal blood pressure. Her cardiovascular and respiratory examinations were normal. Her abdomen was soft with mild peri-incisional tenderness, the wound was healthy with no discharge. Lab investigations were significant for a neutrophilic leukocytosis of 14.5 K/uL. Intravenous piperacillin/tazobactam was commenced, cefazolin and gentamycin were discontinued. Imaging with both ultrasound and contrast enhanced computed tomography (CT) of the abdomen and pelvis was performed to evaluate for intra-abdominal source of the fever. However, the results of all these investigations were within normal limits. Blood and urine cultures were also sterile.
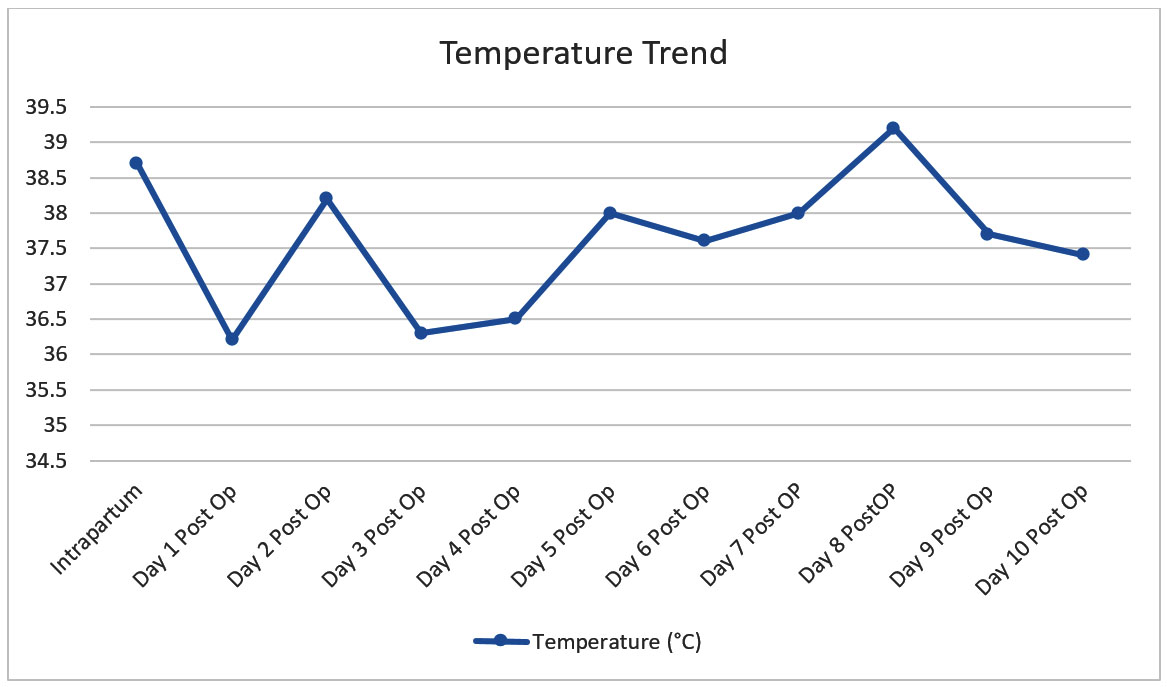
Despite broad spectrum antibiotic coverage, she was noted to be persistently febrile (Figure 1). Testing for coronoavirus disease (COVID-19) and Influenza were both negative. Repeat blood cultures were sterile and her laboratory investigations showed a persistent leukocytosis and normochromic normocytic anemia (Table 1 and Table 2). Given the above, the decision for repeat imaging was made. Contrast-enhanced CT abdomen and pelvis on day 7 postpartum revealed bilateral iliac filling defects. She was referred to hematology, and therapeutic anticoagulation was commenced with enoxaparin at a dose of 1 mg/kg Q12 hours. On advice of infectious disease specialty, the doses of metronidazole and piperacillin/tazobactam were increased from Q8 hourly to Q6 hourly. In spite of this, she was again febrile on day 8 postpartum; fluconazole was commenced to cover for possible fungal etiology. Following a temperature spike of 38 °C Day 10 postpartum, intravenous meropenem 1 g Q 8 hourly was started, and all other antibiotics and antifungals discontinued. There was good clinical response thereafter with gradual defervescence (Figure 2). She was discharged after completion of one week of meropenem to complete a total of six months of anticoagulation.
Discussion
Septic pelvic thrombophlebitis (SPT) is a rare, potentially life-threatening condition during puerperium, occurring in 1/2000 deliveries (1/9000 vaginal and 1/800 caesarean deliveries) [2]. There are two types of SPT, ovarian vein thrombophlebitis (OVT) and deep septic pelvic thrombophlebitis (DSPT) [3]. This was first described in the late 1800s by Von Recklinghausen, in a case series of women presenting with pelvic infection and ovarian vein thrombosis [4].
The pathophysiology of this disease is thought to involve Virchow’s triad (endothelial injury, venous stasis, and hypercoagulability). While SPT is not unique to the pregnancy and postpartum state, these time periods are an especially favorable host to these changes [5]. Endothelial damage can occur as a result of intrapartum trauma to vascular structures, as a result of uterine infection, or from pelvic surgery itself [6]. Venous stasis occurs as a result of pregnancy-induced ovarian venous dilatation and low postpartum ovarian venous pressures [7]. These adaptations, in addition to the hypercoagulable state of pregnancy, lead to increased propensity for developing complications such as SPT.
When compared with vaginal delivery, the risk is significantly higher with caesarean births. Additionally, peripartum or postpartum infections, such as chorioamnionitis, as was suspected in this patient, and endometritis increase the risk of SPT [8]. A multi-center cohort evaluation by Dotters-Katz et al. identified black race, young age, preeclampsia, and multiple gestation as other predisposing factors to developing SPT [8].
After the intrapartum fever, our patient next became febrile on day two postoperatively. Patients with DSPT usually present three to five days after delivery or surgery with an unfocalized fever, which may be the only symptom; that symptom commonly persists despite antibiotics. Patients appear clinically well between fever spikes and pain is usually absent [3]. Throughout the course of her admission, two sets of blood and urine cultures were done, both of which were sterile. Microbial cultures are rarely positive; a positive blood culture is almost an exception to the rule with an incidence below 3% [2].
Due to the prolonged length of her febrile illness, the decision was made for repeat imaging. The key to diagnosis of SPT is its inclusion in differential diagnosis [9]. Diagnostic workup involves an imaging study added to a high level of suspicion in those patients with the aforementioned risk factors [4]. Options include CT and magnetic resonance imaging (MRI). Both methods are superior to ultrasonography, CT being the standard technique and MRI gaining in popularity owing to the absence of ionizing radiation. Both methods have high sensitivity and specificity (contrast-enhanced CT 100% vs. 92% and 99% vs. 100%, respectively) [2].
Computed tomography findings comprise enlargement of the involved vein, enhancement or sharply defined vessel wall, perivascular inflammatory stranding, and a low-density filling defect of the gonadal vessel representing the thrombus [4]. It is important to note however that small uterine vein branches are difficult to visualize, thus, a negative imaging study cannot exclude SPT [3]. In this case, her initial CT scan did not show evidence of thrombosis, however, it was subsequently confirmed on repeat imaging.
Our patient was managed conservatively. Surgical removal of the clot has been reported as a treatment for SPT. However, recent reports suggest a shift from surgical approaches to those centered around antimicrobial and anticoagulation therapies due to advances in therapeutic drugs [1]. The selection of antibiotics for managing SPT is extrapolated from the literature on postpartum endometritis, since at the time when a presumptive diagnosis of SPT is made, most patients will have already been receiving broad-spectrum parenteral antibiotics to cover the common pathogens of endometritis. The duration of the antibiotic therapy is not strictly defined. The use of anticoagulants for the treatment of SPT is advocated by many authors in order to prevent further thrombosis and embolism and reduce the spread of septic emboli. However, there are no defined guidelines for anticoagulation therapy [3].
In the case of our patient, she did not initially respond to broad-spectrum antibiotics however leukocytosis resolved and defervescence was achieved with the administration of meropenem (Figure 2). We decided to maintain anticoagulation for six months after discharge.
Recurrence is possible [2]. Postpartum complications such as SPT, increase the risk of thrombosis in subsequent pregnancies. According to Royal College of Obstetricians and Gynaecologists (RCOG), women with a prior venous thromboembolic event, antenatal prophylaxis with low molecular weight heparin is indicated and should be continued for at least six weeks postnatally [10]. This treatment would be indicated for our patient should she desire future pregnancies.
Pulmonary embolism of septic emboli is a known complication. Others include migration and spread of thrombosis to abdominal veins and veins of lower extremities. Mortality is very low (2%), mainly attributed to systemic spread of infection or to septic emboli to the lungs [2].
Conclusion
Septic pelvic thrombophlebitis is a frequently misdiagnosed condition due to its nonspecific symptomatology. A high level of clinical suspicion is required and should be considered for postpartum patients presenting with persistent fever despite antibiotics. Early diagnosis and intervention are essential for successful management of these patients.
REFERENCES
1.
Miyamori D, Shigemoto N, Une K, Kinoshita H, Harimoto S, Sakashita T, et al. Delayed onset septic pelvic thrombophlebitis treated by tissue-plasminogen activator following initial treatment for massive right ovarian vein thrombosis and methicillin-resistant Staphylococcus aureus bacteremia: A case report. J Obstet Gynaecol Res 2024;50(8):1408–14. [CrossRef]
[Pubmed]

2.
Kozar S, Šavc H. Deep septic pelvic thrombophlebitis – A life-threatening condition in postpartum period. Acta Clin Croat 2021;60(4):773–6. [CrossRef]
[Pubmed]

3.
da Silva Cunha M, Godinho AB, Botelho R, de Almeida JP. Postpartum septic pelvic thrombophlebitis after caesarean delivery: A case report. Case Rep Womens Health 2018;17:5–7. [CrossRef]
[Pubmed]

4.
Roepke RML, de Campos FPF, Lovisolo SM, Santos EHS. Septic pelvic thrombophlebitis of unknown origin: An ever threatening entity. Autops Case Rep 2014;4(3):39–46. [CrossRef]
[Pubmed]

5.
Barnes AV, Noman R, Shakfeh S. Beyond postpartum fever: Case report uncovering deep septic pelvic thrombophlebitis. Cureus 2023;15(8):e43034. [CrossRef]
[Pubmed]

6.
Collins CG. Suppurative pelvic thrombophlebitis. A study of 202 cases in which the disease was treated by ligation of the vena cava and ovarian vein. Am J Obstet Gynecol 1970;108(5):681–7.
[Pubmed]

7.
Hodgkinson CP. Physiology of the ovarian veins during pregnancy. Obstet Gynecol 1953;1(1):26–37.
[Pubmed]

8.
Dotters-Katz SK, Smid MC, Grace MR, Thompson JL, Heine RP, Manuck T. Risk factors for postpartum septic pelvic thrombophlebitis: A multicenter cohort. Am J Perinatol 2017;34(11):1148–51. [CrossRef]
[Pubmed]

9.
French RA, Cole C. An “enigmatic” cause of back pain following regional anaesthesia for caesarean section: Septic pelvic thrombophlebitis. Anaesth Intensive Care 1999;27(2):209–12. [CrossRef]
[Pubmed]

10.
Reducing the risk of thrombosis and embolism during pregnancy and the puerperium (green-top guideline No. 37a) [Internet]. RCOG. [cited 2025 Nov 13]. [Available at: https://www.rcog.org.uk/guidance/browse-all-guidance/green-topguidelines/reducing-the-risk-of-thrombosis-andembolism-during-pregnancy-and-the-puerperium-green-top-guideline-no-37a/]

SUPPORTING INFORMATION
Author Contributions
Keturah Murray - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthor declares no conflict of interest.
Copyright© 2026 Keturah Murray. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.